Clear Sky Science · en
Spatial pattern separation deficits in early Alzheimer’s disease are comparable in humans and animal models
A Closer Look at Subtle Memory Changes
Many people worry about occasionally misplacing objects or forgetting where they parked the car. This study digs into a very specific kind of "where was it?" memory that falters early in Alzheimer’s disease, long before full-blown dementia. By testing both older adults and specially bred rats with nearly identical tasks, the researchers show that a precise kind of spatial memory breaks down in the earliest stages of the disease, and that this breakdown looks remarkably similar across species. 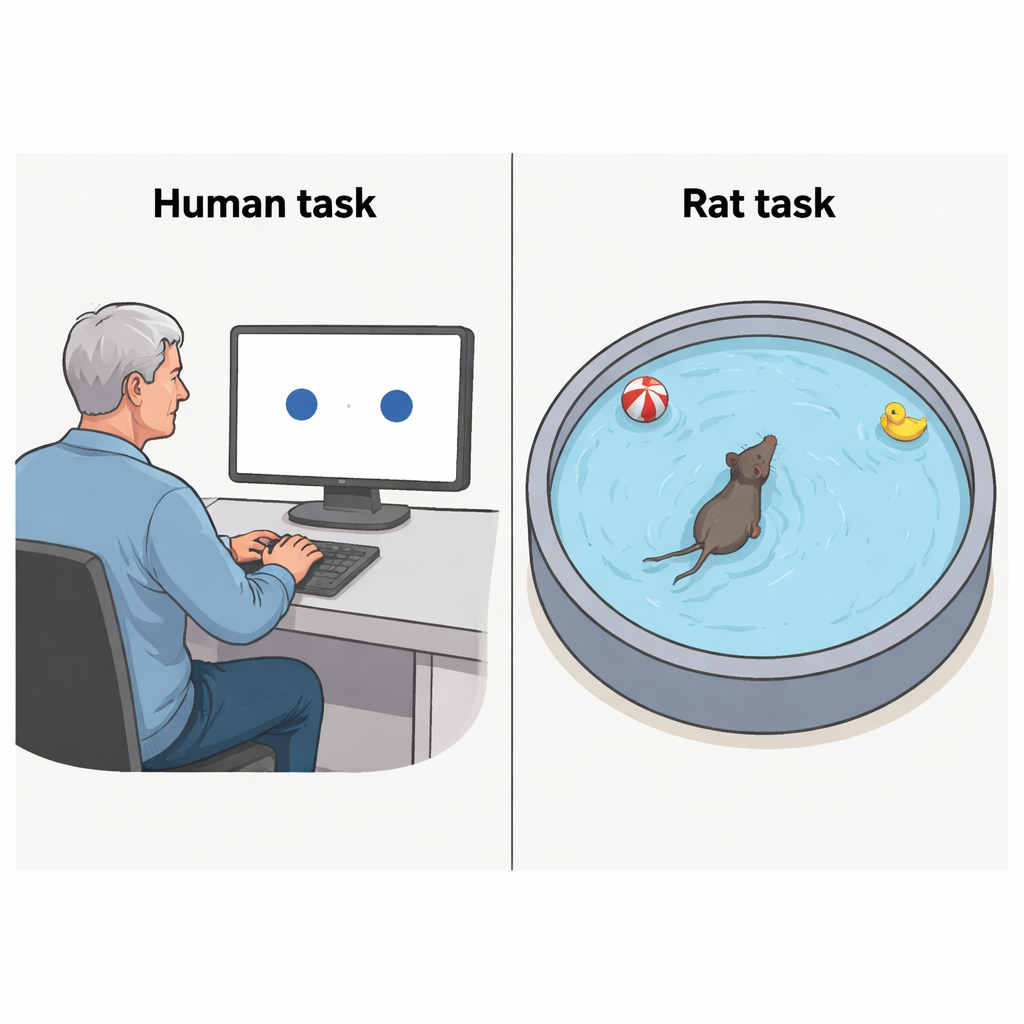
Telling Apart Very Similar Places
The work centers on “spatial pattern separation,” a process our brains use to distinguish between locations that are almost, but not quite, the same—like two neighboring parking spots. To study this in humans, the team used a simple computer task. Older volunteers saw a single blue circle on a screen and were asked to remember its exact position. After a short delay, two identical circles appeared side by side, very close together. One was in the original position, the other slightly shifted. Participants pressed a button to choose which circle was in the right spot. The researchers varied how far apart the circles were, from just touching edges to a modest distance, and measured how often people chose correctly.
Early Alzheimer’s Leaves a Distinct Footprint
The study compared 56 older adults with memory problems caused by early Alzheimer’s disease (confirmed by brain scans and spinal fluid tests) to 60 cognitively healthy peers. Those with early Alzheimer’s were less accurate overall, and everyone did better when the circles were farther apart. Crucially, the early Alzheimer’s group still showed this same pattern: shrinking the distance made the task harder for both groups, but the affected group performed consistently worse at every distance. These differences remained even after the researchers adjusted for performance on standard memory tests, suggesting that this is not just “being more forgetful,” but a specific difficulty in teasing apart similar locations.
Inside the Brain’s Navigation Hubs
To find out what might be going wrong in the brain, the team examined MRI scans from most of the human participants. They focused on structures deep in the temporal lobes that are known to support navigation and fine-grained memory for places. People with smaller volumes in the back portion of the hippocampus and in a rear subsection of the entorhinal cortex tended to do worse on the spatial task. A small region in the basal forebrain, which sends chemical signals that help tune these memory circuits, also mattered: when it was shrunken, performance on the task suffered. Interestingly, the amount of amyloid protein seen on brain scans—often highlighted in Alzheimer’s research—did not clearly track with how well people performed on this spatial discrimination test.
Rats in a Water Maze Tell a Similar Story
To test whether the same kind of spatial problem appears in an animal model, the researchers adapted the idea for rats using a version of the well-known Morris water maze. In this setup, rats learn the fixed location of a hidden platform in a circular pool, guided by visual cues placed around the room. On special test trials, the rats swam with either one or two prominent cues placed at different angles relative to the learned platform location. For one group, the cues were separated by 90 degrees—closer and more easily confused. For another, they were 180 degrees apart on opposite sides of the pool. Young adult rats carrying Alzheimer’s-like gene changes were compared with normal rats. Both groups learned the basic platform location equally well, showing that overall spatial memory was still intact. Yet, when the cues were only 90 degrees apart, the Alzheimer’s-model rats swam less precisely toward the correct area in the first few seconds of the trial, indicating a difficulty separating nearby locations. This difference largely disappeared when the cues were a full 180 degrees apart and easier to tell apart. 
Bridging Lab Animals and Human Patients
Because the human computer task and the rat water-maze task were built around the same core idea—choosing between very similar locations—the researchers could directly compare how well each test distinguished early Alzheimer’s from normal aging. In both species, the tasks had similar power to separate affected from unaffected individuals. That makes spatial pattern separation a promising “translational” measure: scientists can use the same underlying concept to track how experimental treatments change brain function in animals and then apply parallel tests in human trials. Over time, this could help close the gap between drugs that look promising in the lab and those that actually improve early cognitive problems in people.
What This Means for Everyday Life
For non-specialists, the key takeaway is that not all memory failures in Alzheimer’s look the same. This study shows that the disease disrupts a very precise ability to tell apart close-by places, and that this can be detected with relatively simple, noninvasive tests. Because the same kind of breakdown appears in people and in well-characterized animal models before broader memory collapses, these spatial tests could become valuable early warning tools and sensitive readouts for new treatments aimed at protecting the brain’s navigation circuits.
Citation: Laczó, M., Maleninska, K., Khazaalova, N. et al. Spatial pattern separation deficits in early Alzheimer’s disease are comparable in humans and animal models. Sci Rep 16, 6020 (2026). https://doi.org/10.1038/s41598-026-36266-y
Keywords: Alzheimer’s disease, spatial memory, pattern separation, hippocampus, translational neuroscience