Clear Sky Science · en
Genetic diversity in the Plasmodium falciparum next-generation blood stage vaccine candidate antigen PfCyRPA in Senegal
Why this malaria study matters
Malaria still kills hundreds of thousands of people every year, most of them in Africa and many of them children. The vaccines now being rolled out save lives, but their protection fades and is not complete. This study looks at a new vaccine target used by the malaria parasite to break into red blood cells. By checking how much this target varies in real infections in Senegal, the researchers ask a key question for future vaccines: is this a stable “Achilles’ heel” of the parasite, or a moving target?
A new bullseye on the parasite
When malaria parasites invade the bloodstream, they must slip into red blood cells to multiply. To do this, they use a small set of proteins that lock onto the cell’s surface, much like a key fitting into a lock. One of these proteins, called PfCyRPA, works together with partners PfRh5 and PfRipr in a tightly linked invasion complex. Because parasites cannot invade without this complex, and because PfCyRPA appears to change little from strain to strain, it has become a leading candidate for next-generation blood-stage vaccines now entering early clinical trials. A vaccine that targets such an essential and stable protein could block disease across many parasite strains and regions.
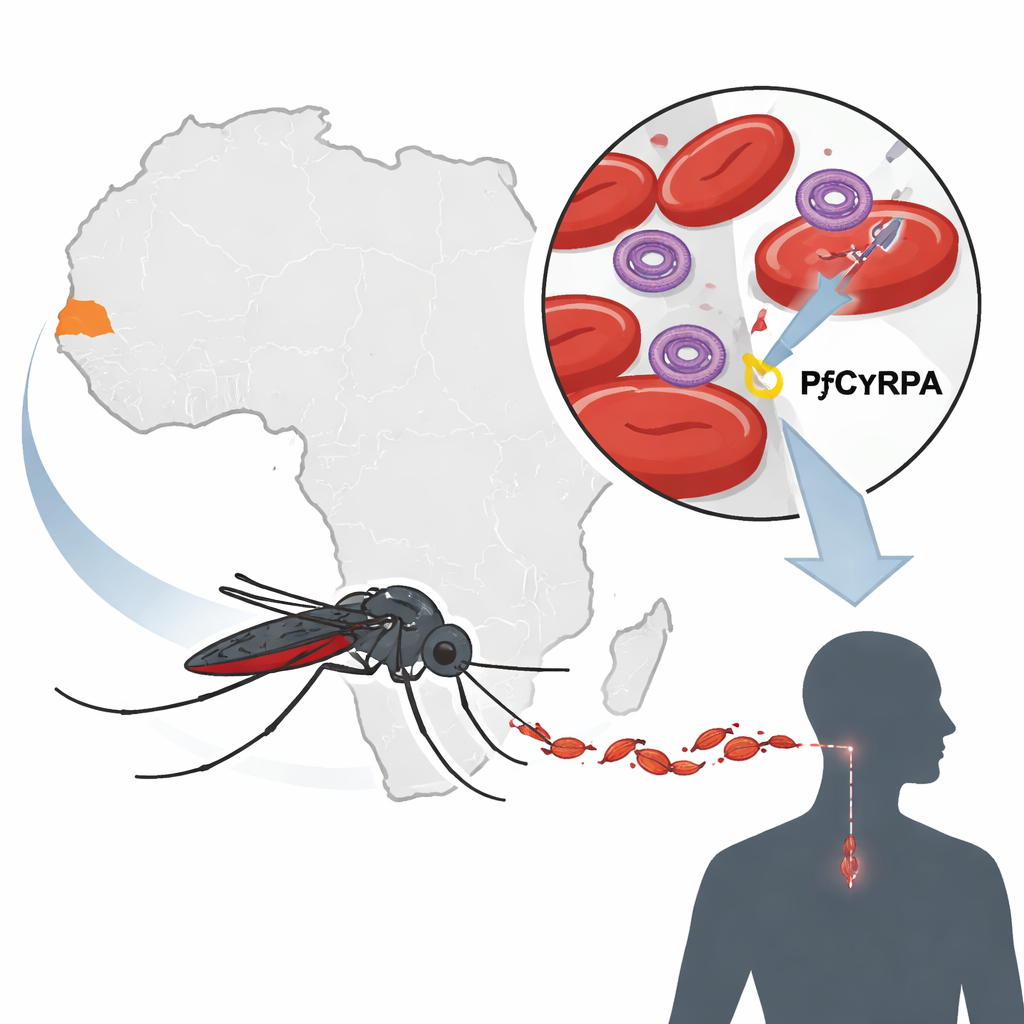
Taking a genetic snapshot in Senegal
To see how stable this protein really is in nature, the team studied 93 malaria infections from patients in Kédougou, a region in southeastern Senegal with intense seasonal transmission. Many patients carried several parasite strains at once, which makes it hard to spot rare genetic changes. To overcome this, the researchers used deep sequencing, a high-throughput method that reads the PfCyRPA gene thousands of times per sample and can detect even low-level variants. They then compared every sequence to the standard laboratory parasite strain, known as 3D7, and catalogued single-letter DNA changes that alter the protein’s building blocks.
Few changes and mostly rare ones
The results show that the reference version of PfCyRPA dominates this parasite population: about 72% of infections carried only the standard form of the gene. Overall, the scientists found just 15 distinct protein-changing mutations, and two-thirds of them appeared in only a single infection each. Only one change, called V292F, reached close to 10% of infections. By measuring how often each mutation appeared among the many parasite genomes within a single patient, they showed that most of these changes were present at high frequency in the infections where they occurred. This suggests that, when a variant does arise, it can become the main version within that infection—but such variants remain uncommon across the community.
What the structure reveals about function
Genetic differences matter for vaccines only if they change how the protein folds, how it works, or how antibodies see it. To explore this, the team used 3D structures of PfCyRPA bound to its partner PfRh5 and to several human antibodies. They “threaded” each mutation onto these structures and used computer tools to predict the effects on shape, stability, and binding. Most mutations were predicted to have only minor consequences for PfCyRPA’s overall form or its contacts with PfRh5 and known antibodies. A few changes, such as D236V and N270T, may subtly affect flexibility or stability by disrupting hydrogen bonds or creating small clashes in the protein. Others, including R50C and F187L, sit close to the contact area with PfRh5 and could tweak that interaction. A small set of mutations lies near regions where strongly blocking antibodies tend to bind, but even there the predicted effects on antibody recognition were mild.
What this means for future vaccines
For non-specialists, the key message is that this crucial parasite protein appears remarkably stable in a real-world, high-transmission setting: most parasites carry the same version, and the rare variants that do occur are predicted to only slightly change the protein’s behavior or how antibodies bind to it. That makes PfCyRPA a promising target for durable malaria vaccines. At the same time, the work highlights a handful of rare changes that could, in principle, give the parasite an edge under vaccine pressure. Pinpointing these sites now allows scientists to design vaccine candidates and lab tests that keep them in mind, helping ensure that tomorrow’s blood-stage vaccines remain effective even as the parasite continues to evolve.
Citation: Ba, A., Thiam, L.G., Pouye, M.N. et al. Genetic diversity in the Plasmodium falciparum next-generation blood stage vaccine candidate antigen PfCyRPA in Senegal. Sci Rep 16, 5661 (2026). https://doi.org/10.1038/s41598-026-36257-z
Keywords: malaria vaccines, Plasmodium falciparum, PfCyRPA, genetic diversity, Senegal