Clear Sky Science · en
A repurposing Dapagliflozin via polymeric nanogels for colorectal cancer therapy
Why a diabetes drug might help fight colon cancer
Colorectal cancer is one of the leading causes of cancer-related deaths worldwide, and many patients still relapse despite surgery and chemotherapy. This study explores an unexpected ally against this disease: dapagliflozin, a common pill for type 2 diabetes. By packaging this drug into tiny soft particles that turn into a gentle gel in the stomach, the researchers aim to deliver more of it to the large intestine—where colon tumors grow—while keeping the dose taken by mouth similar to existing tablets.
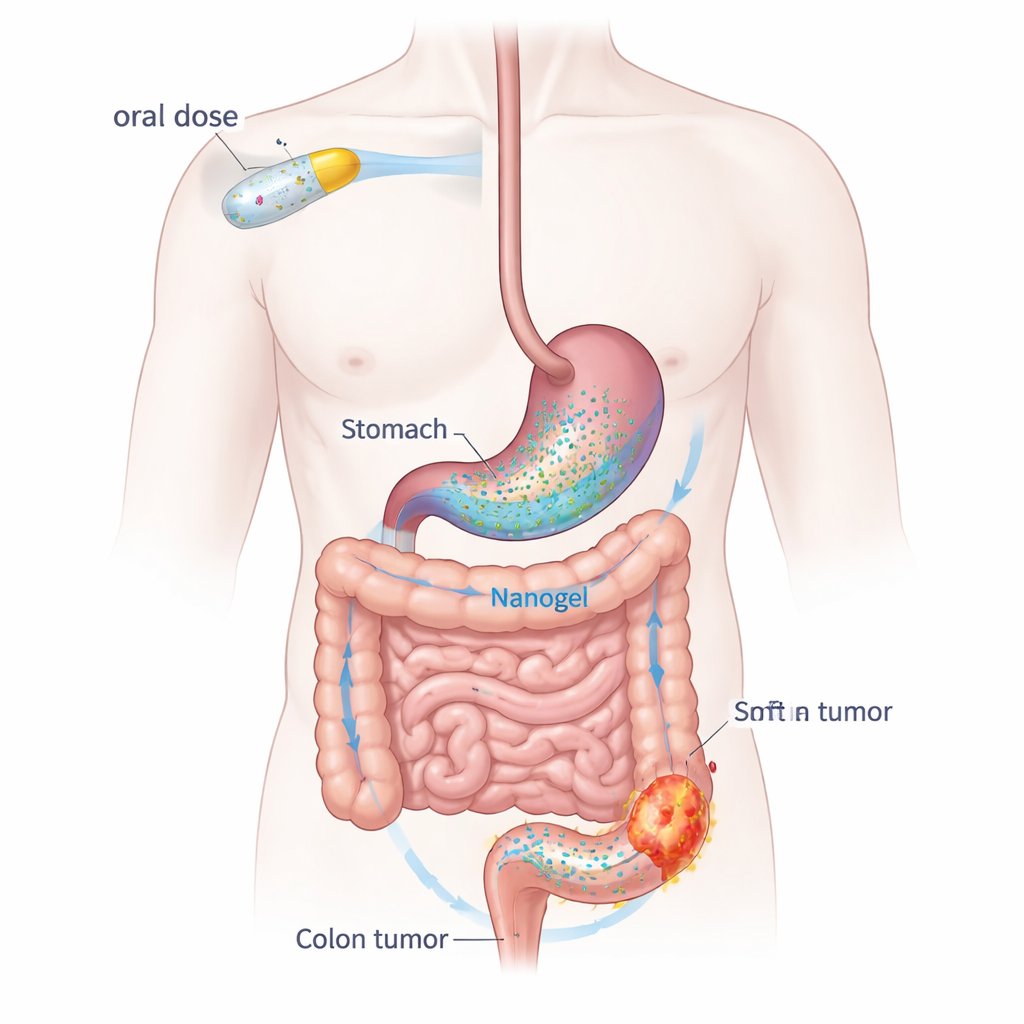
Turning an everyday pill into a cancer-targeting payload
Dapagliflozin normally works in the kidneys to lower blood sugar, but it also calms inflammation and slows cell growth—two processes that cancers rely on. On its own, however, the drug dissolves poorly in water and is cleared from the body quite quickly. To overcome this, the team built a “nanogel” delivery system using two food-like polymers: sodium alginate, extracted from seaweed, and polyvinyl alcohol, a widely used, biocompatible thickener. The drug is trapped inside nanoparticles about one ten-thousandth the width of a millimeter. When these particles encounter stomach acid, they settle and knit together into a soft hydrogel mass that lingers, then gradually releases the drug as it travels toward the colon.
Making the drug more soluble and slower to wash out
The scientists first optimized how the particles were made, fine-tuning the ratio of the two polymers so the nanoparticles stayed small, evenly sized, and stable in liquid. They confirmed that the drug was well-embedded in the polymer network and partially transformed from a crystalline to a more amorphous form, which generally dissolves better. In test liquids that mimic stomach and intestinal fluids, the nanogel formulation increased dapagliflozin’s apparent solubility by about 1.7–1.8 times compared with the raw drug. When they monitored how quickly the drug leaked out, the nanogel showed a gentle “slow-release” profile over several hours, rather than the rapid burst seen with the plain drug. One particular recipe, called F2, formed a compact, durable gel that held together for at least two hours in acidic conditions while steadily releasing the medicine.
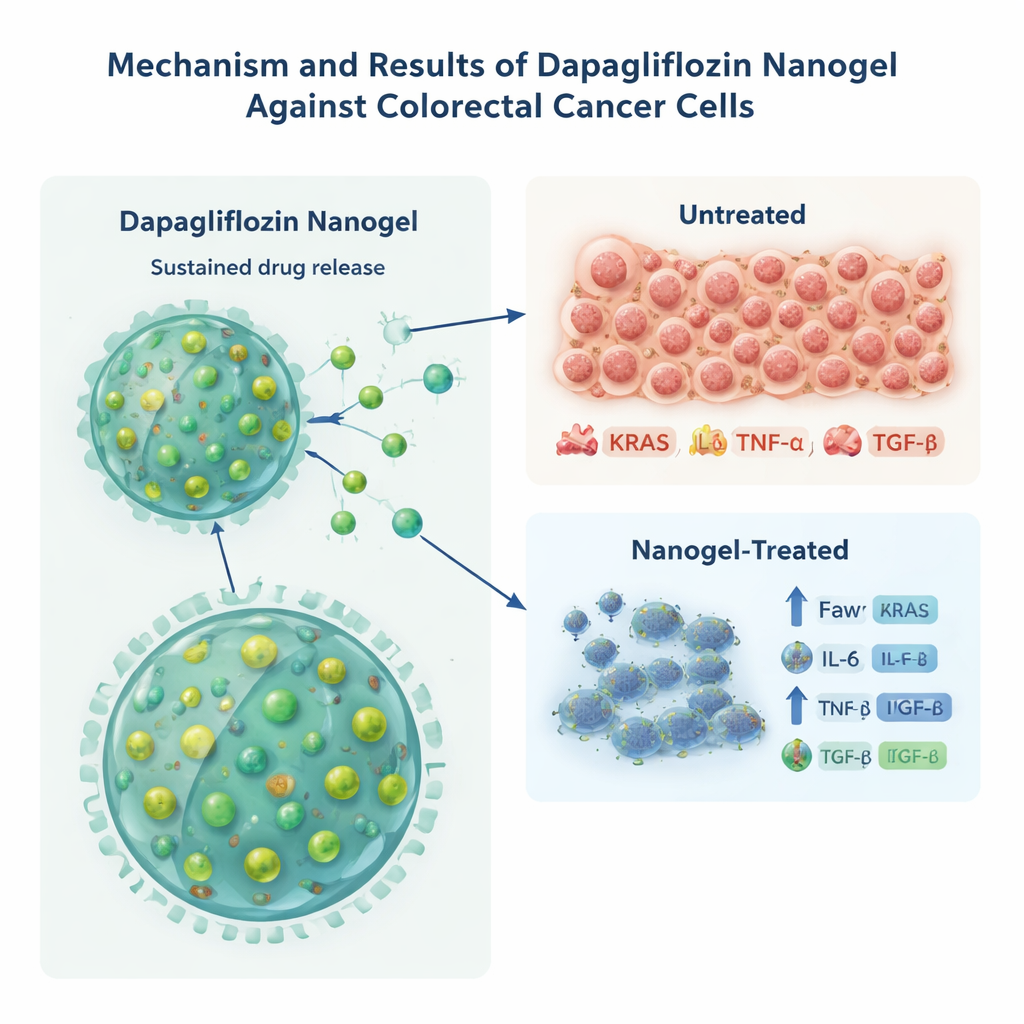
Putting cancer cells under stress in the lab
To see whether this new form of dapagliflozin could actually harm cancer cells, the team tested it on HCT-116 colorectal cancer cells grown in lab dishes. Compared with the same amount of free drug, the nanogel version killed cancer cells at lower concentrations, cutting the dose needed to halve cell growth by about one third. The researchers then looked at molecules linked to aggressive tumor behavior and chronic inflammation, including KRAS (a key cancer-driving protein) and the signaling proteins IL-6, TNF-α, and TGF-β. Cells treated with the drug-loaded nanogel showed much lower levels of all these markers than cells treated with the free drug or with empty particles, suggesting that sustained exposure from the nanoparticles more effectively dialed down cancer- and inflammation-related signals. Because the cell line already carries a fixed KRAS mutation, the authors caution that this drop in KRAS protein might reflect stress responses rather than direct genetic correction, but it still supports a strong biological impact.
Changing how the body handles the medicine
The team then moved to animal studies, giving rats either a simple suspension of dapagliflozin powder or the optimized nanogel, both by mouth at the same dose. In the nanogel group, peak drug levels in the blood appeared later and were lower, but the drug stayed in circulation for longer. The overall exposure (measured as area under the curve) rose modestly by about 7%, and the apparent half-life roughly doubled. These changes mean the body sees a smoother, more extended dose over time instead of a sharp spike and quick decline. That behavior matches what the researchers designed the gel to do: hold the drug in the gut, release it slowly, and potentially keep higher levels of it bathing the lower intestine and colon where tumors arise.
What this could mean for future cancer care
In everyday terms, this study takes a well-known diabetes drug and outfits it with a new “delivery suit” so it can better reach and linger near colon tumors. The nanogel makes the drug easier to dissolve, slows its escape from the gut, and appears to make it more toxic to colorectal cancer cells while dialing down key inflammation and growth signals. In rats, it gently stretches out the drug’s presence in the bloodstream without dramatically increasing the total dose. The work is still at an early stage—limited to one cancer cell line and short-term animal testing—and has not yet shown direct tumor shrinkage in living animals or people. Even so, it lays a detailed foundation for repurposing dapagliflozin as part of future colorectal cancer treatments, using smart oral nanogels to turn a familiar pill into a more targeted anticancer tool.
Citation: Abdullah, S., Thiab, S., Altamimi, A.A. et al. A repurposing Dapagliflozin via polymeric nanogels for colorectal cancer therapy. Sci Rep 16, 5625 (2026). https://doi.org/10.1038/s41598-026-36232-8
Keywords: colorectal cancer, drug repurposing, nanoparticles, oral drug delivery, dapagliflozin