Clear Sky Science · en
Nationwide genomic surveillance of carbapenem- and colistin-resistant Klebsiella pneumoniae bloodstream isolates in Thailand (2020–2024)
Deadly Blood Infections on the Rise
Hospitals around the world are grappling with a disturbing trend: common bacteria that once bowed to our strongest antibiotics are becoming nearly impossible to treat. This study focuses on Klebsiella pneumoniae, a microbe that can invade the bloodstream and cause life-threatening infections, and tracks how it has evolved across Thailand to resist even last-resort drugs. Understanding where these superbugs are spreading and how they evade medicines is crucial for protecting patients, guiding doctors’ choices, and shaping public health policy.
Tracking a Hospital Superbug Across a Nation
The researchers analyzed 227 Klebsiella pneumoniae samples taken from the blood of patients in hospitals across Thailand between 2020 and 2024. These samples came from a nationwide surveillance network that routinely collects data on drug-resistant infections. Laboratory tests showed that over 93% of these bloodstream isolates were resistant to carbapenems, a powerful class of antibiotics often used when other drugs fail. Worryingly, nearly one third of the carbapenem-resistant strains were also resistant to colistin, one of the last options doctors turn to when nothing else works. By pairing standard drug tests with whole-genome sequencing, the team could see not only which drugs failed, but also which genetic tricks the bacteria used to survive.
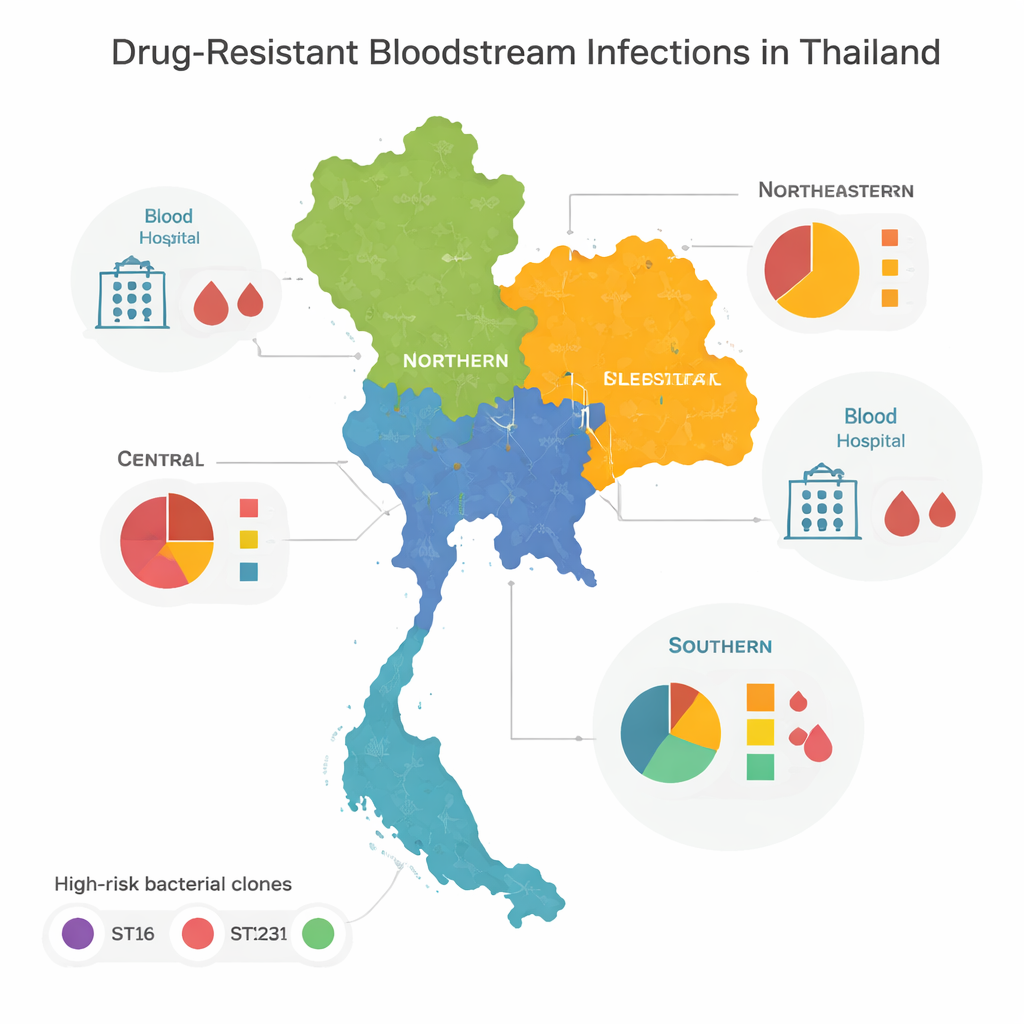
High-Risk Clones and Regional Patterns
When the team compared the genomes, they found that most of the resistant bacteria belonged to a few “high-risk” families, or clones, known globally for causing hospital outbreaks. Three in particular—called ST16, ST147, and ST231—dominated in central, northern, and northeastern Thailand. These clones had spread broadly across regions, suggesting frequent movement of patients, staff, or bacteria between hospitals. The southern part of the country looked different: instead of being dominated by a few types, it harbored a wide variety of bacterial lineages, many seen only once. This diversity hints at a more complex mix of local outbreaks, environmental sources, and cross-border movement rather than one or two runaway clones.
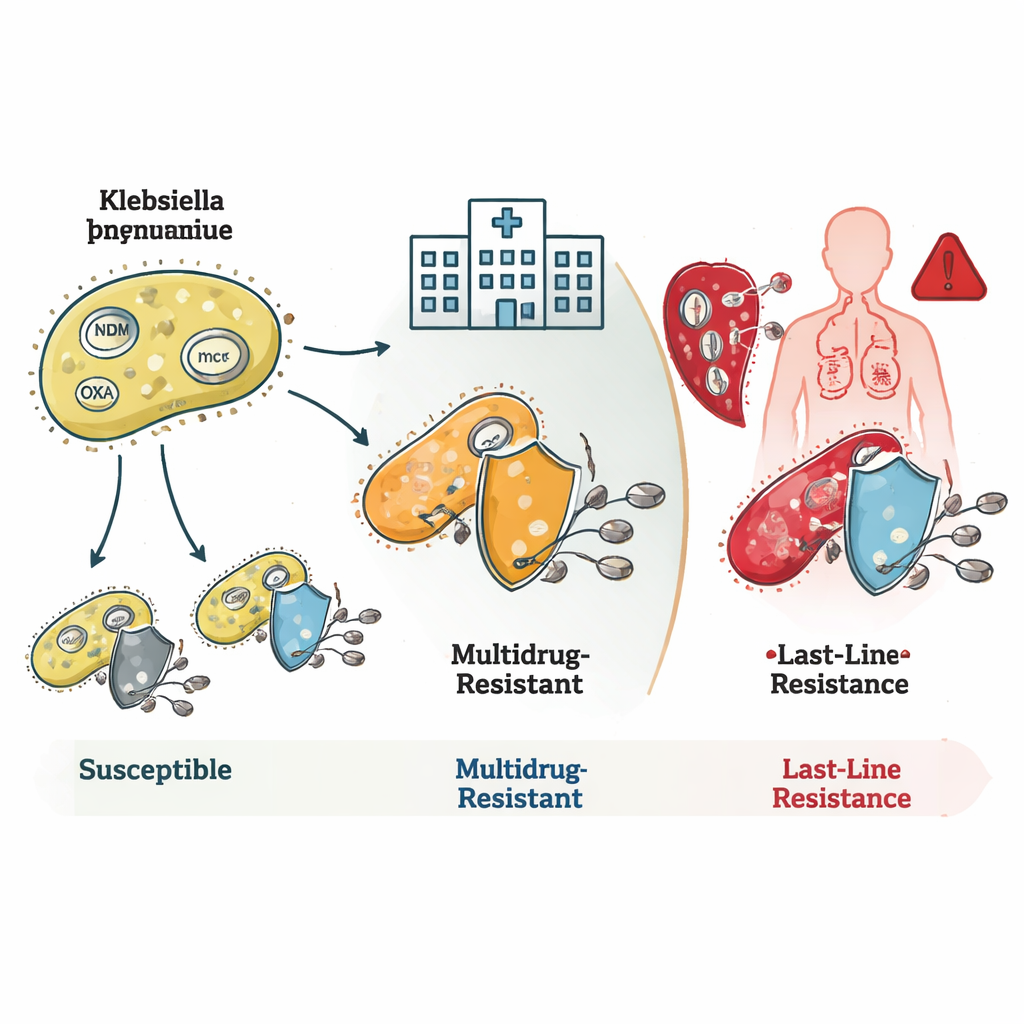
How the Bacteria Outsmart Antibiotics
The study revealed how these bacteria stack multiple resistance mechanisms. Many ST16 and ST231 strains carried genes known as blaNDM and blaOXA, which produce enzymes that break down carbapenem antibiotics before they can act. Some also had changes in their outer membrane that reduce drug entry, combining to create very high-level resistance. ST231 strains often carried an additional gene, rmtF1, that blocks an important group of drugs called aminoglycosides, further narrowing treatment options. ST147 stood out for its genetic flexibility: it mixed and matched different resistance genes and plasmids—small, mobile DNA rings—making it a potential hub for spreading resistance traits to other bacteria.
Last-Line Failures and Hidden Dangers
The most alarming finding involved resistance to colistin, a drug doctors reserve for desperate situations. Some rare bacterial families carried mcr genes, which can jump between species on plasmids and directly cause colistin resistance. Others, especially within the ST16 clone, were resistant to colistin even though they lacked any known resistance genes or mutations. This suggests that scientists have not yet fully mapped all the ways bacteria can neutralize this last-line drug. The team also identified hypervirulent strains—bacteria that are especially good at causing serious disease—including one lineage that combined high virulence with resistance to all tested carbapenems, a particularly dangerous mix.
Why Genomic Surveillance Matters
To understand how genetic changes translated into real-world drug failure, the researchers used statistical models to link specific resistance genes to actual lab test results. Genes such as blaNDM-1 and its close relatives showed extremely strong associations with carbapenem resistance, confirming their central role in driving treatment failure. Yet for colistin, most resistant bacteria had no recognized resistance markers, pointing to hidden mechanisms still to be discovered. The authors argue that routine use of whole-genome sequencing in hospital surveillance can act like an early-warning radar, spotting emerging high-risk clones, tracking their movement across regions and borders, and guiding rapid infection-control responses.
What This Means for Patients and Policy
For a layperson, the message is stark but clear: some bloodstream infections in Thailand are now caused by bacteria that resist nearly every drug doctors can throw at them, and a few dangerous families of these microbes are spreading widely. At the same time, the study shows that with the right tools—nationwide sampling, genetic analysis, and rapid data sharing—health systems can see these threats coming and adapt. Expanding genomic surveillance, developing quick molecular tests, and tightening infection control could help slow the advance of these superbugs and buy time for new treatments and vaccines to be developed.
Citation: Krobanan, K., Kamjumphol, W., Leethongdee, P. et al. Nationwide genomic surveillance of carbapenem- and colistin-resistant Klebsiella pneumoniae bloodstream isolates in Thailand (2020–2024). Sci Rep 16, 5853 (2026). https://doi.org/10.1038/s41598-026-36228-4
Keywords: antimicrobial resistance, Klebsiella pneumoniae, bloodstream infection, genomic surveillance, Thailand healthcare