Clear Sky Science · en
Aberrantly expressed long noncoding RNAs in adipose-derived mesenchymal stem cells differentiation to nucleus pulposus-like cells
Why Back Pain and Hidden RNA Matter
Low back pain is one of the leading reasons people miss work or visit a doctor, and a major culprit is the slow breakdown of the soft cushions between our spinal bones, known as intervertebral discs. Inside each disc sits a gel-like core called the nucleus pulposus, which depends on a healthy population of specialized cells to keep it springy and well hydrated. As these cells are lost or grow old, the disc dries, collapses, and can trigger chronic pain. Scientists are exploring whether stem cells—versatile cells that can turn into many tissues—could be guided to replace these worn-out disc cells. This study asks a surprisingly basic question: which “silent” genetic signals switch on when stem cells are coaxed to become new disc cells, and how might those signals be harnessed to improve future therapies?
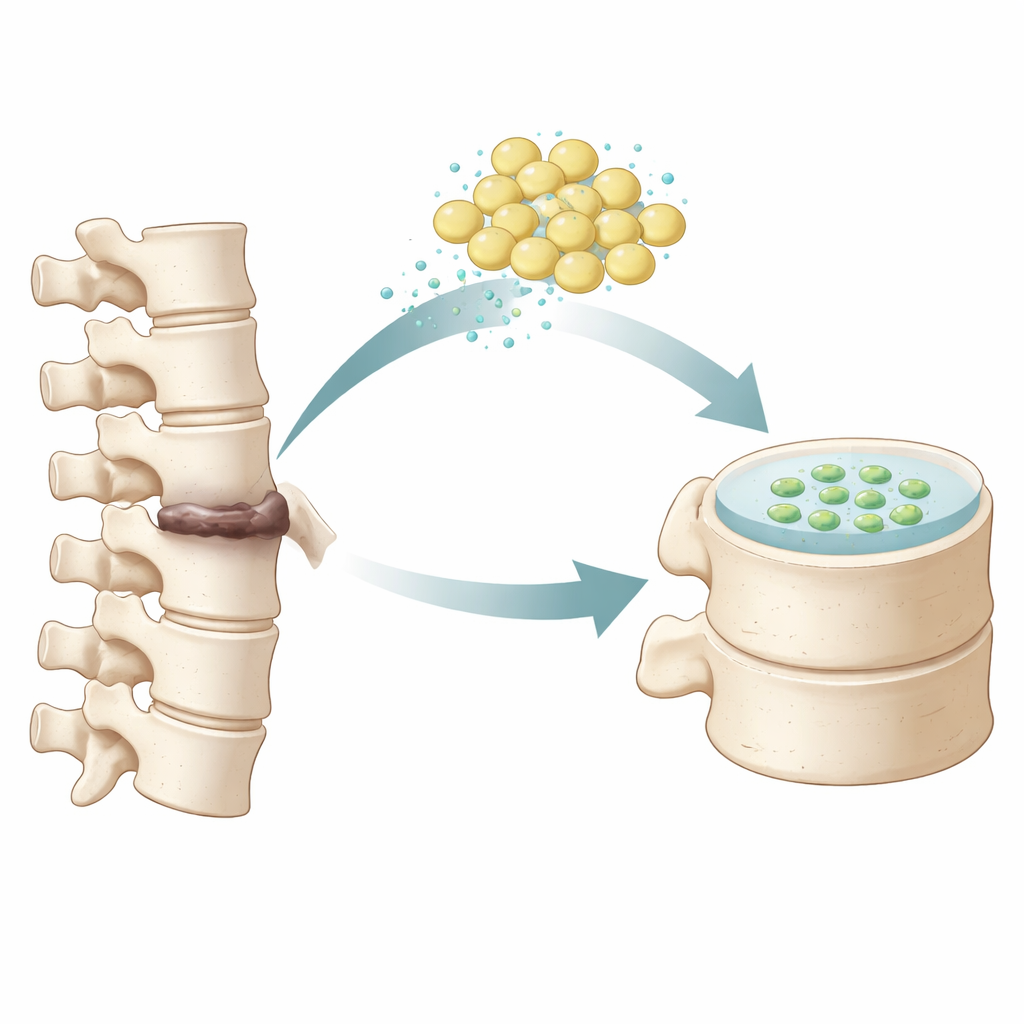
From Fat Tissue to Disc Repair
The researchers focused on human adipose-derived mesenchymal stem cells, which are collected from body fat and are relatively easy to obtain. In the laboratory, they grew these cells in small three-dimensional pellets and bathed them in a carefully chosen mix of growth factors that earlier work had shown could nudge cells toward a nucleus pulposus-like identity. Over several weeks, the team monitored the pellets with standard microscopic stains and fluorescent tagging. The treated cells began to look and behave more like the gel-core cells of a healthy disc, producing hallmark components of disc tissue such as collagen and aggrecan, and switching on key genes associated with nucleus pulposus identity.
Listening in on the Cell’s “Background Chatter”
Beyond the familiar protein-coding genes, cells also produce long noncoding RNAs—stretches of RNA that do not make proteins but can subtly steer which genes are turned on or off. These molecules are emerging as important conductors in the orchestra of cell differentiation. Using high-throughput RNA sequencing, the team measured both traditional messenger RNAs and long noncoding RNAs in stem cells undergoing disc-like differentiation, and compared them to undifferentiated control cells. They found extensive remodeling of the genetic landscape: 500 long noncoding RNAs and 601 messenger RNAs changed their activity levels, with some becoming more abundant and others dialed down as the cells shifted toward a nucleus pulposus-like state.
Key Pathways and Master Regulators
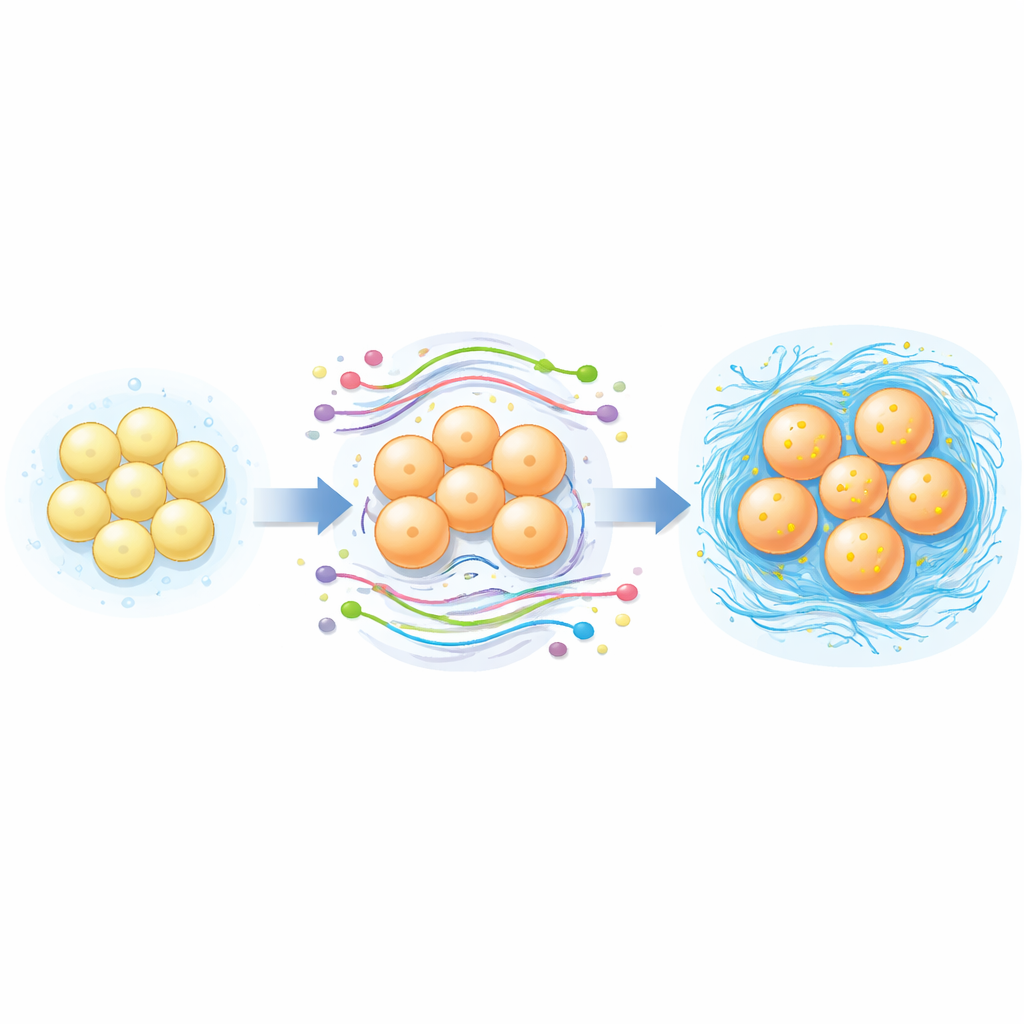
To make sense of this large list of changing molecules, the scientists used bioinformatic tools that group genes by their roles in the cell. Many of the altered genes were tied to building and organizing the extracellular matrix—the meshwork of collagen, sugars, and other molecules that gives disc tissue its cushioning properties. Pathway analysis highlighted the PI3K–Akt signaling route and systems that govern the cell’s internal skeleton as particularly active during differentiation, hinting that these circuits help drive the transformation from fat-derived stem cells to disc-like cells. By constructing interaction networks, the team pinpointed a set of long noncoding RNAs, including MALAT1, MEG3, GAS5, ZNF331, and JARID2-linked RNAs, that appear to sit at the hub of communication between regulatory RNAs, messenger RNAs, and microRNAs. These hubs may act as master switches controlling how efficiently stem cells commit to a disc-like fate.
Clues for Better Stem Cell Therapies
Intervertebral disc degeneration presents an especially harsh environment—acidic, low in oxygen, and rich in inflammatory molecules—that can thwart transplanted stem cells. By mapping which RNAs and pathways change during the successful laboratory conversion of fat-derived stem cells into nucleus pulposus-like cells, this study offers a catalog of molecular targets that might be tuned to boost survival and performance of therapeutic cells in real patients. Although the work was done in vitro and in a limited number of samples, it lays groundwork for future experiments that could, for example, tweak specific long noncoding RNAs or signaling pathways to enhance disc regeneration.
What This Means for People with Back Pain
For people living with chronic low back pain, these findings will not change treatment tomorrow, but they help fill in a crucial missing piece: how stem cells learn to become the exact type of disc cells needed for repair. The study shows that this transformation is guided not just by well-known genes, but also by a complex network of long noncoding RNAs and signaling pathways that shape the cell’s behavior. By decoding this hidden layer of control, scientists move closer to designing smarter stem cell therapies that can better withstand the hostile environment of a damaged disc and more reliably rebuild its cushioning core, potentially offering longer-lasting relief in the future.
Citation: Zhu, J., Jin, L., Jin, K. et al. Aberrantly expressed long noncoding RNAs in adipose-derived mesenchymal stem cells differentiation to nucleus pulposus-like cells. Sci Rep 16, 8029 (2026). https://doi.org/10.1038/s41598-026-36219-5
Keywords: intervertebral disc degeneration, stem cell therapy, nucleus pulposus cells, long noncoding RNA, RNA sequencing