Clear Sky Science · en
Pyridinium- and bromine-substituted distyryl-BODIPY dyes for mitochondria-targeted photodynamic therapy
Lighting Up Cancer Cells from the Inside
Photodynamic therapy is a cancer treatment that uses special light-activated drugs to kill tumor cells while largely sparing healthy tissue. This study explores a new family of fluorescent dyes designed to home in on the cell’s power plants—mitochondria—and, when illuminated with deep-red light, generate toxic oxygen species that can destroy cancer cells. The work shows both the promise and the pitfalls of engineering such dyes at the molecular level.
Why Target the Cell’s Power Stations?
Mitochondria are crucial for energy production and help decide whether a cell survives or dies. Cancer cells tend to have a more highly charged mitochondrial membrane than normal cells, which makes them attract positively charged molecules far more strongly. The researchers took advantage of this by attaching a positively charged pyridinium group to a well-known fluorescent dye scaffold called BODIPY. This design helps steer the dyes into mitochondria, where light-triggered chemical reactions can inflict maximum damage on cancer cells while limiting harm elsewhere.
Designing Smarter Light-Activated Dyes
The team created three closely related dyes, called PyBHI, PyBMI, and PyBBrI, that differ mainly in how many bromine atoms they carry (none, one, or two). All share a long, extended structure that shifts their light absorption and emission into the near-infrared region—a useful window where light can penetrate deeper into tissue. In theory, these dyes could use two different internal pathways to reach a “triplet” excited state that produces reactive singlet oxygen: one driven by the heavy bromine atoms and another driven by a temporary shift of electrical charge within the molecule. 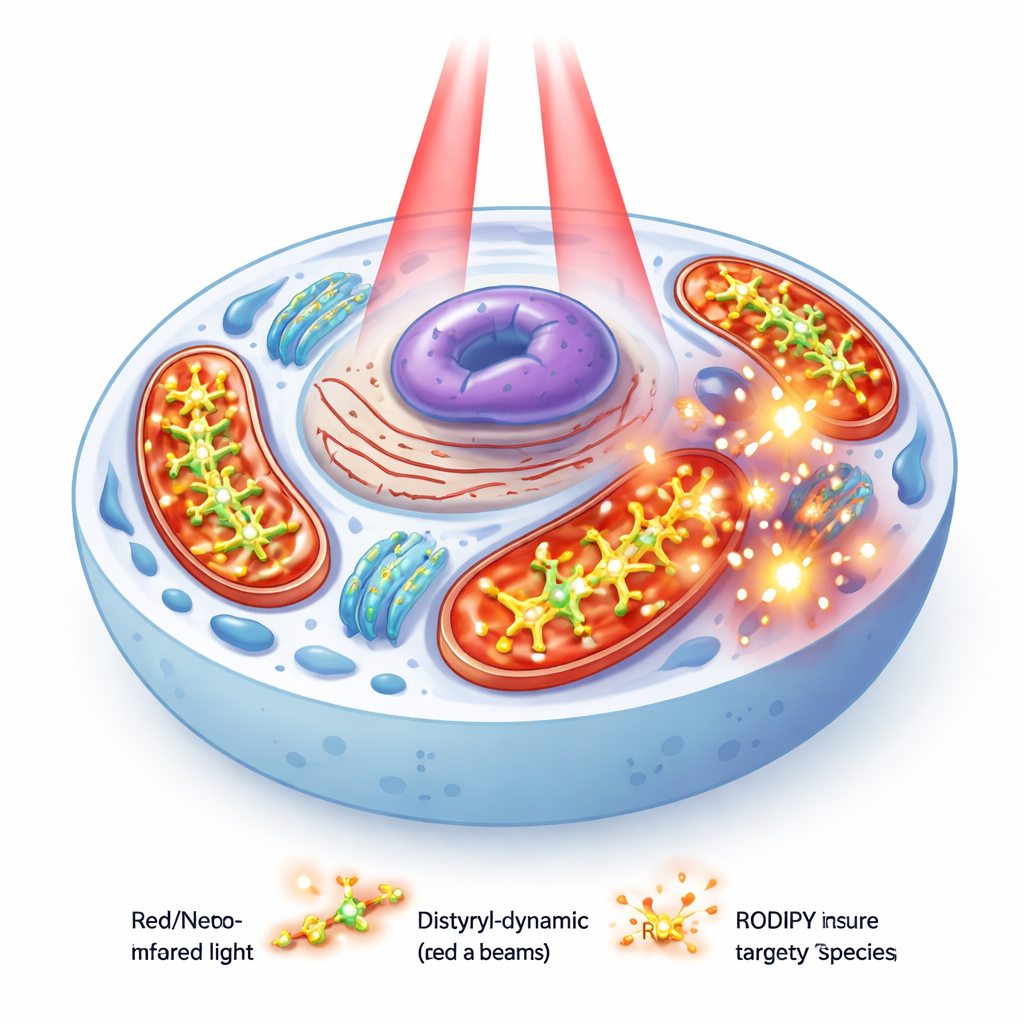
When Theory Meets Experimental Reality
Despite the elegant design, detailed tests showed that the dyes did not generate singlet oxygen as efficiently as hoped. Using chemical probes that lose their color upon reacting with singlet oxygen, the researchers measured very low oxygen-generation yields for all three dyes, with only a modest improvement as more bromine atoms were added. Ultrafast laser experiments revealed why: after light absorption, the dyes quickly enter charge-shifted states that mostly relax harmlessly as heat instead of converting into the desired triplet state. In the brominated versions, heavy atoms still open a pathway to the triplet state, but this route is much slower than the competing loss of energy, limiting overall efficiency.
Targeting Tumor Mitochondria in Living Cells
The story changes in a biological setting. In cultured human breast (MCF-7) and cervical (HeLa) cancer cells, all three dyes were shown by confocal microscopy to accumulate strongly in mitochondria, co-localizing with a standard mitochondrial stain. Toxicity tests in the dark indicated that the dyes are largely harmless without light. Under red-light irradiation, however, the di-brominated dye PyBBrI caused substantial cancer cell death, with low nanomolar doses cutting cell viability by half. The other two dyes were much less effective. 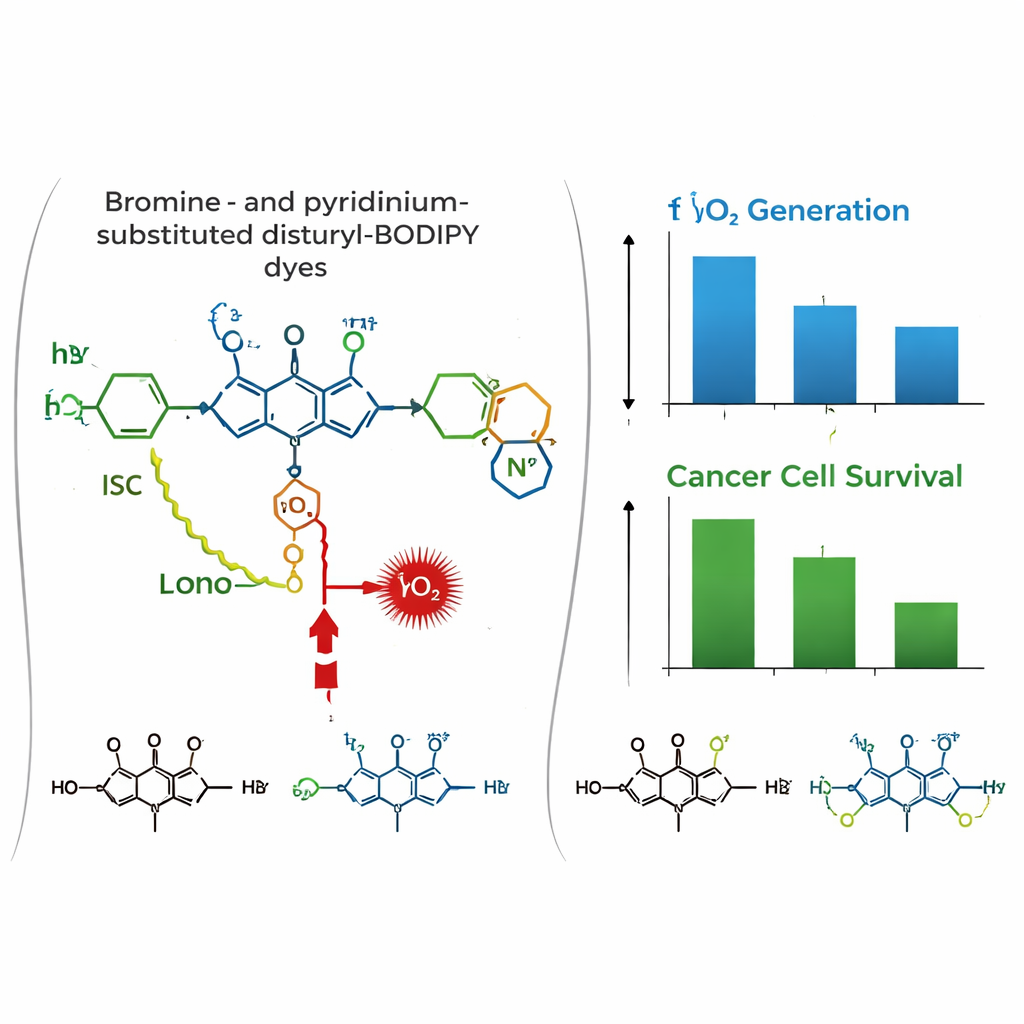
What This Means for Future Cancer Therapies
For a non-specialist, the key message is that small chemical tweaks can drastically change how light-activated cancer drugs behave, and that their performance in real cells can be better—or worse—than predicted from basic measurements. In this case, the most heavily modified dye, PyBBrI, showed only modest reactive oxygen production in a test tube but proved highly potent against cancer cells once it reached their mitochondria. The work highlights both the importance of directing drugs to vulnerable structures inside cancer cells and the complexity of predicting photodynamic efficiency. It points the way toward next-generation, mitochondria-targeted therapies that use near-infrared light to selectively “switch on” cell-killing chemistry exactly where it is needed.
Citation: Kim, C., Badon, I.W., Jo, J. et al. Pyridinium- and bromine-substituted distyryl-BODIPY dyes for mitochondria-targeted photodynamic therapy. Sci Rep 16, 6789 (2026). https://doi.org/10.1038/s41598-026-36213-x
Keywords: photodynamic therapy, mitochondria-targeted dyes, BODIPY, singlet oxygen, cancer cells