Clear Sky Science · en
Development of Dirofilaria immitis adult worms in NSG mice, detection of parasite-derived microRNA and comparative analysis of laboratory isolates
Why Heartworms Matter Beyond the Vet’s Office
Heartworm disease is best known as a threat to dogs, but the parasite that causes it, Dirofilaria immitis, can also affect cats and even humans. As the worms spread with changing climates and growing drug resistance, it becomes harder and more expensive to protect pets. This study describes a powerful new way to study heartworms in the lab using a special strain of mouse, and shows how tiny genetic signals in the blood could one day help detect infections earlier and guide better treatments. 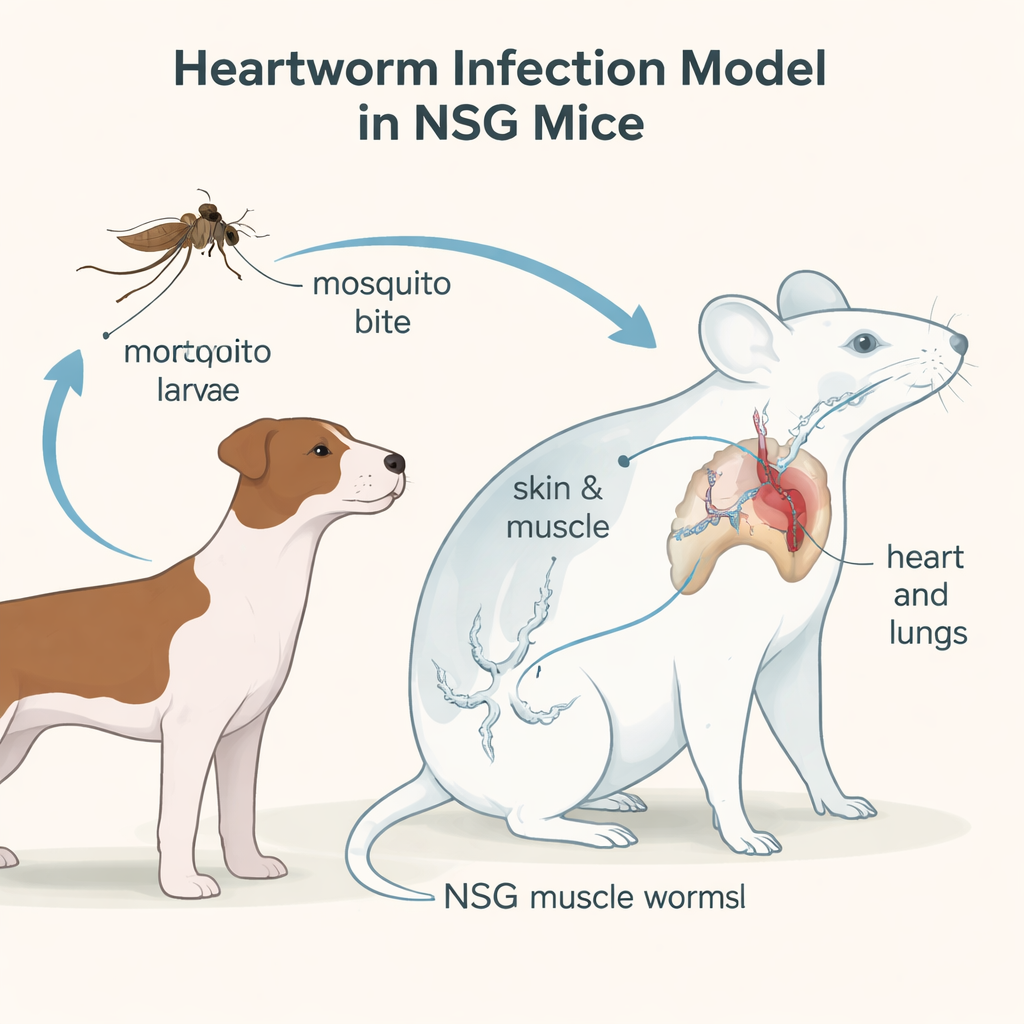
A Tiny Worm with a Big Journey
Heartworms have a surprisingly complex life. They start as microscopic larvae carried by mosquitoes, enter a new host through a bite, then migrate under the skin and through muscles before finally settling in the blood vessels of the heart and lungs. Until now, only dogs reliably supported the full development of these worms, which meant that testing new drugs or diagnostics required large, long, and costly dog studies. The researchers set out to see whether immunodeficient NSG mice—animals whose immune systems are largely switched off—could host heartworms from the early larval stages all the way to adulthood in a way that mimics what happens in dogs.
Following the Worms into the Heart and Lungs
By infecting NSG mice with a laboratory heartworm strain called JYD-34 and examining them over nearly six months, the team tracked where the worms were and how they changed over time. Early on, larvae were found in skin and muscle, just as in dogs. Later, they appeared in body cavities and, after about 80 days, in the heart and lungs. Careful microscope work showed that both male and female worms completed their major developmental steps, reaching sizes similar to those seen in dogs and developing fully formed reproductive organs. One key difference was that, even after almost six months, the female worms in mice had not yet released the next generation of microscopic offspring into the bloodstream, suggesting that some final trigger or extra time might be needed. 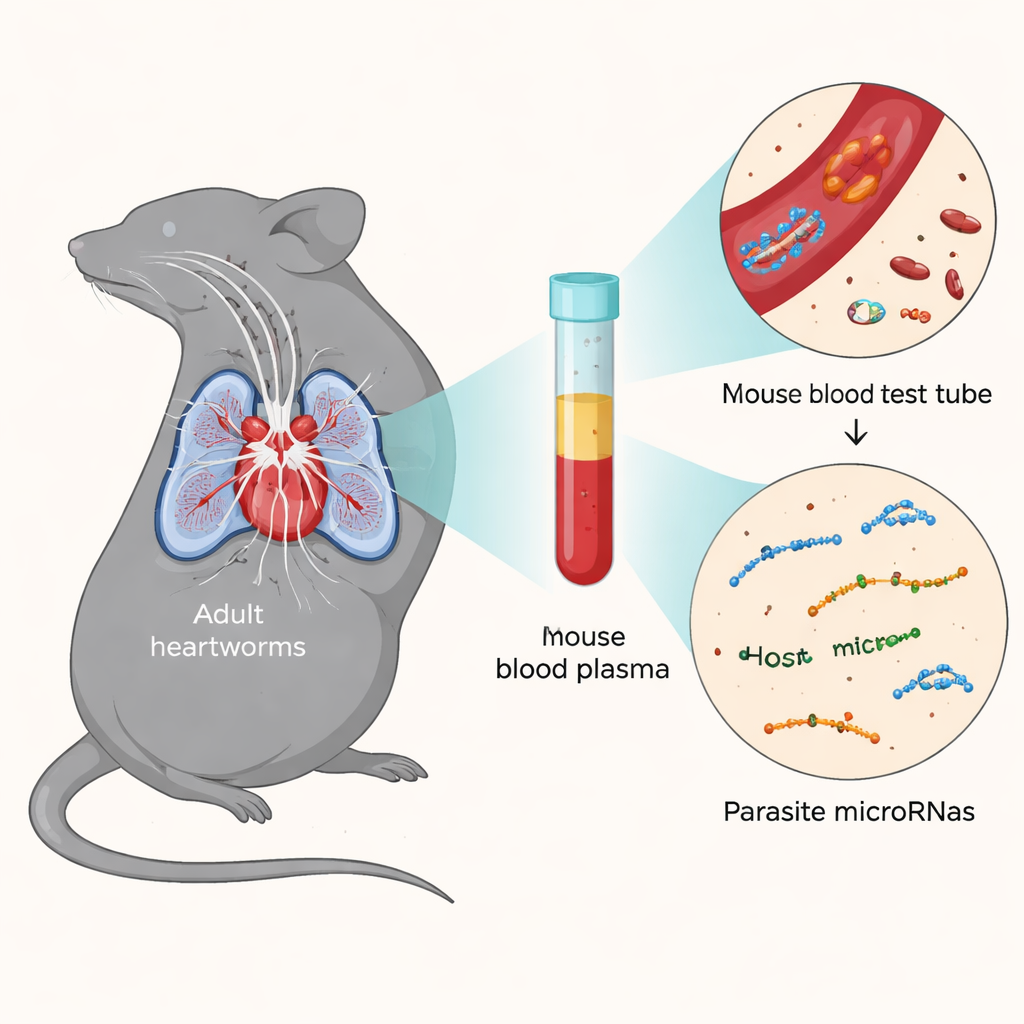
What the Mice’s Organs and Blood Revealed
Despite carrying adult worms in their hearts and lungs, the NSG mice showed only mild tissue changes, with little inflammation and limited signs of damage in the lungs, kidneys, and blood vessels. That contrasts with heavily infected dogs, which can develop coughing, breathing problems, and severe heart and lung disease. Because NSG mice lack many immune defenses, they may not develop the full-blown illness seen in pets, but that also makes them a controlled system for testing how drugs act on the worms without the complications of strong immune reactions. The scientists also examined the mice’s blood for microRNAs—short pieces of genetic material released by both host and parasite—and found 31 distinct heartworm-derived microRNAs along with nine mouse microRNAs that changed consistently during infection.
Early-Alert Signals and Strain-to-Strain Differences
The discovery of parasite microRNAs in mouse blood is important because the worms were still in relatively early stages that are hard to detect with today’s commercial tests. Many of the same microRNAs have been reported in infected dogs, suggesting that these tiny molecules might serve as reliable early warning signs of heartworm infection. The team also compared five different heartworm isolates, some resistant and some susceptible to common preventive drugs. All grew in the mice, but they did not behave the same: some strains reached the heart and lungs sooner, while one never made it there within the study period. These differences echo genetic and physical variations seen among heartworm populations in the real world and underscore why drug resistance and treatment failures can be so complex.
What This Means for Pets and People
By showing that NSG mice can host heartworms from the infective larval stage through to sexually mature adults, and by tying this to subtle organ changes and distinctive microRNA signals in the blood, this study delivers a versatile new model for heartworm research. For non-specialists, the key takeaway is that scientists now have a faster, more ethical way to probe how heartworms grow, how they damage the body, why some strains resist drugs, and how to spot infections earlier using molecular fingerprints in blood. In time, this mouse model and the microRNA markers it revealed could help drive better preventives, safer treatments, and more sensitive tests that protect both animals and the people who care for them.
Citation: Nakhale, M., Hess, J.A., Oliver, E. et al. Development of Dirofilaria immitis adult worms in NSG mice, detection of parasite-derived microRNA and comparative analysis of laboratory isolates. Sci Rep 16, 6764 (2026). https://doi.org/10.1038/s41598-026-36209-7
Keywords: heartworm, Dirofilaria immitis, NSG mouse model, microRNA biomarkers, drug-resistant parasites