Clear Sky Science · en
γ-Radiation induces region-specific subcellular alterations of amyotrophic lateral sclerosis and frontotemporal dementia markers in swine brain
Why tiny doses of radiation and pig brains matter
Most of us think of radiation as purely harmful, especially for the brain. But our world is filled with low levels of radiation from medical scans, air travel, and the environment. At the same time, scientists are still puzzling over what causes amyotrophic lateral sclerosis (ALS), the deadly disease that paralyzed baseball star Lou Gehrig, and its close cousin, frontotemporal dementia (FTD), which attacks behavior and personality. This study asks a surprisingly hopeful question: could a single, modest dose of gamma radiation subtly reshape brain chemistry in ways that are not purely damaging, and perhaps even protective, against the molecular changes linked to ALS and FTD?
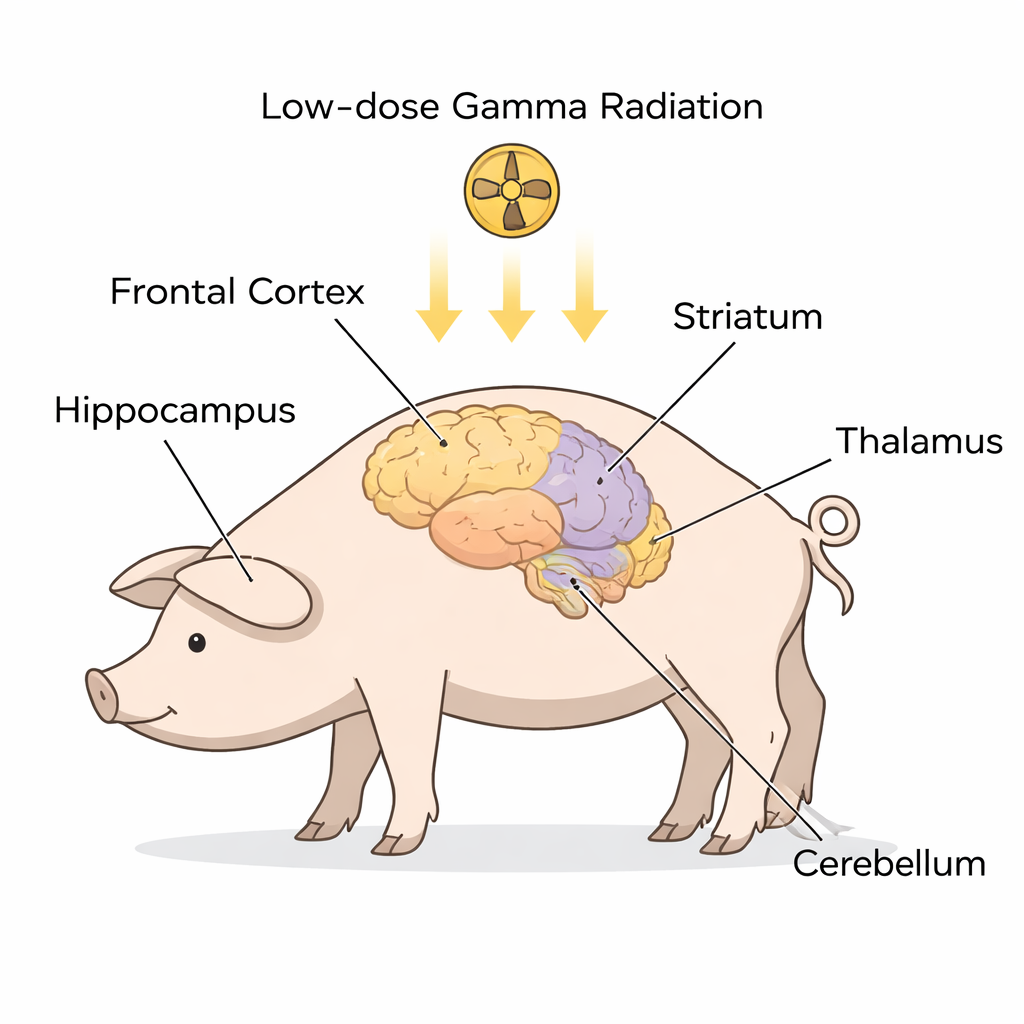
A closer look at ALS warning signs in the brain
Instead of examining patients who already had ALS, the researchers worked with healthy Göttingen minipigs, whose brains are anatomically similar to ours. Nine animals received a one-time, whole‑body exposure to low‑dose gamma radiation, while six were anesthetized but not irradiated and served as controls. About a month later, the scientists dissected five key brain regions often implicated in ALS and FTD—the frontal cortex, hippocampus, striatum, thalamus/hypothalamus, and cerebellum. In each region they separated the cell contents into two main compartments: the cytoplasm (the cell’s fluid interior) and the nucleus (the compartment that holds DNA). Using a technique called Western blotting, they measured levels of several proteins that are strongly tied to ALS and ALS‑FTD, including SOD1, FUS, C9orf72, STMN2, ubiquitin, TDP‑43, and a modified form called phosphorylated TDP‑43.
Radiation leaves some markers unchanged—and others shifted
One reassuring finding was that total levels of SOD1, a protein whose mutations cause some inherited ALS, did not change in any region after radiation. Likewise, the main forms of TDP‑43—the protein most often found in ALS‑related brain clumps—stayed stable in both the nucleus and cytoplasm across all regions. But not everything was static. The team observed region‑specific and compartment‑specific shifts in several other proteins. For example, FUS, a protein that normally lives in the nucleus but can misbehave and clump in ALS, decreased in the cytoplasm of the thalamus/hypothalamus after radiation, while its nuclear levels rose in the frontal cortex and fell in the hippocampus. The ALS‑linked protein C9orf72 did not change in the cell fluid but showed a drop inside hippocampal nuclei. STMN2, a protein important for maintaining nerve fibers, also declined in the hippocampal nuclei, hinting at a coordinated response between these two molecules in that memory‑related region.
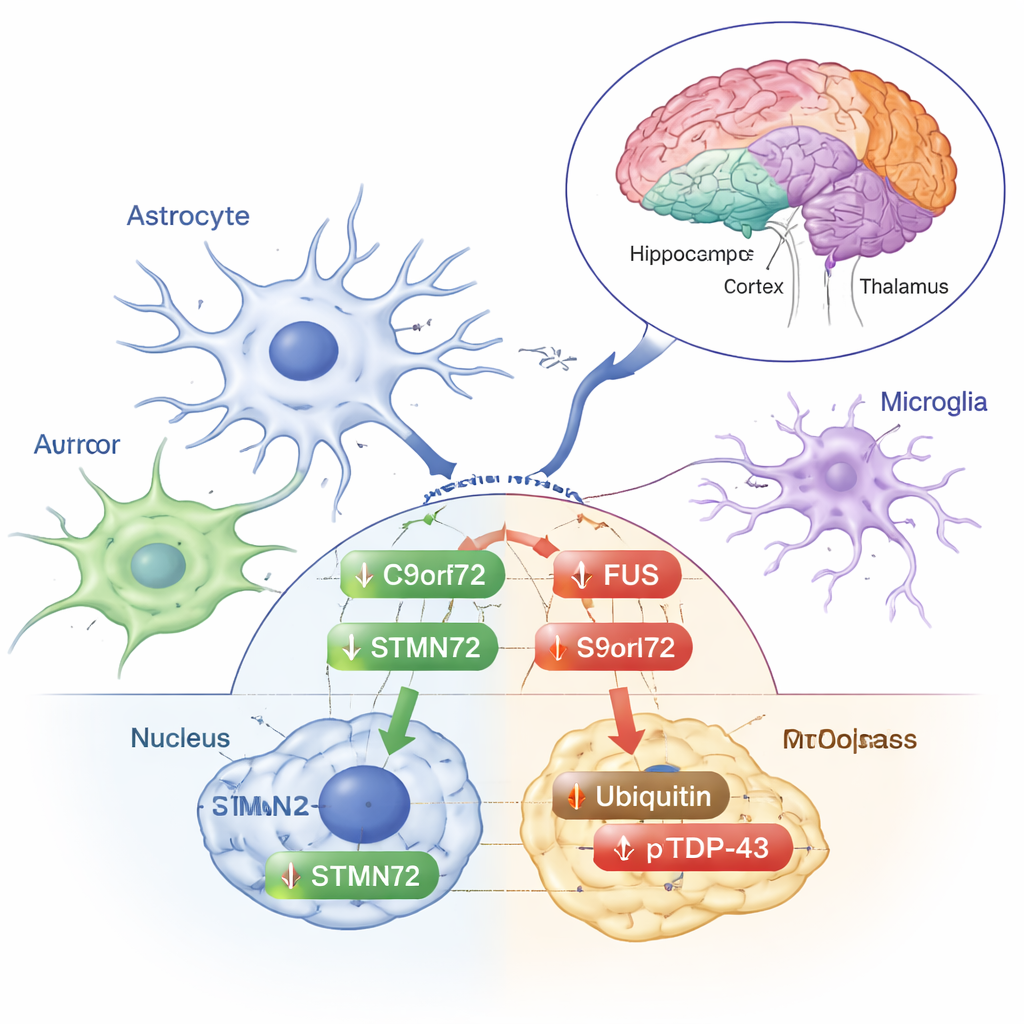
Stress signals or early protection?
Other changes looked more like classic stress responses. Ubiquitin—the small tag the cell uses to mark damaged proteins for cleanup—increased in the cytoplasmic fraction of the frontal cortex. The phosphorylated form of TDP‑43, which is often enriched in the abnormal deposits seen in ALS and FTD, rose in the cytoplasm of the frontal cortex, striatum, and thalamus/hypothalamus, even though total TDP‑43 itself did not. These shifts suggest that low‑dose radiation nudges the brain’s protein‑handling machinery without triggering the full‑blown patterns of misfolding and aggregation that define disease. The authors emphasize that these responses vary by brain region and by whether a protein is inside or outside the nucleus, underscoring how finely tuned the brain’s reaction to stress can be.
What this could mean for ALS risk—and possible benefits
Does this mean low‑dose radiation causes ALS? The data argue against a simple yes. The researchers did not see the hallmark mislocalization or buildup of the main ALS proteins, and many changes pointed toward altered regulation rather than outright damage. Intriguingly, these results fit with earlier work from the same group and others suggesting that small amounts of radiation can sometimes trigger “hormesis”—a phenomenon where mild stress activates repair pathways and reduces harmful proteins, such as those linked to Alzheimer’s or Parkinson’s disease, in animal models. The current study hints that, at least in the short term, a single low dose of gamma radiation reshapes ALS‑related markers in ways that may be adaptive rather than destructive.
Big questions and cautious hope
For lay readers, the key takeaway is that the relationship between radiation and brain health is more nuanced than “all radiation is bad.” In this large‑animal model, low‑dose gamma exposure produced subtle, region‑specific shifts in proteins associated with ALS and FTD, without reproducing the damaging patterns seen in patients’ brains. These changes might reflect the brain’s attempt to cope with and repair stress, and in some cases could even prove beneficial. However, the study looked only a month after a single exposure, and only in the brain (not the spinal cord, where ALS strikes hardest). Long‑term and repeated‑dose studies are needed before anyone considers translating such findings to people. Still, this work opens a new window on how everyday‑level radiation interacts with the molecules that underlie devastating neurodegenerative diseases—and raises the possibility that, in carefully controlled settings, small doses might one day be harnessed to nudge the brain’s own defenses.
Citation: Iacono, D., Murphy, E.K., Perl, D.P. et al. γ-Radiation induces region-specific subcellular alterations of amyotrophic lateral sclerosis and frontotemporal dementia markers in swine brain. Sci Rep 16, 5627 (2026). https://doi.org/10.1038/s41598-026-36208-8
Keywords: amyotrophic lateral sclerosis, frontotemporal dementia, low-dose radiation, brain proteins, minipig model