Clear Sky Science · en
Leukocyte telomere attrition following radiotherapy in prostate cancer: a prospective study
Why this research matters for cancer survivors
People are living longer after cancer, but many survivors notice problems that feel like “fast‑forwarded” aging—fatigue, frailty, and other chronic conditions that seem to arrive too soon. This study asks a simple but important question: does radiation treatment for prostate cancer leave a lasting mark on the body’s basic aging clock, hidden in our chromosomes?
The tiny caps that protect our DNA
At the ends of our chromosomes sit telomeres, tiny stretches of DNA that work like the plastic tips on shoelaces, preventing genetic material from fraying. Each time a cell divides, these caps get a little shorter. When they become too short, the cell is pushed into a shutdown state or dies, a process tied to natural aging. Because white blood cells are easy to sample from blood and reflect what happens in many tissues, their telomere length is often used as a yardstick for “biological age,” which can differ from the number of years we have lived.
Taking a closer look during and after treatment
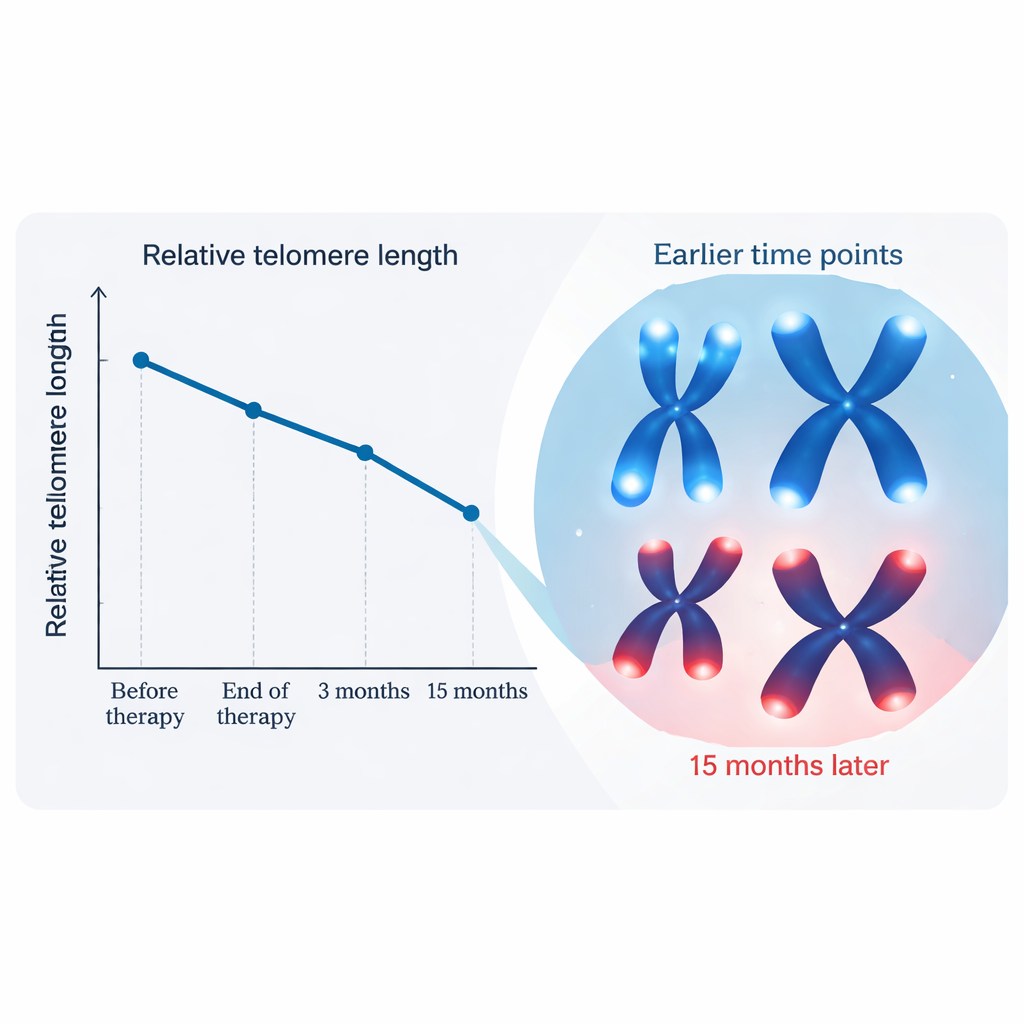
To see how modern prostate radiotherapy might influence this aging yardstick, researchers followed 314 men aged 65 and older receiving curative radiation at one medical center. Blood samples were taken four times: before treatment, at the end of the several‑week course of radiotherapy, and then 3 months and 15 months later. In each sample, the team measured the average relative length of telomeres in white blood cells using a sensitive DNA test. This design let them track how each man’s telomeres changed over time, rather than just comparing different people at a single snapshot.
What happened to telomeres over time
Telomere length stayed broadly similar from before treatment to the end of radiotherapy and again at the 3‑month check, with small ups and downs that were not statistically meaningful. The striking change appeared at the 15‑month mark: on average, telomeres were about 15 percent shorter than at the start, a drop much larger than what would be expected from normal aging over just a little more than a year in older adults. This pattern suggests that the long‑term biological impact of radiation on the blood may emerge slowly, well after visible side effects have faded. The researchers also explored whether factors such as age, high blood pressure, smoking, treatment details, or radiation to nearby lymph nodes might influence these changes. Age and high blood pressure showed modest links to how much shortening occurred, while most treatment variations did not clearly change the outcome in this group.
How radiation might speed cellular wear and tear
Why would a local radiation treatment to the prostate affect telomeres in circulating blood cells? The authors point to two connected processes. First, ionizing radiation generates reactive oxygen molecules that can damage DNA, and telomeres are particularly vulnerable to this kind of harm and are repaired less efficiently than other regions. Second, radiotherapy can trigger a lingering inflammatory reaction in the body, driving white blood cells to divide more often and releasing more damaging oxygen by‑products. Both mechanisms could accelerate the gradual erosion of telomeres, nudging cells toward a state of senescence—an irreversible “old” state in which they no longer divide but continue to influence surrounding tissues.
What this could mean for patients
For men in this study, prostate radiotherapy remained a curative treatment, but it was also associated with signs of faster biological aging in their blood cells a year after therapy. Shorter telomeres alone do not prove that a person will become frail or ill, yet they are linked to a higher risk of age‑related problems such as heart disease, diabetes, and cognitive decline. The findings highlight that successful cancer treatment can carry hidden long‑term costs and underscore the need for follow‑up care that considers aging‑related health, not just tumor control. The authors argue that larger, longer studies—including comparisons with people who are not treated with radiation—are needed to confirm these results and to test protective approaches, such as targeting inflammation or oxidative stress, that might help preserve patients’ biological resilience after therapy.
Citation: Langsenlehner, T., Paal, K., Thurner, E.M. et al. Leukocyte telomere attrition following radiotherapy in prostate cancer: a prospective study. Sci Rep 16, 5905 (2026). https://doi.org/10.1038/s41598-026-36205-x
Keywords: prostate cancer, radiotherapy, telomeres, biological aging, cellular senescence