Clear Sky Science · en
Eccentric sombor index of graphs and its role in the structure-property relationship analysis of polycyclic aromatic compounds
Why hidden patterns in molecules matter
Chemists and drug designers are surrounded by countless possible molecules, but testing each one in the lab is slow and expensive. This study explores a mathematical shortcut: using the shape of a molecule, written as a simple network, to predict important physical and safety properties without touching a test tube. The authors focus on a new network-based number, the “eccentric Sombor index,” and show that it can reliably forecast how certain ring-shaped pollutants behave.
A network view of familiar ring-shaped chemicals
Many common chemicals, including pollutants from combustion, are built from fused benzene rings and are known as polycyclic aromatic hydrocarbons (PAHs). In a drawing of such a molecule, chemists can treat each non-hydrogen atom as a point and each bond as a line, forming a network, or graph. Classic graph-based measures count how many neighbors an atom has. The authors instead look at how far each atom sits from the most distant atom in the same molecule, a quantity called its eccentricity. By combining these eccentricities over all bonds with a specific formula, they obtain the eccentric Sombor index, a single number that captures how spread out and ring-like the whole structure is.
From abstract numbers to real-world properties
The core idea of the paper is to connect this abstract index to everyday measurable properties of PAHs, such as molecular weight, boiling point, how easily they polarize in an electric field, and the temperatures at which they melt or ignite. To do this, the researchers calculated the eccentric Sombor index for 32 widely studied PAHs, ranging from the simple two-ring naphthalene to larger, more complex ring systems. For each molecule, they pulled standard physicochemical data from the PubChem database and then used linear regression, a standard statistical method, to see how well a straight-line relationship links the index to each property.
Strong straight-line links emerge
The analysis revealed that the eccentric Sombor index tracks several key properties with striking accuracy. Measures such as molecular weight, boiling point, molar refractivity, polarizability, molar volume, and flash point all showed coefficients of determination (R²) above 0.9, meaning over 90% of the variation in these values across the 32 PAHs can be explained by a simple line drawn through the scatter of points. Some traits, like melting point and a general “complexity” measure, were less tightly linked, but the overall picture is that this single graph-based number encodes a surprisingly rich amount of physical information about these ringed molecules.
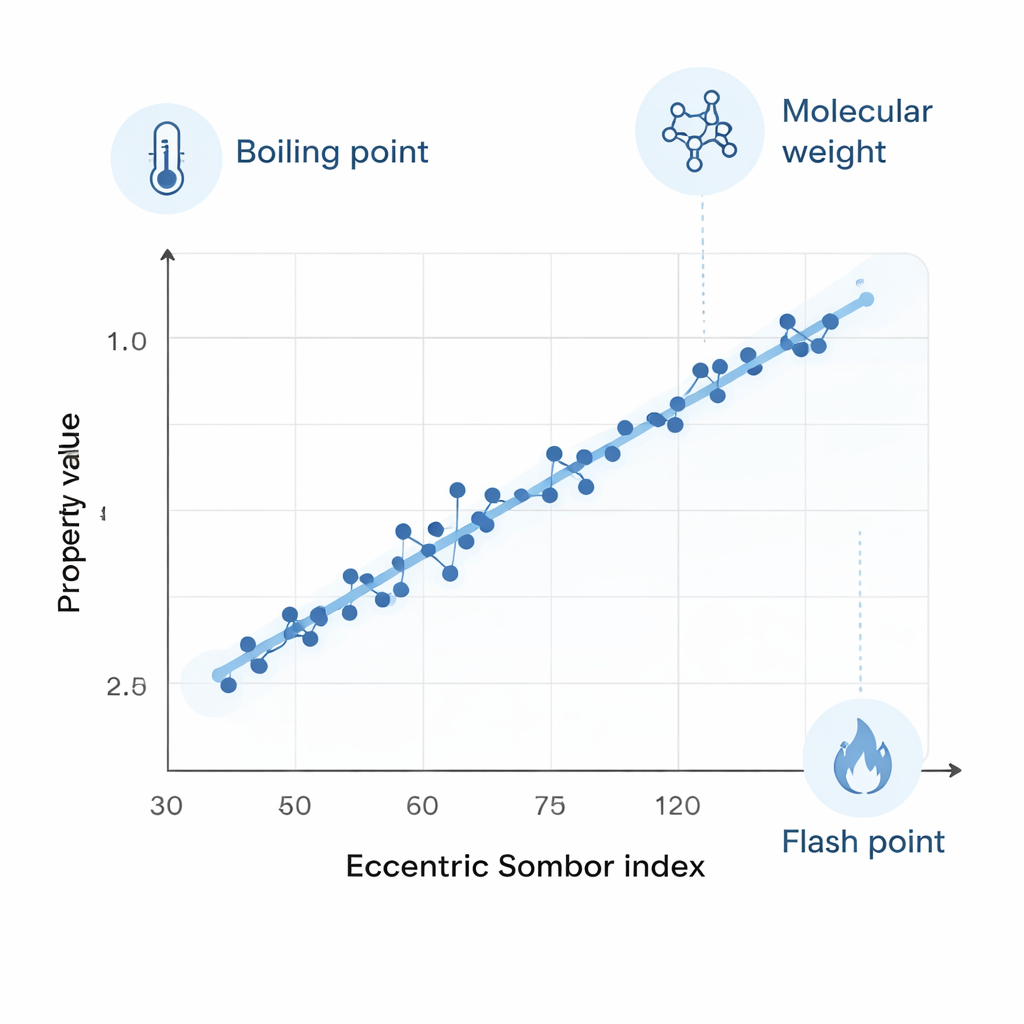
Beating an older yardstick
The eccentric Sombor index is part of a family of tools; an earlier, more traditional version used simple neighbor counts instead of eccentricities. The authors directly compared the predictive power of the new and old versions for the same set of PAHs. While the degree-based Sombor index already worked reasonably well, the eccentric version generally did better, sometimes by a large margin. For example, molar volume, which reflects how much space a molecule effectively occupies, was predicted far more accurately by the eccentric Sombor index. This suggests that taking long-range layout within the molecule into account gives a more faithful picture than just counting local connections.
What this means and where it can lead
For non-specialists, the takeaway is that the detailed way atoms are arranged in a molecule can be boiled down to a single, cleverly designed network number that predicts many laboratory properties with high accuracy. In this study, that number—the eccentric Sombor index—works especially well for fused-ring pollutants known as PAHs and outperforms an older, simpler measure. While the work is limited to one class of compounds and to straight-line models, it points toward a future in which chemists can rapidly screen new molecules, guide safer design, and narrow down candidates for drugs or materials using math on a computer before ever mixing chemicals in the lab.
Citation: Khaji, B., Hanif, S. & Arathi Bhat, K. Eccentric sombor index of graphs and its role in the structure-property relationship analysis of polycyclic aromatic compounds. Sci Rep 16, 6282 (2026). https://doi.org/10.1038/s41598-026-36192-z
Keywords: polycyclic aromatic hydrocarbons, topological indices, quantitative structure–property relationships, graph-based molecular descriptors, eccentric Sombor index