Clear Sky Science · en
The Lysis cassette of jumbophage PhiKZ
How Giant Viruses Burst Open Bacteria
Viruses that infect bacteria—called bacteriophages or simply phages—are being explored as precision weapons against dangerous infections like those caused by Pseudomonas aeruginosa, a hospital superbug. One especially large phage, phiKZ, has fascinated scientists because it builds a protective “nucleus-like” compartment inside its host. Yet until now, no one really knew how this giant phage actually breaks its host open to escape. This study uncovers the set of genes and molecular tools phiKZ uses to time and execute that final explosive step.
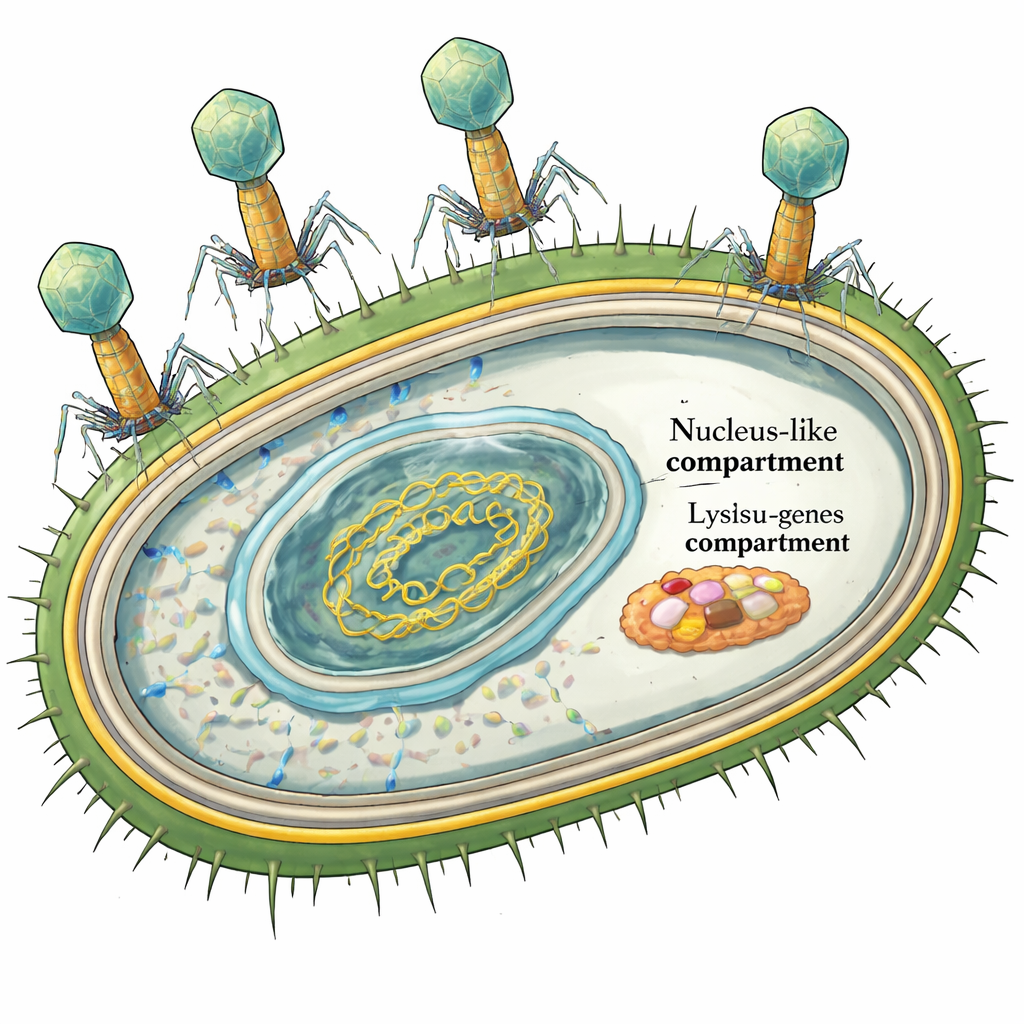
The Giant Phage and Its Escape Plan
PhiKZ is a “jumbo” phage, with a genome more than 200,000 DNA letters long. Earlier work showed in detail how it hides its DNA inside a protein shell to dodge bacterial defenses. But the last act of the infection—the controlled destruction of the bacterial cell—was a mystery. Most phages that infect bacteria with two membranes, like Pseudomonas, use a four-part lysis system: one protein pokes holes in the inner membrane, another cuts the cell wall, and a two-part complex collapses the outer membrane. Because phiKZ’s genome seemed to lack a recognizable “hole-forming” gene, some researchers proposed that it might use a completely different strategy to get its wall-cutting enzyme into place.
Finding the Hidden Lysis Toolkit
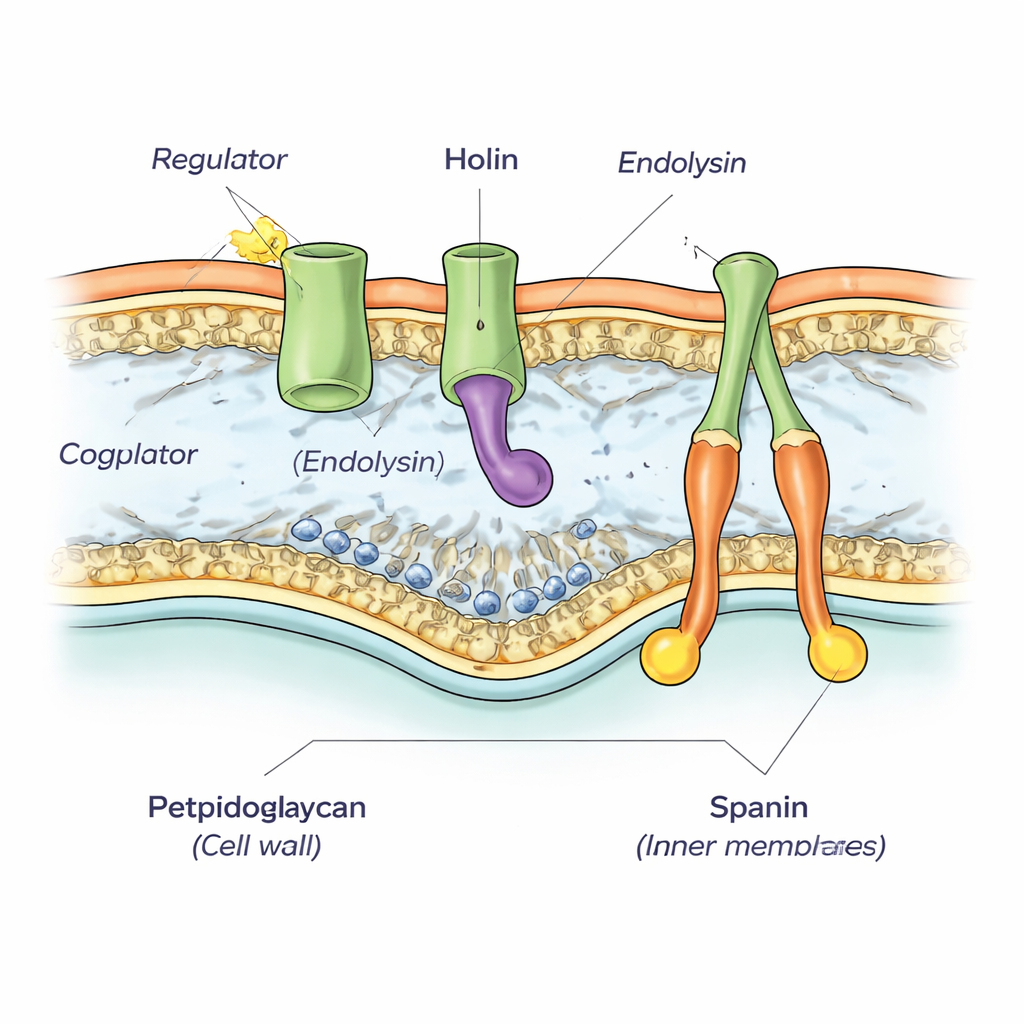
By re-examining the region of the phiKZ genome surrounding a known wall-cutting enzyme (called an endolysin), the authors discovered a tightly packed cluster of five genes. Through sequence analysis and computer predictions, they showed that two of these genes encode the spanins, a pair of proteins that link the inner and outer membranes and later help them fuse. Another gene encodes the endolysin itself. A fourth gene turned out to be the missing holin—the protein that quietly builds up in the inner membrane before suddenly opening large holes. The researchers confirmed these functions experimentally by transplanting phiKZ genes into well-studied lambda phage and Escherichia coli systems and showing that the phiKZ versions could stand in for missing parts.
A Timing Switch for Cell Destruction
The fifth gene in the cluster encodes a small protein that remains in the bacterial cytoplasm rather than embedding in a membrane. When the authors expressed this protein alongside the holin in E. coli, cells died noticeably earlier than when the holin acted alone, even though the rest of the lysis machinery was absent. This suggests that the extra protein acts as a regulator, nudging the holin toward earlier or enhanced pore formation. When the team trimmed away sections of the holin’s long inside-facing tail, they found that the regulator could no longer speed things up and, in some cases, even blocked lysis. Structural models produced with AlphaFold software supported a physical partnership between the holin’s inner tail and the regulator, forming a stable two-protein complex.
Hints of a Delay Mechanism at High Virus Doses
When the researchers infected bacterial cultures with different numbers of phiKZ particles, they noticed an intriguing pattern. At low virus doses, the culture lysed relatively quickly and sharply. At high doses, however, lysis was delayed and stretched out over more than two hours. This counterintuitive slowdown echoes a classic phenomenon called lysis inhibition, known from another phage called T4, in which additional infections tell the virus to postpone bursting the cell so that more viral particles can be produced. Because phiKZ has a holin-regulator pair capable of both advancing and, under some conditions, blocking lysis, the authors suggest that a similar delay system may exist in this jumbo phage.
Why This Matters for Phage Therapy
By establishing that phiKZ carries a conventional, yet sophisticated, lysis cassette—complete with holin, endolysin, spanins, and a timing regulator—this work shows that even exotic jumbo phages rely on familiar tools to break out of their hosts. For non-specialists, the key takeaway is that the “burst timing” of therapeutic phages can be tuned by small regulatory proteins that talk to the pore-forming holins. Understanding and eventually engineering these timing switches could help design phage treatments that either kill bacteria as fast as possible or, when beneficial, hold off lysis to maximize viral amplification before the final blow.
Citation: Manohar, P., Wan, J., Ganser, G. et al. The Lysis cassette of jumbophage PhiKZ. Sci Rep 16, 5840 (2026). https://doi.org/10.1038/s41598-026-36188-9
Keywords: bacteriophage lysis, phiKZ jumbo phage, holin endolysin system, Pseudomonas aeruginosa phage, lysis inhibition