Clear Sky Science · en
Diagnostic performance evaluation of Elecsys anti-SARS-CoV-2 assay against RT-PCR for SARS-CoV-2 detection and surveillance in Ethiopian referral hospitals: cross-sectional study
Why this study matters for everyday life
More than four years after COVID-19 first appeared, many countries still struggle to test people quickly and accurately, especially in low-resource settings. This study from Ethiopia’s Oromia region asks a practical question with global relevance: can a commonly used blood test for COVID-19 antibodies stand in for the more complex PCR test when it comes to finding who is currently infected—and how many people have already been exposed? The answer has big implications for how hospitals monitor outbreaks, protect patients and staff, and plan for future epidemics.
Taking a closer look inside busy hospitals
Researchers worked in five large referral hospitals in Oromia during August 2021, a period when COVID-19 was still actively spreading. They enrolled 1,360 patients coming to these hospitals for care, regardless of whether they felt sick with COVID-19-like symptoms. Each person gave two types of samples: a swab from the nose for the RT-PCR test, which detects the virus itself, and a blood sample for the Elecsys anti-SARS-CoV-2 test, which looks for antibodies that show past infection. Alongside the lab tests, trained staff collected information on age, residence, symptoms, medical conditions, and contact with known COVID-19 cases.
How common were current and past infections?
The results revealed that COVID-19 had left a deep footprint among hospital patients. Nearly one in five participants (about 18%) had an active infection detected by RT-PCR at the time of the study. Even more striking, just over half (53.2%) had antibodies, indicating they had been infected at some point in the past—often without ever having had a confirmed diagnosis. Urban residents were far more likely than rural residents to have an active infection, and classic symptoms such as fever, cough, muscle aches, and contact with confirmed cases were linked to a higher chance of testing positive by RT-PCR. In contrast, the presence or absence of symptoms and contact history did not clearly predict who had antibodies, reflecting the virus’s widespread and often silent circulation over many months.
Putting the antibody test to the test
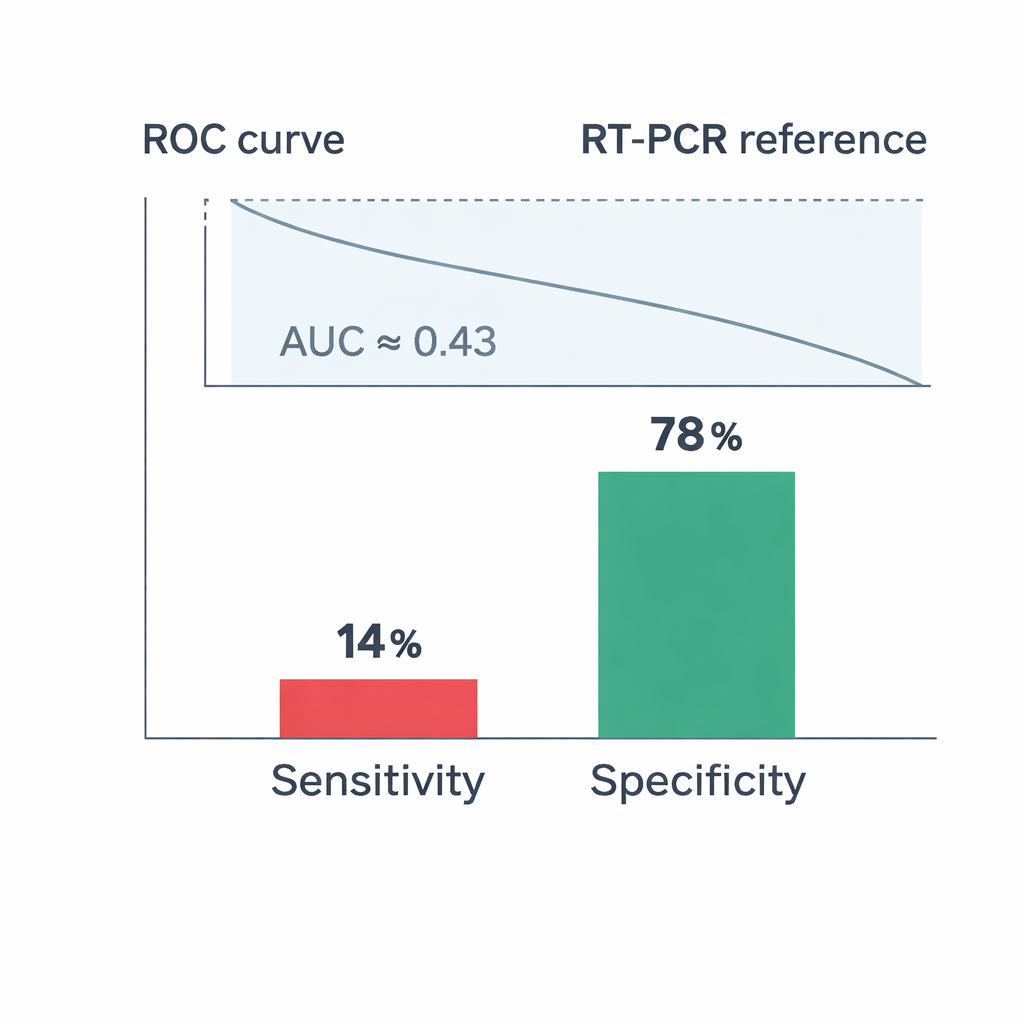
The core of the study was to see how well the Elecsys antibody test could stand in for RT-PCR when identifying who was currently infected. Here the findings were sobering. Among people who were PCR-positive, only 14% had a positive Elecsys result at the same time, meaning the blood test missed the vast majority of active infections. Its specificity—how often it correctly identified people without current infection—was higher at about 78%, but overall agreement between the two tests was poor. A statistical measure called Cohen’s kappa was close to zero, and the area under the ROC curve (AUC), which summarizes overall diagnostic performance, was only 0.43—worse than what would be expected by chance for distinguishing infected from non-infected individuals.
What these numbers tell us about test roles
Why did the antibody test perform so poorly for catching active cases? The two tests are looking at different stages of infection. RT-PCR detects viral genetic material during the brief window when the virus is actively replicating and a person is contagious. Antibodies, in contrast, usually rise later and can remain in the blood for months. When both tests are done on the same day, many truly infected people may not yet have built up enough antibodies to be detected. In a population where more than half of people have already been infected at some point, this timing mismatch makes agreement between the tests look especially low. The study’s hospital-based sample—likely including many severely ill or previously exposed patients—further highlighted this disconnect.
What it means for patients and public health
For patients and policymakers, the message is clear but two-sided. RT-PCR remains essential for diagnosing who is currently infected and needs isolation or treatment. The Elecsys antibody test, at least as used in this real-world Ethiopian setting, should not be relied upon to detect active COVID-19 on its own. However, its relatively high specificity and simple blood-based method make it useful for a different task: tracking how widely the virus has spread in the community over time. In regions where PCR machines and trained staff are scarce, antibody testing can help estimate total exposure and guide long-term planning for vaccines and future outbreaks—even if it cannot safely replace RT-PCR at the bedside.
Citation: Gobena, D., Gudina, E.K., Degfie, T.T. et al. Diagnostic performance evaluation of Elecsys anti-SARS-CoV-2 assay against RT-PCR for SARS-CoV-2 detection and surveillance in Ethiopian referral hospitals: cross-sectional study. Sci Rep 16, 5207 (2026). https://doi.org/10.1038/s41598-026-36177-y
Keywords: COVID-19 testing, SARS-CoV-2 antibodies, RT-PCR diagnosis, Ethiopia hospitals, sero-surveillance