Clear Sky Science · en
FcIgG-GE11-Melittin as a novel EGFR targeted peptibody with potent cytotoxic activity against cancer cells
Bee Venom Meets Precision Cancer Therapy
Many promising cancer drugs are simply too harsh on the rest of the body to be used safely. One striking example is melittin, the main toxin in bee venom: it can tear apart cancer cells, but it can also damage red blood cells and healthy tissues. This study explores a clever workaround—building a “guided missile” that carries melittin directly to tumor cells that display a specific surface marker, while largely sparing normal cells.
Why Target a Common Cancer Switch
A key player in many tumors is a molecule called the epidermal growth factor receptor, or EGFR. This receptor sits on the surface of cells and helps control growth and survival. In several cancers—including lung, colorectal, prostate, and some head-and-neck and kidney cancers—EGFR is overproduced, driving uncontrolled cell division. Because of this, drugs that latch onto EGFR have become an important class of targeted therapies. However, existing EGFR drugs mainly block signals; they do not actively punch holes in cancer cells. The researchers asked whether they could harness melittin’s powerful cell-lysing ability and deliver it selectively to EGFR-rich tumor cells.
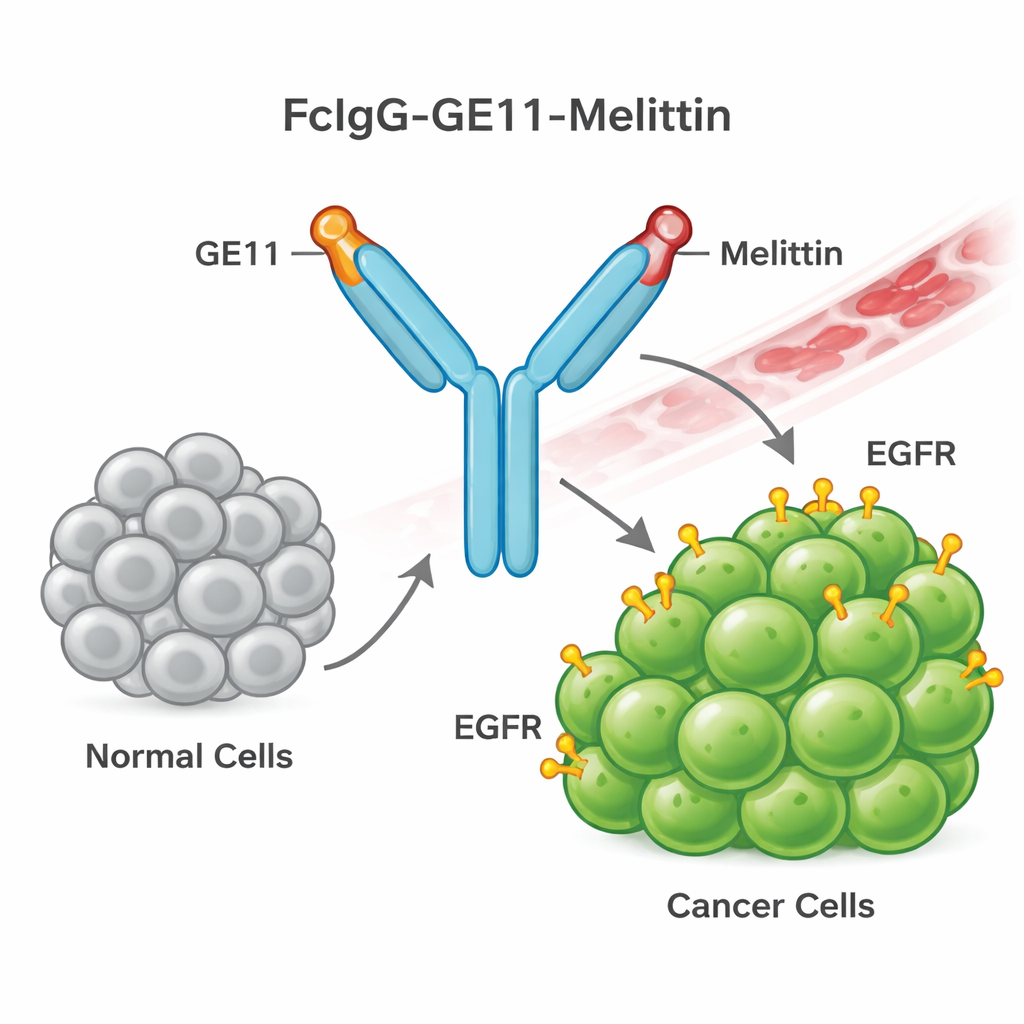
Building a Guided Bee-Venom Weapon
To do this, the team designed a new fusion protein they call FcIgG-GE11-Melittin. It combines three parts: an antibody “tail” (the Fc region of human IgG) that improves stability in the bloodstream and makes purification easier; a short guiding peptide called GE11 that recognizes and binds EGFR on cancer cells; and melittin, the bee-venom peptide that can rupture cell membranes. Bacteria were engineered to produce this fusion protein in large amounts. The protein was then refolded and purified, and lab tests confirmed that it formed the expected dimeric shape and carried the human antibody tag, showing that the construct had been built correctly.
Locking Onto Tumor Cells, Not Just Any Cell
The next question was whether this designer molecule actually finds and sticks to EGFR-rich cancer cells. Using flow cytometry, the authors tested binding on A549 lung cancer cells, which are known to overexpress EGFR. Cells treated with FcIgG-GE11-Melittin showed a strong increase in fluorescence, indicating that many fusion molecules had attached to their surface. An unrelated peptibody, Romiplostim, showed almost no binding, serving as a negative control. The binding profile of the new construct closely matched that of Cetuximab, a well-known EGFR antibody, supporting the idea that the GE11 component is correctly guiding the fusion protein to its target.
Killing Cancer Cells While Sparing Blood Cells
The team then tested how well the fusion protein could kill different cancer cell lines in culture. Free melittin alone was extremely potent but indiscriminate, reducing cell survival sharply even at low doses in lung (A549), prostate (DU145), and colorectal (SW480) cancer cells. By contrast, FcIgG-GE11-Melittin showed dose-dependent killing that was strongest in EGFR-positive cells, while an EGFR-low breast cancer line (MDA-MB-453) was relatively resistant. This pattern suggests that the fusion construct’s attack depends on EGFR targeting rather than random membrane damage. Most importantly, in experiments with human red blood cells, free melittin caused nearly complete hemolysis at modest doses, whereas the fusion protein produced less than 5% hemolysis even at very high concentrations. In other words, attaching melittin to the EGFR-seeking carrier dramatically reduced its tendency to burst red blood cells.
What This Could Mean for Future Cancer Care
Overall, the work shows that coupling a potent membrane-disrupting toxin like melittin to a precise targeting system can shift it from a broadly toxic substance to a more tumor-selective weapon. FcIgG-GE11-Melittin binds strongly to EGFR-overexpressing cancer cells, kills them in a controlled, dose-dependent way, and shows minimal damage to red blood cells in the lab. While further animal studies and safety testing are needed before any use in patients, this approach illustrates how re-engineering natural toxins into guided therapies could open new avenues for treating EGFR-positive cancers more effectively and with fewer side effects.
Citation: Hallaji, M., Fayaz, S., Allahyari, M. et al. FcIgG-GE11-Melittin as a novel EGFR targeted peptibody with potent cytotoxic activity against cancer cells. Sci Rep 16, 7047 (2026). https://doi.org/10.1038/s41598-026-36167-0
Keywords: targeted cancer therapy, EGFR, bee venom melittin, peptibody, tumor-selective toxins