Clear Sky Science · en
Multinomial probability model of radiation induced DSB and non-DSB clusters: tandem and bistranded damage clusters
Why tiny DNA injuries matter
When we think about radiation damage to our genes, we usually picture dramatic snaps in the DNA double helix. But most radiation injuries are subtler and more crowded together than a single clean break. This paper explores those hidden clusters of small DNA wounds that may quietly drive cancer risk from medical radiation, occupational exposure, and cosmic rays in space.
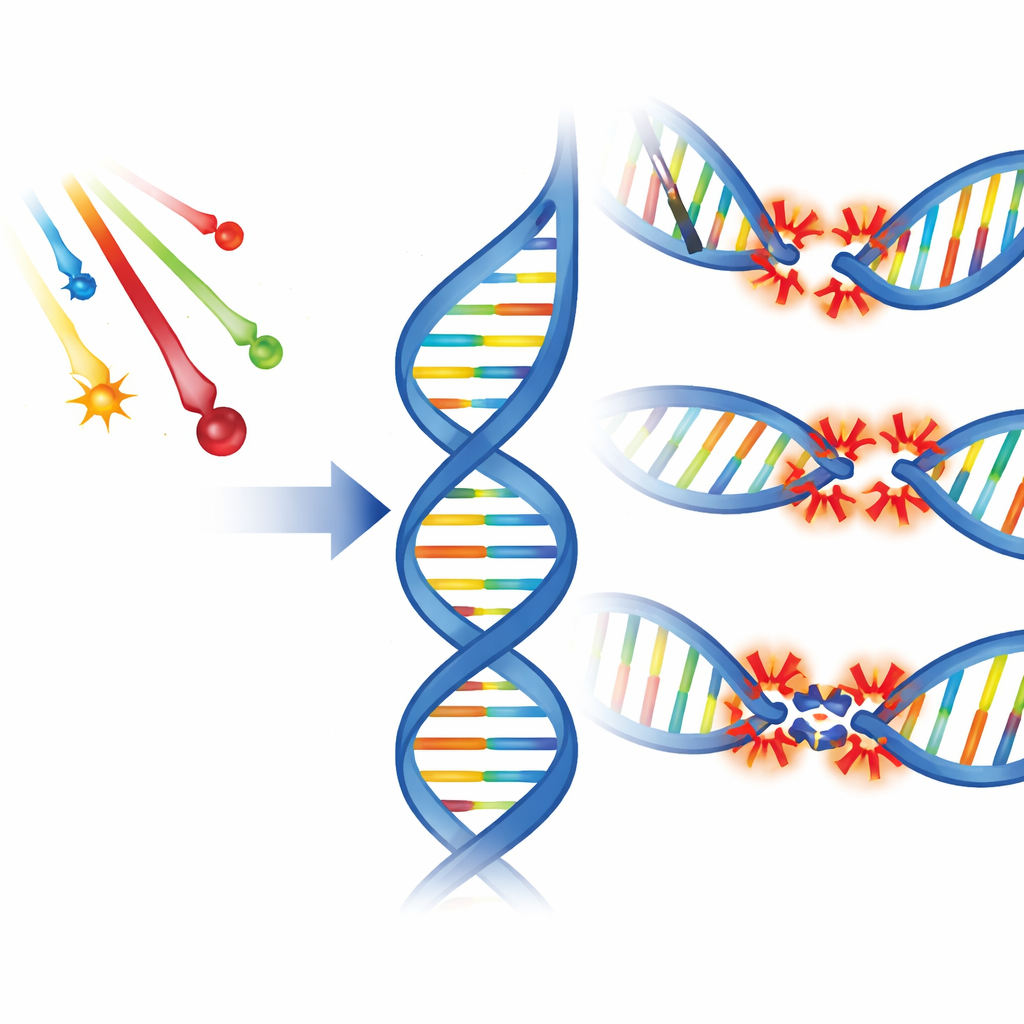
Different kinds of breaks in the DNA ladder
Ionizing radiation damages DNA in several basic ways. It can nick one strand of the double helix, cut both strands close together, or chemically alter the bases that encode genetic information. Scientists call these single-strand breaks, double-strand breaks, and base damage. Importantly, radiation often causes several such injuries packed within a few turns of the DNA helix. These crowded sites, known as clustered damage, come in two main flavors: bistranded clusters, where injuries face each other on opposite strands, and tandem clusters, where multiple injuries line up along the same strand. Experiments have shown that non–double-strand clusters—made mostly of base damage and single-strand breaks—are more common than outright double-strand breaks, especially at low doses.
Filling in the blind spots of experiments
Existing laboratory techniques can only see part of this picture. Enzyme-based assays and high-resolution imaging can detect many bistranded clusters, where injuries on opposite strands convert into visible breaks. But methods for counting tandem clusters, with multiple lesions along one strand only, are still lacking. That means our current measurements underestimate how many clustered injuries truly occur. To bridge this gap, the author builds a theoretical model that does not rely on seeing each lesion directly. Instead, it uses how much energy radiation deposits into a tiny DNA-containing volume, and how that energy is probabilistically split into different types of elementary lesions.
A probabilistic map of microscopic chaos
The heart of the work is a multinomial probability model: a mathematical framework that tracks how an incoming burst of energy can lead to several outcomes at once. In a nanoscale cylinder containing about 73 DNA base pairs, the model considers four possibilities whenever energy is deposited: direct hits to the DNA backbone that cause strand breaks, direct hits to bases that cause chemical damage, indirect hits to surrounding water that produce reactive radicals, and harmless energy absorbed by nearby proteins and other molecules. By combining these probabilities with detailed spectra of energy deposition for electrons and various ions, the model enumerates how often different combinations of lesions will appear—and crucially, how close they will lie to one another along or across the DNA strands.
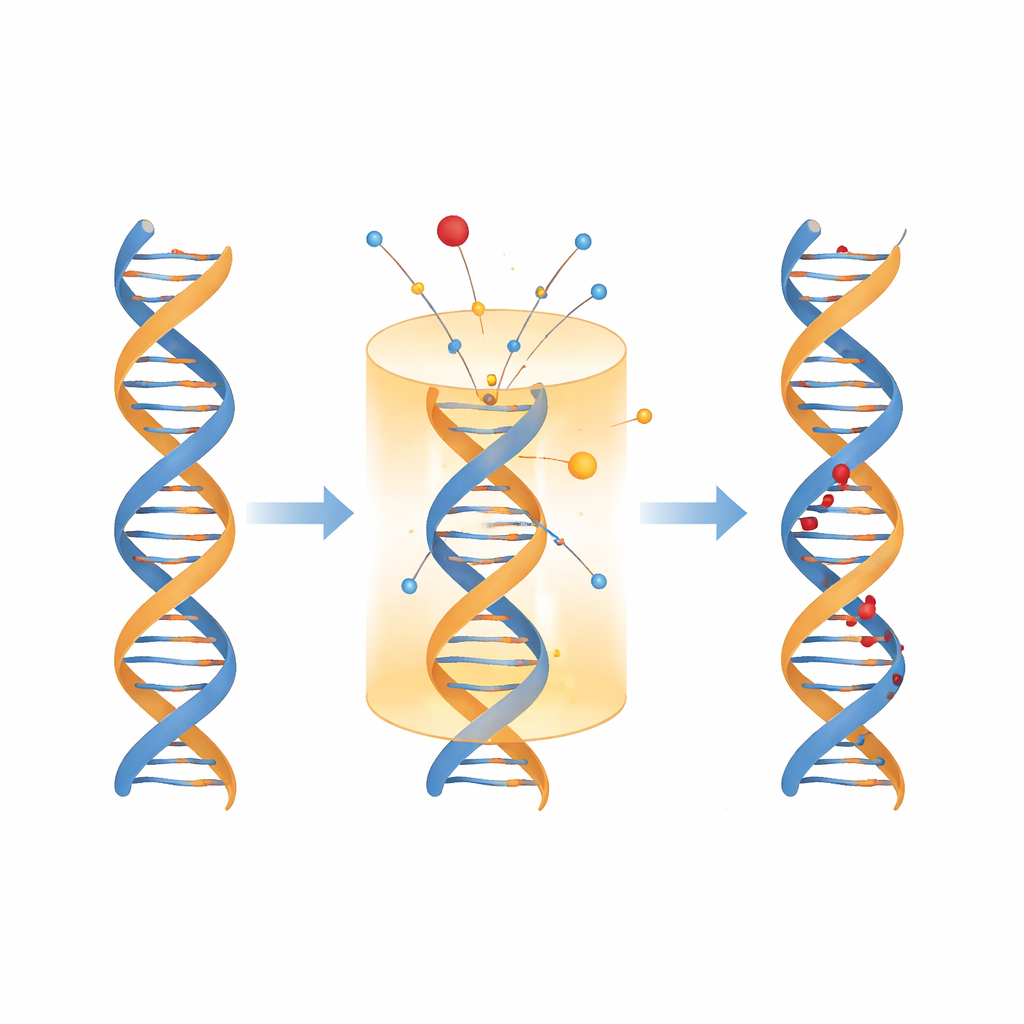
What the model reveals about radiation tracks
Applying this framework to electrons and ion beams used in cancer therapy and space environments, the study predicts more than 30 categories of DNA damage, including 24 types of clustered lesions. The calculations reproduce existing measurements of double-strand breaks for protons, helium, carbon, and iron ions, lending confidence to the unseen predictions about clusters. For typical medical and space-relevant conditions, the total number of non–double-strand clusters is estimated to be roughly four to six times higher than the number of double-strand breaks. Within those non-DSB clusters, tandem lesions appear surprisingly often: about half to three-fifths as frequent as bistranded clusters, with only a modest decline as radiation tracks become denser. The model also shows that most double-strand breaks themselves are “complex,” containing extra base damage nearby, which may complicate repair.
Implications for health, therapy, and spaceflight
Clustered, non-DSB damage is not just a bookkeeping detail. These crowded lesions are mainly processed by base excision repair pathways that can be slow and error-prone when many injuries sit in a small region. Repair attempts can convert non-DSB clusters into delayed double-strand breaks or mutations long after the initial exposure. The new probabilistic model provides a fast way to estimate these hidden injuries for any radiation type, without the heavy computational cost of full Monte Carlo track simulations. Its predictions suggest that radiation protection standards, cancer radiotherapy planning, and assessments of astronaut risks should pay as much attention to these subtle clusters as to the more obvious double-strand breaks.
Take-home message
In summary, this work shows that radiation produces far more tightly packed, non–double-strand DNA lesions than previously appreciated—several times more than outright double-strand breaks—and that tandem clusters along a single strand are nearly as common as bistranded ones. By marrying energy deposition data with multinomial probabilities, the model offers a practical tool for estimating these invisible injuries across many radiation types. For the layperson, the key message is that the most dangerous DNA damage from radiation may not be the rare, dramatic breaks we can easily see, but the numerous small wounds clustered together that quietly challenge the cell’s repair systems.
Citation: Cucinotta, F.A. Multinomial probability model of radiation induced DSB and non-DSB clusters: tandem and bistranded damage clusters. Sci Rep 16, 7877 (2026). https://doi.org/10.1038/s41598-026-36157-2
Keywords: clustered DNA damage, radiation biology, ionizing radiation, cancer radiotherapy, space radiation