Clear Sky Science · en
Photodissociation and electron-collision induced dissociation of C5H2F10 using photoelectron–photoion coincidence spectroscopy and quantum chemistry
Cleaning Up the Invisible Workhorses of Chipmaking
Modern smartphones, data centers, and AI hardware all rely on computer chips carved with astonishing precision. That carving is often done by reactive gases in plasma etching tools—workhorse chemicals that quietly shape the nanoscale tunnels and trenches inside advanced processors and memory. Many of the traditional gases are powerful greenhouse pollutants. This paper explores a promising replacement, a fluorinated gas called C5H2F10, and asks a practical question: when this gas is energized in a plasma, exactly how does it break apart and what useful fragments does it produce?

A New Gas for Smaller, Greener Devices
As chipmakers stack more and more layers vertically, they must etch very deep, very narrow holes with clean, straight walls. Fluorocarbon and hydrofluorocarbon gases excel at this, but some long-used choices have global warming potentials tens of thousands of times higher than carbon dioxide. C5H2F10 belongs to a newer family of molecules engineered to have far lower climate impact while still delivering fast, directional etching. To judge whether it can truly replace older gases, we must understand which charged and neutral fragments it turns into inside a plasma, because those fragments determine both etching performance and side effects such as damage or unwanted deposits.
Watching Molecules Break Apart in Real Time
The authors combined three powerful tools to track how C5H2F10 falls apart when energized. First, they used a synchrotron light source to fire vacuum‑ultraviolet photons at a jet of the gas and then recorded, in coincidence, the departing electrons and resulting ions. This photoelectron–photoion coincidence method allowed them to link specific energy inputs to specific breakup products. Second, they used an electron‑impact mass spectrometer, which mimics the way hot electrons in a real plasma collide with the gas. Finally, they ran detailed quantum‑chemistry calculations to map how bonds stretch, snap, or rearrange, and how much energy each step costs. Together, these approaches provide both a movie of what happens and a map explaining why.
Key Building Blocks Born from the Breakup
One striking result is that the original C5H2F10 ion is so unstable that it essentially never survives; it breaks instantly into fragments. At modest energies, the gas mainly splits by cutting carbon–carbon links near the most heavily fluorinated parts of the chain. That yields a handful of relatively large fragments, especially CF3+ and related pieces, that dominate the ion population. As the energy is raised, those larger fragments themselves begin to crack, and a particularly important product, CHF2+, appears in large amounts. Unlike fragments made by a single bond break, CHF2+ requires atoms to shuffle positions before part of the molecule departs. The authors used calculations to trace these rearrangements and showed that fluorine atoms migrate along the carbon backbone through low‑lying transition states, a prediction that matches the timing and intensity of the measured ion signals.
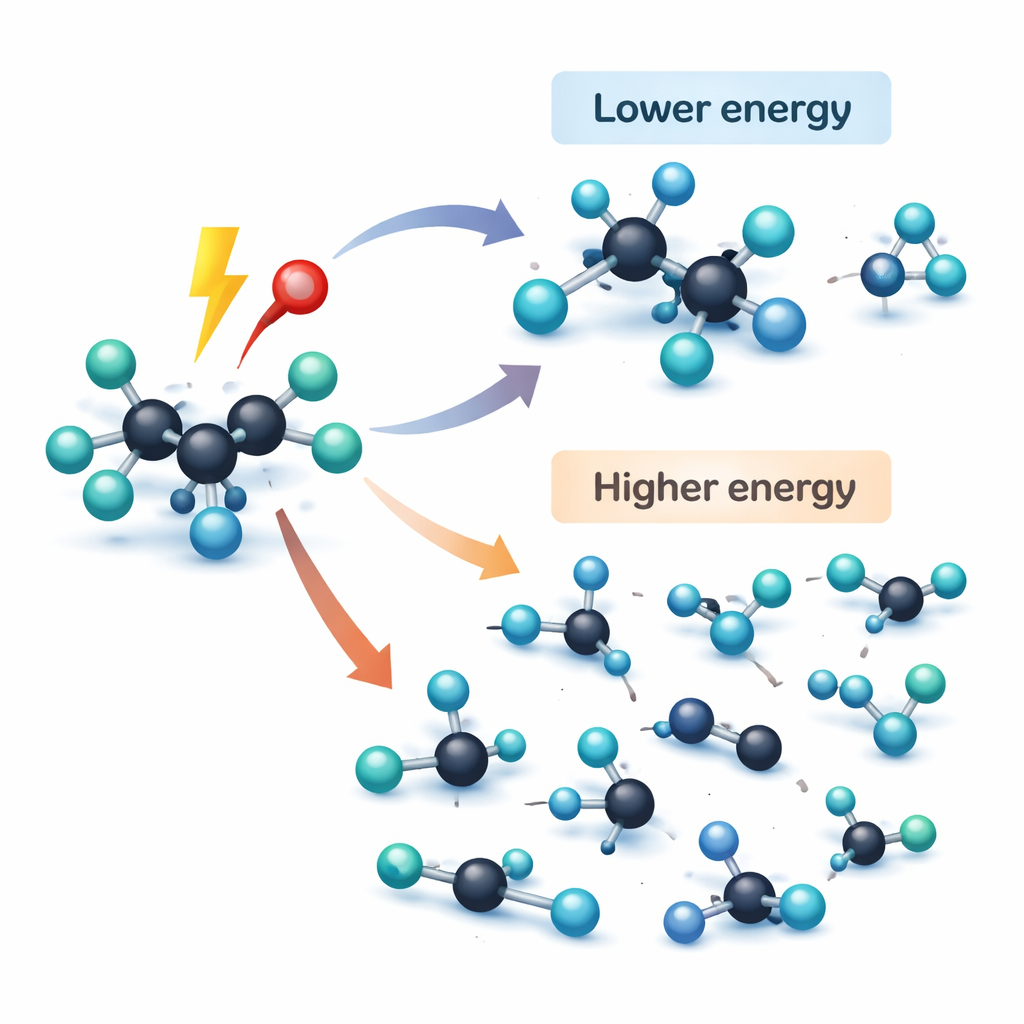
Tuning Fragmentation with Energy, Like a Control Knob
Comparing photon‑driven and electron‑driven experiments reveals that the way C5H2F10 breaks can be tuned like a control knob. At lower electron energies, similar to the milder parts of a plasma, the gas largely follows the same primary breakup channels seen in the light‑based measurements, producing a manageable set of fragments helpful for controlled etching. At higher electron energies, fragmentation becomes much more violent: larger ions disappear and are replaced by a swarm of smaller pieces. This behavior mirrors what happens in harsh industrial plasmas, where multiple collisions and high energies produce atomic fluorine and tiny fluorocarbon fragments that aggressively remove material but can also roughen surfaces if not balanced by gentler, hydrogen‑bearing species.
Why This Matters for Future Chips and the Climate
By pinning down exactly how C5H2F10 responds to different kinds and amounts of energy, the study offers chipmakers a recipe for using this lower‑warming gas without sacrificing precision. Knowing which fragments dominate under which conditions helps engineers design plasma settings that generate just enough fluorine‑rich ions to etch quickly, while also creating hydrogen‑containing species that protect sidewalls and improve selectivity between materials. The work shows that C5H2F10 can supply the same crucial mix of reactive building blocks as older, more climate‑damaging gases, but in a way that can be carefully tuned. In short, it lays the scientific groundwork for semiconductor manufacturing processes that are not only smaller and faster, but also significantly kinder to the planet.
Citation: Tran, N.T., Hayashi, T., Iwayama, H. et al. Photodissociation and electron-collision induced dissociation of C5H2F10 using photoelectron–photoion coincidence spectroscopy and quantum chemistry. Sci Rep 16, 5312 (2026). https://doi.org/10.1038/s41598-026-36140-x
Keywords: plasma etching, semiconductor manufacturing, low GWP fluorocarbons, molecular fragmentation, sustainable electronics