Clear Sky Science · en
Cytoplasmic TDP-43 leads to early behavioral impairments without neurodegeneration in a serotonergic neuron-specific C. elegans model
Why Tiny Worms Matter for Big Brain Diseases
Conditions like amyotrophic lateral sclerosis (ALS) and frontotemporal dementia (FTD) slowly rob people of movement, speech, and personality. A key suspect in these diseases is a protein called TDP-43, which clumps in the wrong place inside nerve cells. This study uses a tiny worm, Caenorhabditis elegans, to watch what happens in living animals when TDP-43 moves out of its normal home and into the cell’s cytoplasm. By focusing on a small, well-understood set of serotonin-producing neurons, the researchers show how behavior can go wrong long before neurons visibly die—offering a window into the earliest, most treatable stages of disease. 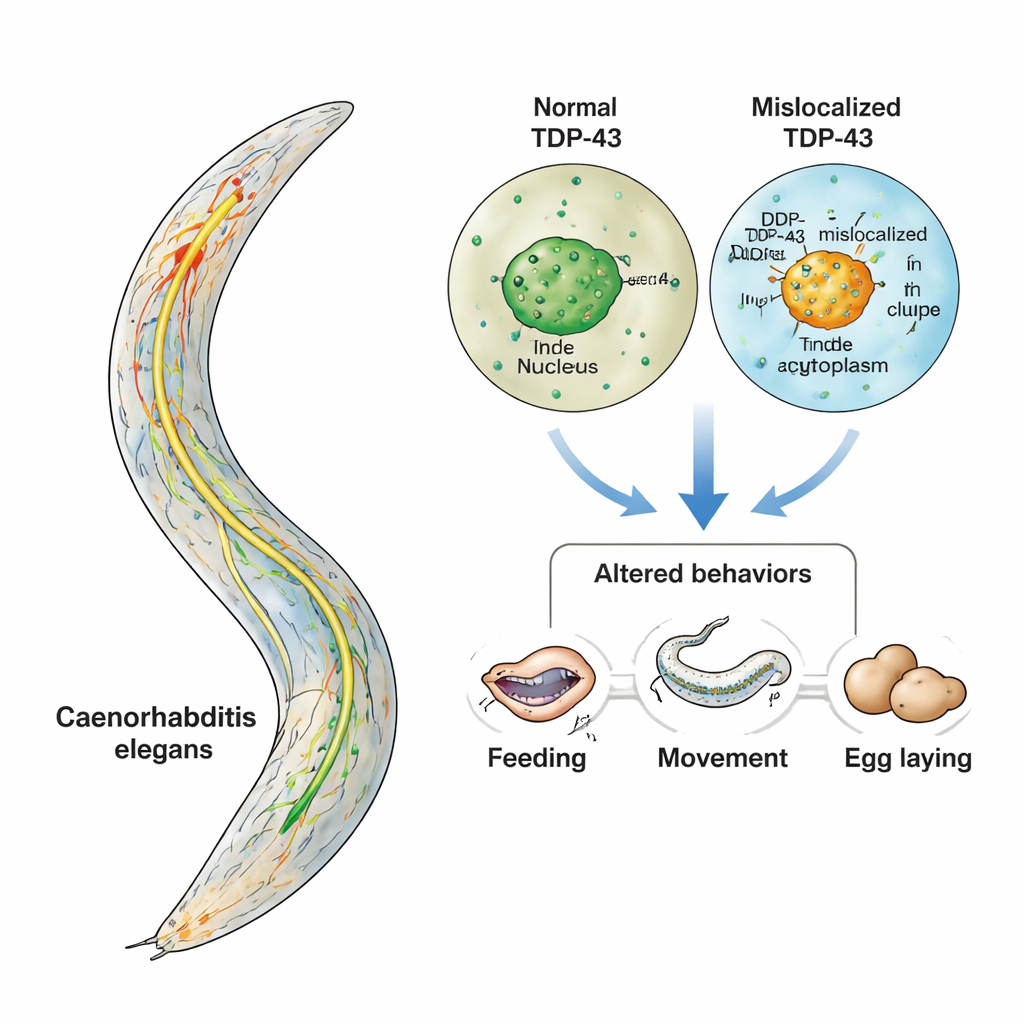
A Clumping Protein and a Chemical Messenger
TDP-43 normally lives in the nucleus, where it helps manage RNA, the cell’s working copies of genetic instructions. In ALS and FTD, however, TDP-43 often leaves the nucleus, builds up in the cytoplasm, and forms sticky aggregates. At the same time, evidence from patients and mouse models suggests that serotonin, a chemical messenger better known for its role in mood, is also disrupted in these diseases. Because serotonin influences many behaviors—from feeding to movement control—the authors asked a focused question: if TDP-43 is misplaced specifically in serotonin-producing neurons, do those neurons malfunction in ways that change behavior, even before they degenerate?
Rewiring a Simple Nervous System
The team turned to C. elegans, a microscopic worm whose entire nervous system of 302 neurons has been carefully mapped. Only three pairs of these neurons make serotonin, and each pair controls a well-defined behavior: feeding (pharyngeal pumping), slowing down when food is found, and egg-laying. Using genetic tools, the researchers engineered worms so that only these serotonergic neurons produced human TDP-43. One strain made the normal, nucleus-localized form; another made a mutant version lacking its nuclear “address tag,” forcing TDP-43 to accumulate in the cytoplasm. Because all other neurons were left untouched, any changes in behavior could be traced directly to problems in this single, tiny circuit.
Behavior Fails Before Neurons Die
Across several tests, the altered worms behaved abnormally in ways that closely mirrored partial or severe loss of serotonin. Worms with the cytoplasmic version of TDP-43 showed slower rhythmic pumping of their feeding organ, a weaker tendency to slow down when they encountered food after fasting, and reduced egg-laying. Worms expressing the normal, nuclear TDP-43 also performed worse than unmodified animals, but not as badly as those with cytoplasmic TDP-43, revealing a clear gradient of impairment. Crucially, when the same animals were tested in a context where serotonin is not needed—swimming in liquid—their movement was normal. This showed that the core locomotor machinery was intact, and that the defects were specific to serotonin-dependent behaviors rather than a general decline in health. 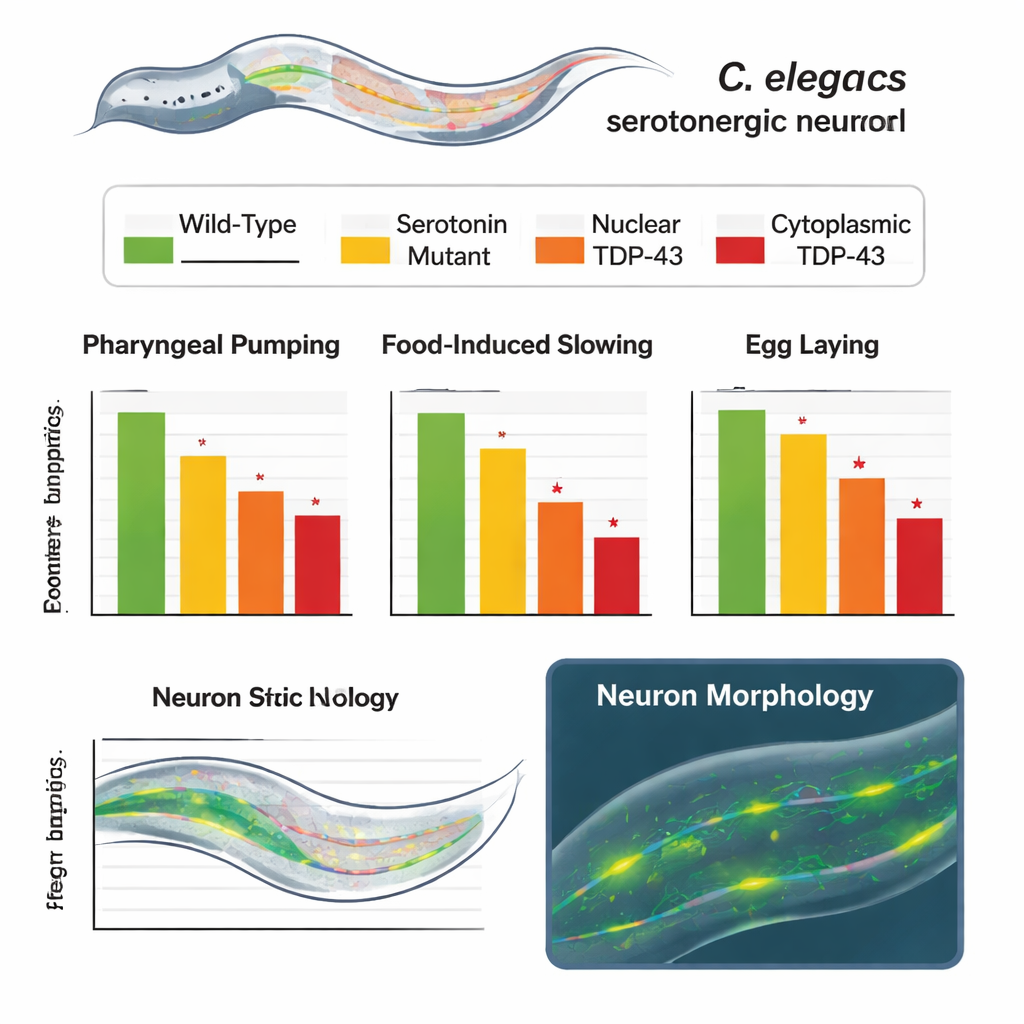
Hidden Damage Revealed by a Drug Test
To find out whether the affected neurons were still alive and functional, the researchers used fluoxetine, better known as Prozac, a drug that enhances serotonin signaling. In normal worms, fluoxetine sharply boosts egg-laying. In the engineered worms, the drug still increased egg-laying, but the response was blunted—especially in those with cytoplasmic TDP-43. This pattern suggests that the serotonin neurons could still release signals, but did so less effectively. Microscopic imaging told a similar story: the serotonin-producing neurons looked structurally normal, with no loss of cell bodies or major distortions in their branches, and their overall fluorescence—a proxy for cell health—matched that of control worms.
Early Warning Signs and New Opportunities
Taken together, the findings paint a picture in which mislocalized TDP-43 can seriously disrupt how serotonin neurons work while leaving them apparently intact to the eye. In the worms, behavior goes wrong—feeding, movement adjustment, and reproduction—before there is any visible neuron death. This echoes clinical observations that subtle behavioral and mood changes often precede obvious brain shrinkage in human neurodegenerative disease. By offering a simple, genetically tractable system where early, circuit-specific defects can be measured precisely, this worm model could help scientists probe how TDP-43 derails neuron function and test therapies aimed at rescuing neurons during this vulnerable, predegenerative window.
Citation: Lacour, A., Vassallu, F., Romussi, S. et al. Cytoplasmic TDP-43 leads to early behavioral impairments without neurodegeneration in a serotonergic neuron-specific C. elegans model. Sci Rep 16, 6068 (2026). https://doi.org/10.1038/s41598-026-36138-5
Keywords: TDP-43, serotonin neurons, C. elegans, ALS and FTD, neurodegeneration