Clear Sky Science · en
The single-cell transcriptional landscape of the pediatric cystic fibrosis lung from minimally invasive respiratory specimens
Why this matters for children with cystic fibrosis
Cystic fibrosis is best known for thick mucus and lung infections, but underneath those symptoms lies a complex battle between immune cells and the delicate lining of the airways. This study zooms in on that battle in children, cell by cell, using tiny samples collected during routine lung checks. By mapping which cells are present and how active they are, the researchers show that harmful patterns of inflammation start early in life and may be hard to undo, even with modern medicines that fix the root gene defect.
Peeking inside young lungs one cell at a time
To explore what is happening inside children’s lungs, the team used minimally invasive tools during flexible bronchoscopy—procedures these patients were already undergoing for medical reasons. They gently brushed the inner surface of the windpipe and bronchi and collected wash fluid from deeper in the lungs. These samples came from two children with cystic fibrosis (an infant and a teenager) and three age-matched children without the disease. Using single-cell RNA sequencing, a technique that reads which genes are switched on in thousands of individual cells, they built a high-resolution map of immune cells and airway-lining cells in each child’s lungs. 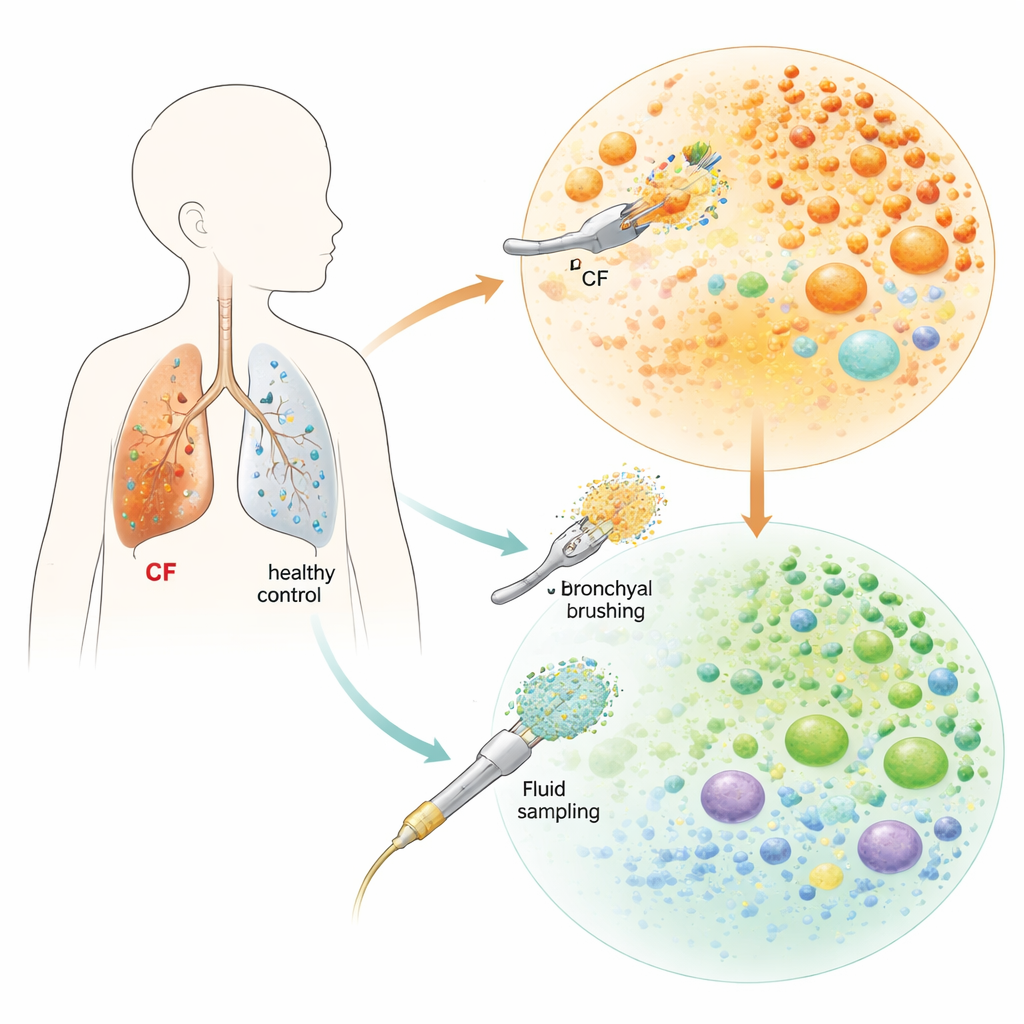
A crowded field of first-responder immune cells
In children without cystic fibrosis, the lung wash samples were dominated by macrophages—cleanup cells that patrol the airspaces and help keep peace. In contrast, the children with cystic fibrosis showed a dramatic shift toward neutrophils, fast-acting “first responder” white blood cells that can damage tissue when they linger. Not only were neutrophils more abundant, they appeared in several distinct flavors. These included immature forms, highly activated inflammatory cells, and a striking subset marked by stress-response proteins known as heat shock proteins. While such stress responses may begin as protective, they can lock neutrophils into a long-lived, aggressive state that keeps inflammation burning and may drive the airway damage seen in cystic fibrosis.
Inflamed defenders and missing reinforcements
The macrophages that remained in cystic fibrosis lungs also looked different. Instead of a mix of common housekeeping types seen in the comparison group, the cystic fibrosis samples were enriched for macrophages that produce strong immune signals, suggesting a shift toward a more inflammatory role. At the same time, there were relatively few T cells—another key arm of the immune system—in the airway fluid of children with cystic fibrosis. Previous work has shown that neutrophils can starve T cells of the nutrients they need to function, effectively pushing them out of the airway. This combination of aggressive neutrophils, activated macrophages, and scarce T cells may help explain why cystic fibrosis lungs remain inflamed and prone to infection over many years.
An airway lining that joins the fight
The brush samples allowed the researchers to look at the cells that line the airways: basal cells (which can replenish the lining), ciliated cells (with hairlike projections that sweep mucus), and secretory cells (which produce mucus and protective molecules). The overall mix of these cell types was similar between cystic fibrosis and comparison children, but their behavior was not. In cystic fibrosis, secretory cells switched on genes linked to immune defense and antimicrobial activity, suggesting they are working overtime as part of the inflammatory response. Ciliated cells also turned on genes tied to inflammation and resistance to a form of cell death related to iron and oxidative damage. Together, these patterns point to an airway surface that is not just a bystander but an active, inflamed participant in the disease process. 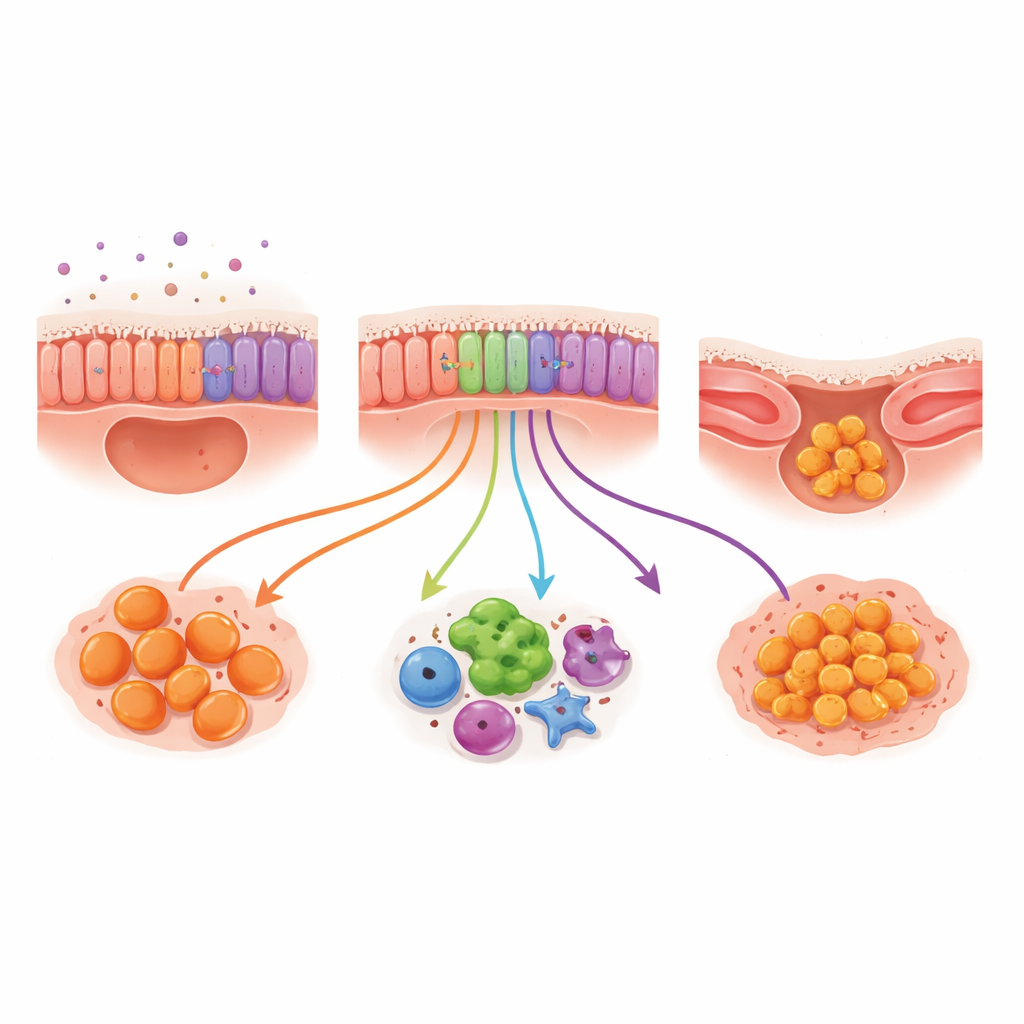
Cell-to-cell conversations that fuel lung damage
To understand how airway-lining cells and neutrophils influence each other, the team analyzed “conversations” between them—signals sent by one cell type and received by another. They found that epithelial cells often acted as senders, using several molecular systems to reach neutrophils. Some of these signals likely help calm inflammation, while others encourage neutrophils to swarm in, activate, and engulf material. This push-and-pull suggests the airway lining is trying to balance damage control and defense, but in cystic fibrosis the pro-inflammatory signals appear to win out, keeping the lung in a chronic state of smoldering injury.
Why early changes are hard to reverse
A key finding is that these inflammatory patterns were present both in an infant and in a teenager who had been taking an advanced cystic fibrosis drug that improves the faulty protein. Despite this targeted therapy, the teenager’s lung cells still resembled those of the untreated infant more than those of the comparison children. This implies that once structural damage and entrenched inflammation have developed, simply restoring the underlying protein function may not be enough to reset the immune and epithelial landscape. The study also shows that gentle sampling of the airways, combined with single-cell and spatial gene analysis, can track these changes over time. This approach may help design and test new anti-inflammatory and antimicrobial treatments aimed at preserving lung structure from the very beginning of life.
Citation: Sun, Y., Vicencio, A.G., Beasley, M.B. et al. The single-cell transcriptional landscape of the pediatric cystic fibrosis lung from minimally invasive respiratory specimens. Sci Rep 16, 8113 (2026). https://doi.org/10.1038/s41598-026-36125-w
Keywords: cystic fibrosis lung, neutrophilic inflammation, single-cell RNA sequencing, airway epithelium, pediatric bronchoscopy