Clear Sky Science · en
Comprehensive assessment of gold nanorod-induced genotoxicity using multi-model biological systems
Why tiny gold rods matter to our DNA
Gold may bring to mind jewelry or finance, but in modern medicine, it is also being reshaped into tiny rods thousands of times thinner than a human hair. These “gold nanorods” can help doctors see tumors more clearly, heat and kill cancer cells, and deliver drugs with high precision. Yet the same unusual properties that make these particles so powerful also raise a crucial question: do they damage the genetic material inside our cells? This study takes a broad look across bacteria, yeast, and human liver cancer cells to find out how gold nanorods interact with DNA and what that might mean for both medical treatments and safety regulations. 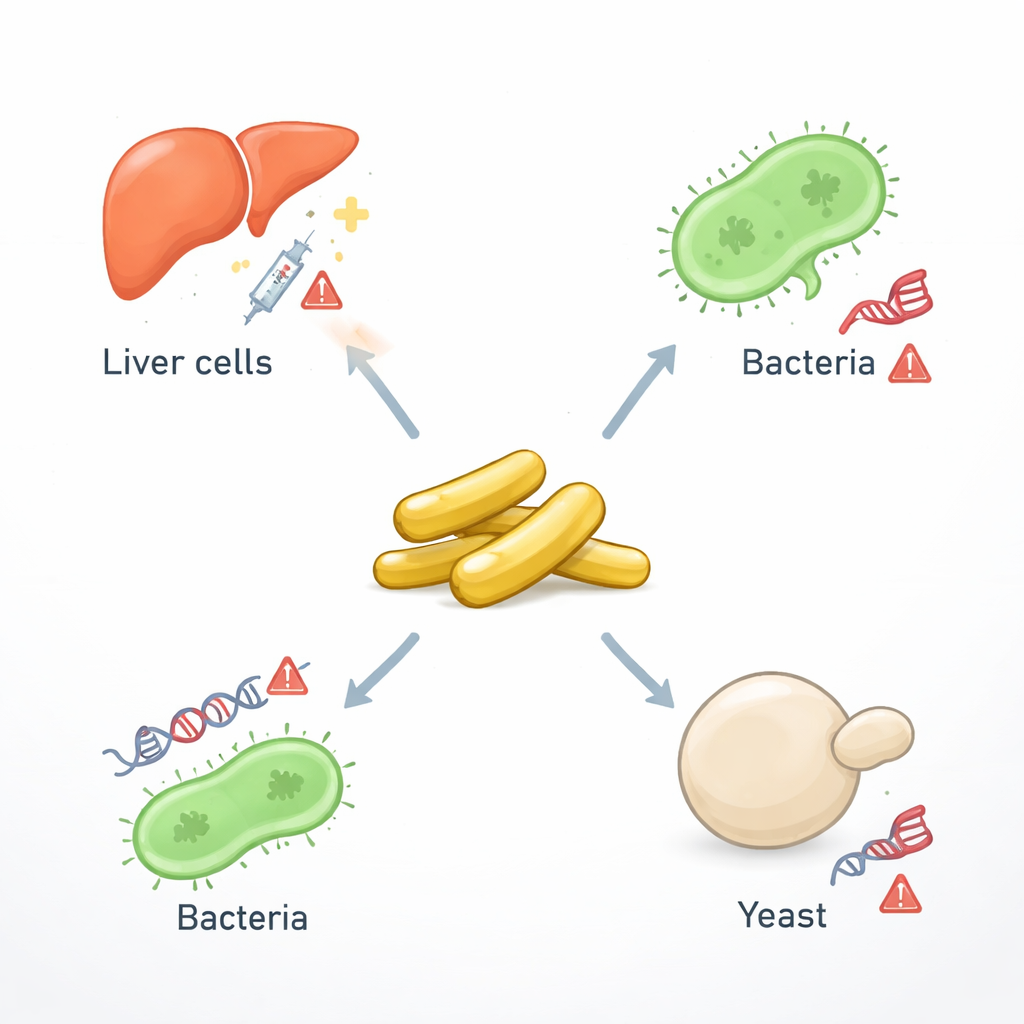
From test tubes to living cells
To explore the risks and benefits of gold nanorods, the researchers first prepared uniform rod-shaped particles about 50 nanometers long—far too small to see with the naked eye, but easily able to slip into cells. They then tested these particles in several biological systems that together form a kind of “toxicity pipeline.” Two common bacteria, Salmonella typhimurium and Escherichia coli, were used as fast, simple sensors of DNA damage. Specially engineered yeast strains, each missing a single gene linked to stress responses or cell death, provided a more human-like model because yeast share many core pathways with us. Finally, human liver cancer cells (HepG2), which are widely used to test drugs and chemicals, were exposed to gold nanorods so the team could track changes in key cancer-related genes.
Watching DNA tear into comet tails
Across bacteria and yeast, the scientists relied on a sensitive technique called the comet assay to “see” DNA damage one cell at a time. In this method, cells are embedded in gel, gently broken open, and placed in an electric field. Intact DNA stays mostly in place, but broken strands stream out, forming a shape that resembles a comet with a bright head and trailing tail. By measuring how long and bright the tail is, researchers can estimate the extent of genetic damage. In both Salmonella and E. coli, gold nanorods caused clear, dose-dependent increases in all comet measures: more cells with tails, longer tails, and a higher percentage of DNA pulled into the tail. Yeast knockout strains showed the same pattern, with some strains—especially those missing genes tied to stress and mitochondrial function—displaying markedly more DNA fragmentation than normal yeast. 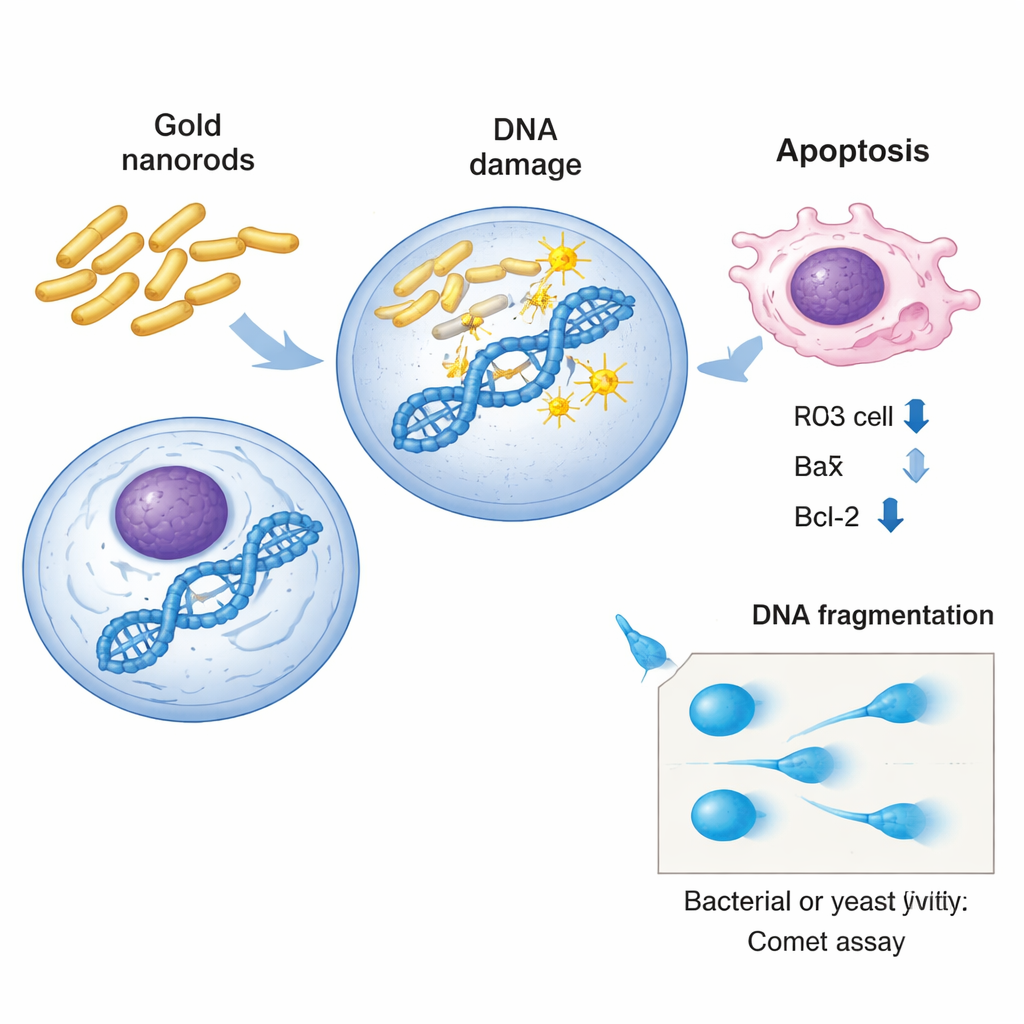
Gene networks and death signals in human cells
In human liver cancer cells, the team looked deeper, beyond physical breaks in DNA, to see how the cells’ internal control systems responded. Using real-time PCR, they measured the activity of three well-known guardians and executioners of cell fate: p53 and Bax, which promote cell death when damage is detected, and Bcl-2, which helps cells survive. After exposure to half-strength toxic doses of gold nanorods, p53 and Bax levels rose, while Bcl-2 levels fell, a molecular signature of programmed cell death (apoptosis). In other words, the nanorods not only injured the DNA but also pushed cancer cells toward self-destruction. To connect the yeast results with human biology, the researchers used the GeneMANIA platform to map interaction networks around the yeast genes they had deleted. This analysis showed dense webs of physical and genetic interactions linked to stress response, DNA repair, and mitochondrial function, reinforcing the idea that the same kinds of pathways are vulnerable in both yeast and human cells.
Balancing medical promise with genetic risk
Taken together, these experiments paint a nuanced picture of gold nanorods. On one hand, they clearly can harm DNA across very different organisms, and the damage rises with increasing dose. Certain genetic backgrounds, such as yeast strains lacking specific stress or mitochondrial genes, are especially sensitive, hinting that people with particular genetic traits might also respond differently. On the other hand, in human liver cancer cells, this DNA damage and the resulting activation of death pathways could be precisely what doctors want when targeting tumors. For a layperson, the key message is that gold nanorods are powerful tools that can both help and hurt: they can kill cancer cells, but they can also pose genetic risks to other cells and to the environment. The study argues that future medical uses of these particles must carefully weigh dosage, target delivery, and patient genetics to harness their benefits while keeping their genotoxic potential firmly under control.
Citation: Rashad, S.E., Haggran, A.A. & Abdoon, A.S.S. Comprehensive assessment of gold nanorod-induced genotoxicity using multi-model biological systems. Sci Rep 16, 5429 (2026). https://doi.org/10.1038/s41598-026-36119-8
Keywords: gold nanorods, DNA damage, nanotoxicology, cancer therapy, genotoxicity testing