Clear Sky Science · en
R5 and R7 positions on fluoroquinolone scaffolds drive F-actin filament disruption
Turning Antibiotics into Brain Helpers
Many common brain disorders, from Alzheimer’s to Parkinson’s disease, share a hidden culprit: tiny structural rods inside nerve cells that become jammed and rigid over time. These rods are made of a protein called actin, normally a flexible part of the cell’s inner scaffolding. This study explores a surprising possibility—that a well-known class of antibiotics, fluoroquinolones, could be redesigned to gently break apart these harmful actin bundles and potentially help protect the brain.
When Cell Scaffolds Go Wrong
Healthy brain cells constantly reshape their internal skeleton to form and remodel connections. Actin filaments are central to this process, forming and dissolving as needed. Under stress, however, actin can lock into stubborn rod-like aggregates that clog the cell, disrupt transport of vital cargo, and encourage the buildup of disease-linked proteins such as amyloid and tau. Over years, these rods can mature into dense structures known as Hirano bodies, which are commonly seen in the brains of people with several neurodegenerative conditions. Because existing actin-targeting drugs are often extremely toxic, researchers are keen to find safer small molecules that can loosen these aggregates without harming cells.
An Unexpected Talent of Familiar Drugs
Fluoroquinolones are broad-spectrum antibiotics used worldwide to treat infections, and some newer members of this family can cross the blood–brain barrier. Earlier hints suggested that certain fluoroquinolones could destabilize actin filaments. In this work, the authors systematically compared seven related compounds, spanning four generations of the drug family, to see how well they break apart pre-formed actin filaments rather than merely preventing them from forming. Using light-scattering measurements, electron microscopy, and gel filtration, they showed that all tested drugs could disrupt actin filaments, but with very different strengths. Two drugs, sparfloxacin (a third-generation compound) and moxifloxacin (fourth generation), stood out by rapidly and irreversibly chopping long filaments into much smaller pieces, even at relatively low drug-to-protein ratios.
Seeing Filaments Fall Apart
High-resolution imaging and biophysical tests revealed how the filaments change under drug treatment. Electron microscopy showed that untreated actin forms long, continuous strands, whereas effective fluoroquinolones convert these into short, scattered fragments. Less potent members produced kinked and bent filaments but did not fully break them. Heat-based measurements of protein stability confirmed this physical disruption: once treated, actin behaved more like its free, unassembled form, with lower melting temperatures, yet its basic building-block structure stayed almost unchanged. This suggests that the drugs do not unfold or damage actin itself—they mainly pry apart the way the units stick together in filaments.
Zooming In on the Molecular Grip
To understand why some fluoroquinolones work better than others, the team combined advanced nuclear magnetic resonance experiments with computer simulations of drug–protein interactions. They found that all compounds rely on a shared ring-shaped “core” to make contact with actin, especially at two positions on that core that repeatedly touch the filament. However, extra chemical groups attached at specific sites—called R5 and R7—strongly influence how firmly and where each molecule binds. In the most effective disruptors, these positions carry an amino group and bulky ring systems that reach into a small pocket at the interface between neighboring actin strands. There, they interfere with key stabilizing contacts, including an important salt bridge that normally helps hold filaments together.
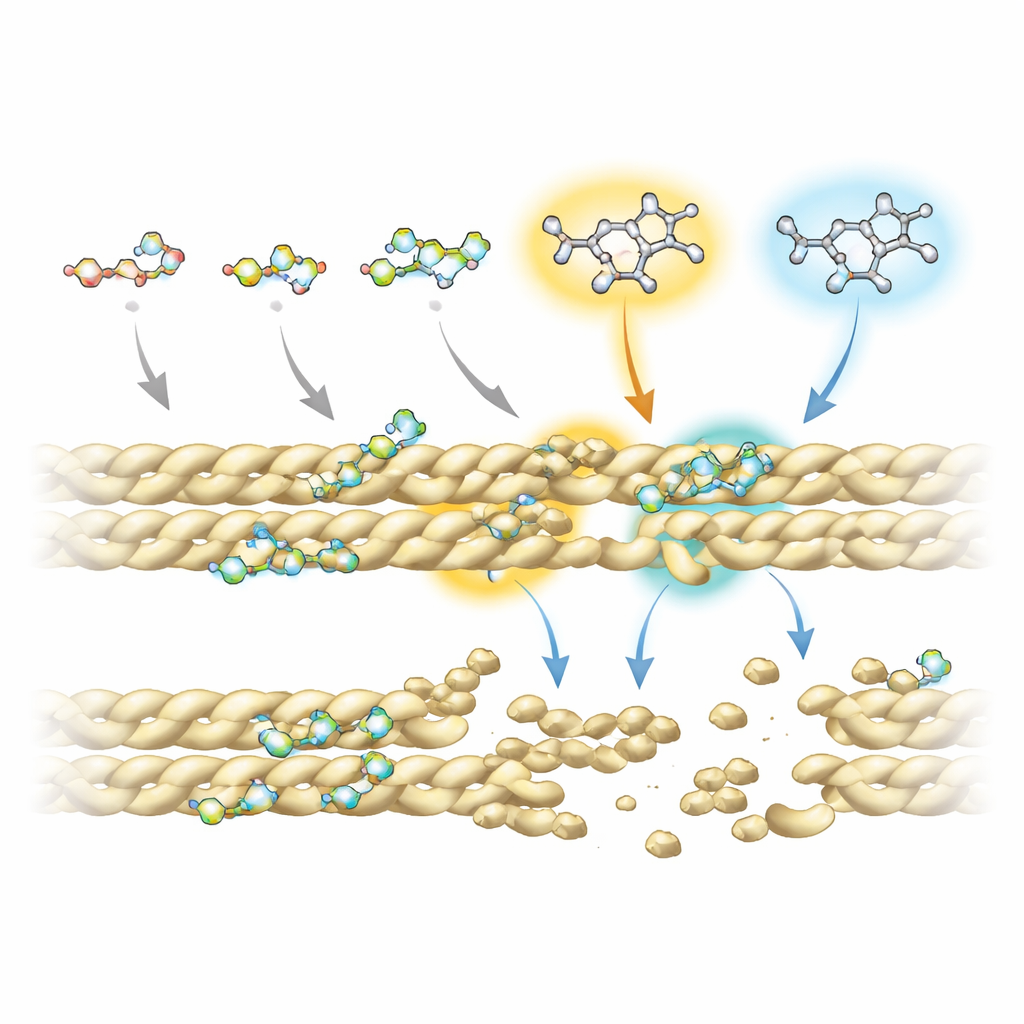
From Yeast Cells to Future Therapies
The researchers also tested the drugs in a yeast strain that naturally accumulates actin bundles, mimicking aspects of diseased cells. Under the microscope, untreated yeast showed large, bright actin clumps, while cells exposed to fluoroquinolones—especially sparfloxacin, moxifloxacin, and, at higher doses, nalidixic acid—displayed far more diffuse, punctate patterns, indicating that the bundles had been broken down. Importantly, the drugs’ action appeared relatively gentle: they weakened filament packing without grossly distorting actin’s underlying shape, and their binding was weaker than that of classic actin toxins, potentially reducing the risk of severe side effects.
Designing Smarter Actin-Targeting Drugs
Overall, this study shows that minor tweaks to well-known antibiotics can dramatically change how they interact with the cell’s internal scaffolding. By pinpointing the R5 and R7 positions as key “control knobs” for both actin disruption and brain penetration, the authors outline a blueprint for creating new fluoroquinolone-inspired molecules that selectively dismantle harmful actin aggregates. While these findings are an early, laboratory-stage proof of principle, they raise the possibility that future drugs derived from familiar antibiotics could help clear toxic scaffolding in nerve cells and slow or prevent some forms of neurodegeneration.
Citation: Gupta, R.J., Nair, H., Sarhadi, T.R. et al. R5 and R7 positions on fluoroquinolone scaffolds drive F-actin filament disruption. Sci Rep 16, 9517 (2026). https://doi.org/10.1038/s41598-026-36089-x
Keywords: actin aggregates, fluoroquinolone antibiotics, neurodegenerative disease, drug repurposing, cytoskeleton