Clear Sky Science · en
The role of cellular senescence-related genes in ischemia–reperfusion injury and the identification of their biomarkers
Why heart damage can worsen when blood returns
When someone has a heart attack or undergoes heart surgery, doctors race to restore blood flow to starving heart muscle. Paradoxically, that life‑saving step can itself cause extra harm—a problem known as ischemia–reperfusion injury. This study explores why some heart cells respond so badly when blood returns, focusing on the biology of cellular aging and identifying a small set of genes that could help doctors spot and eventually treat this hidden damage more precisely.
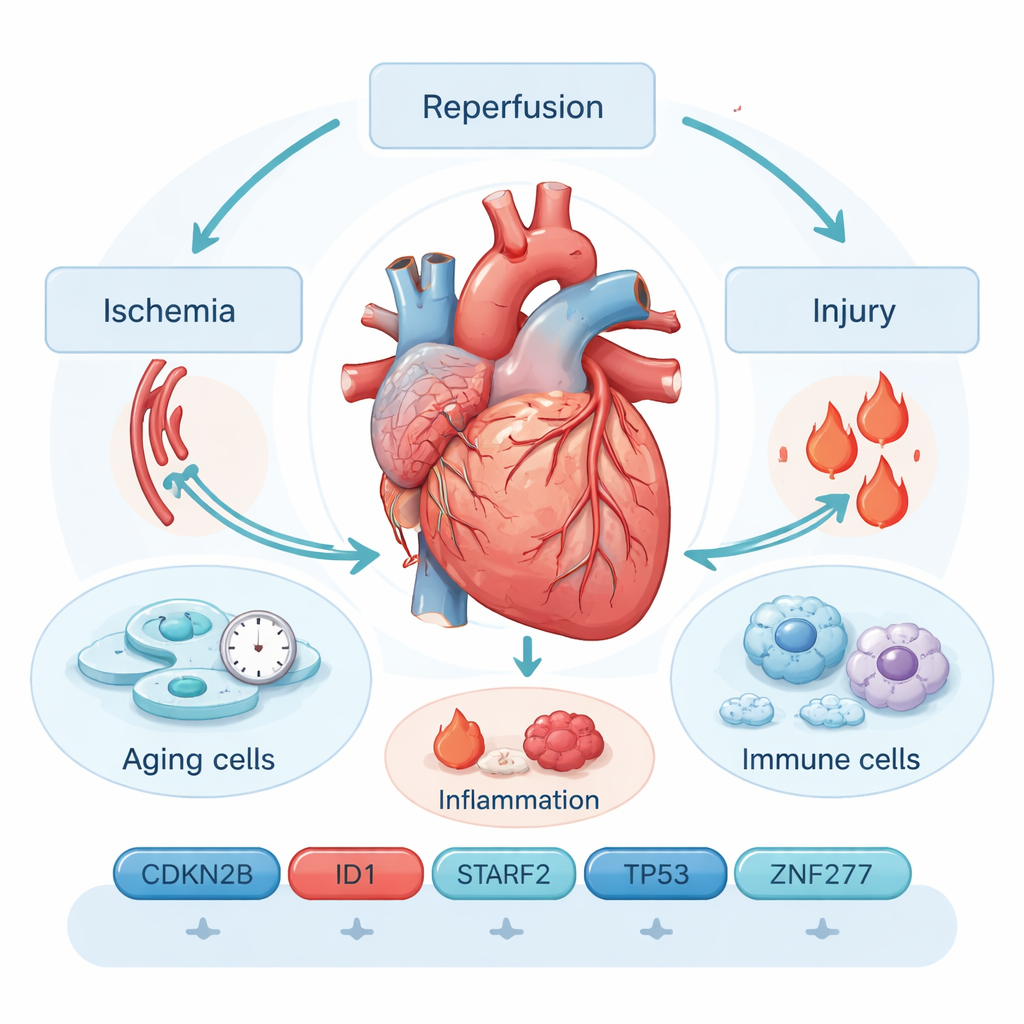
When rescue turns into renewed harm
Ischemia–reperfusion injury happens in two acts. First, a coronary artery is blocked and heart tissue is starved of oxygen (ischemia). Then, when the blockage is cleared and blood rushes back (reperfusion), a burst of reactive molecules and inflammation can damage already fragile cells. Current tools—such as ECGs, imaging scans, and blood tests for proteins like troponin—often miss early or subtle injury and cannot easily predict who will suffer long‑term problems. That gap has driven researchers to look deeper, at the molecular events inside heart cells themselves.
Cellular aging as a hidden driver
The authors focus on cellular senescence, a state in which cells stop dividing permanently and adopt an “alarm‑sending” behavior. Senescent heart cells secrete a stew of inflammatory molecules and enzymes known as the senescence‑associated secretory phenotype. This chemical cloud can worsen scarring, attract immune cells, and interfere with tissue repair, creating a vicious cycle of injury and chronic dysfunction. By mining public gene‑expression datasets from mouse hearts exposed to ischemia–reperfusion, and cross‑referencing them with hundreds of genes linked to cellular aging, the team narrowed the field to 26 genes that change markedly after injury.
Six genes that form a diagnostic fingerprint
To find the most informative signals among those 26 genes, the researchers used several machine‑learning methods. These algorithms search for combinations of genes that best distinguish injured hearts from healthy ones. After logistic regression, LASSO, and support vector machine analyses, six genes emerged as a powerful diagnostic panel: CDKN2B, ID1, STAT3, TERF2, TP53, and ZNF277. Together, their activity patterns could identify ischemia–reperfusion injury with very high accuracy in the combined datasets, outperforming many traditional blood markers in internal tests. Gene‑pathway analyses tied these markers to stress‑response circuits involving oxidative damage, cell‑death programs, and well‑known signaling routes such as MAPK and PI3K–AKT, all of which shape whether a cell recovers or slides into permanent senescence.
How the immune system joins the story
Damage from restored blood flow does not happen in isolation; immune cells quickly flood into the heart. By estimating immune cell activity from the same gene‑expression data, the study found clear signs of heightened immune involvement in injured hearts, including several types of T cells, B cells, macrophages, and mast cells. One aging‑related gene, CDKN2B, strongly tracked with “central memory” CD4 T cells, while TP53 correlated with activated mast cells. These links suggest that aging pathways in heart cells and changes in the immune system reinforce each other: older‑acting cells call in immune help, and those immune responses can, in turn, deepen injury and scarring.
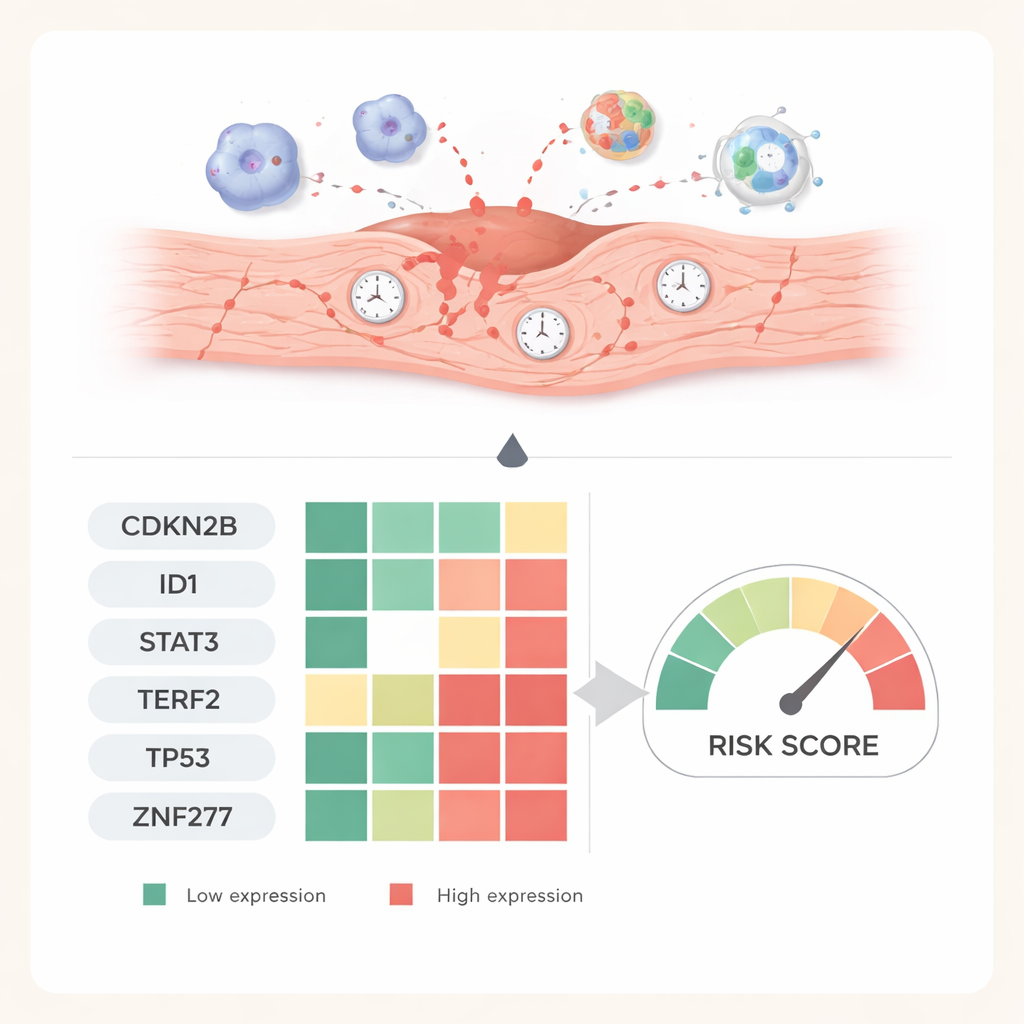
From computer predictions to living hearts
To test whether these six genes matter beyond computer screens, the researchers created a mouse model of ischemia–reperfusion injury. They measured gene activity at different times after blood flow was restored and used tissue staining to visualize the corresponding proteins in heart slices. One gene, CDKN2B, steadily rose over a week, hinting that it helps lock damaged cells into a long‑term senescent state. Others, including ID1, STAT3, TP53, TERF2, and ZNF277, dipped sharply early on and then gradually recovered, mirroring shifts between acute stress, active damage, and later repair. These time‑dependent patterns support the idea that each gene captures a different phase of the heart’s response.
What this could mean for patients
For a layperson, the key message is that the study links heart‑attack‑related injury to the biology of cellular aging and identifies a six‑gene “fingerprint” that flags this damage in experimental models. While the work is still preclinical and based mainly on mouse data and public databases, it offers a roadmap for new blood or tissue tests that could detect ischemia–reperfusion injury earlier and more specifically than today’s tools. In the long run, understanding how these aging‑related genes steer inflammation, scarring, and repair may open doors to treatments that not only reopen blocked arteries, but also help the rejuvenated heart heal more completely.
Citation: Sun, L., Liu, H., Jia, T. et al. The role of cellular senescence-related genes in ischemia–reperfusion injury and the identification of their biomarkers. Sci Rep 16, 5211 (2026). https://doi.org/10.1038/s41598-026-36076-2
Keywords: ischemia reperfusion injury, cellular senescence, heart attack, biomarkers, immune inflammation