Clear Sky Science · en
Expression of sphingosine-1-phosphate receptor 1 in the brain of fatal cerebral malaria
Why this brain–malaria link matters
Cerebral malaria is one of the deadliest complications of malaria infection, killing many patients even when they receive modern drugs. This study looks inside the brain to understand why blood vessels start to leak and fail in the sickest patients. By focusing on a fatty signaling molecule called sphingosine-1-phosphate (S1P) and its receptor S1PR1, the researchers ask whether this chemical conversation between blood and brain might explain who lives, who dies, and how we might better protect the brain in future therapies.
What happens to the brain in severe malaria
In cerebral malaria, red blood cells infected with the parasite Plasmodium falciparum become sticky and latch onto the inner lining of tiny brain blood vessels. The team examined preserved brain tissue from people who died of malaria and compared them with patients who had milder, non-cerebral malaria and with non-malaria controls. In the fatal cerebral cases, they saw blood vessels crammed with parasite-filled red cells, tiny pinpoint bleeds, ring-shaped areas of bleeding, and clumps of support cells known as Dürck granulomas. These changes signal clogged vessels, poor oxygen delivery, and damage to the normally tight blood–brain barrier that keeps the brain’s environment stable. 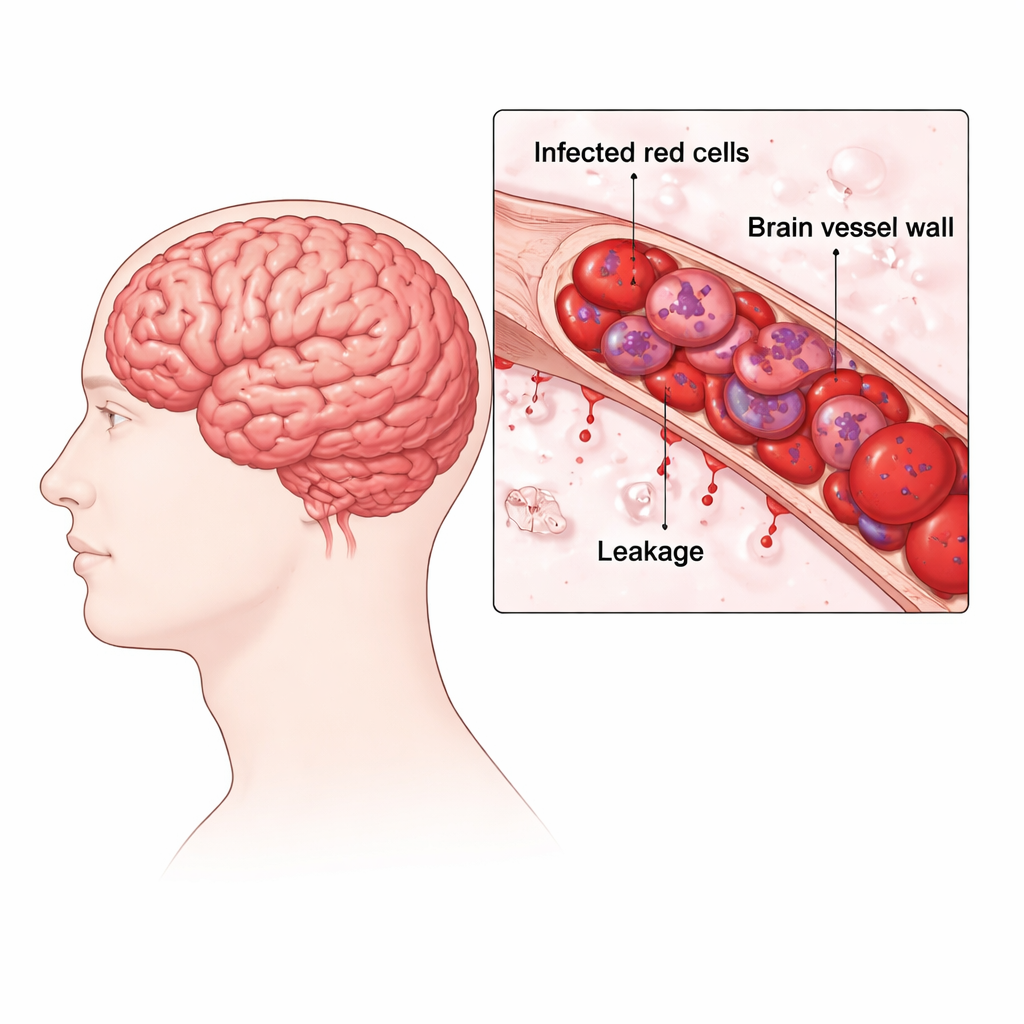
A chemical guardian of vessel tightness
Under healthy conditions, S1P—stored mainly in red blood cells and platelets—helps blood vessels stay sealed. It does this by binding to receptors such as S1PR1 on the surface of vessel-lining cells and brain cells, tightening the junctions between them. The researchers measured S1P levels in blood samples and found a striking drop in patients with cerebral malaria: their S1P levels were roughly four times lower than in people without malaria, and about half those of patients with non-cerebral malaria. This suggests that as malaria destroys or deforms red blood cells and injures vessel cells, the body’s supply of this protective molecule dwindles just when it is most needed.
Where and how the receptor lights up
Next, the team used immunohistochemistry—a staining method that makes specific proteins visible under the microscope—to map S1PR1 in the brain. In healthy control brains and in non-cerebral malaria, S1PR1 staining was weak in neurons and blood vessels. In contrast, brains from cerebral malaria patients showed intense S1PR1 staining in the vessel wall cells and in neurons, while neighboring support cells called glia showed little to no signal. When the scientists quantified this, S1PR1 “scores” in vessels and neurons were dramatically higher in cerebral malaria than in the other groups. The more parasite-filled red cells were stuck in the vessels, the higher the S1PR1 expression and the lower the S1P level in the blood, revealing a tight link between parasite burden, receptor activation, and loss of the protective signal. 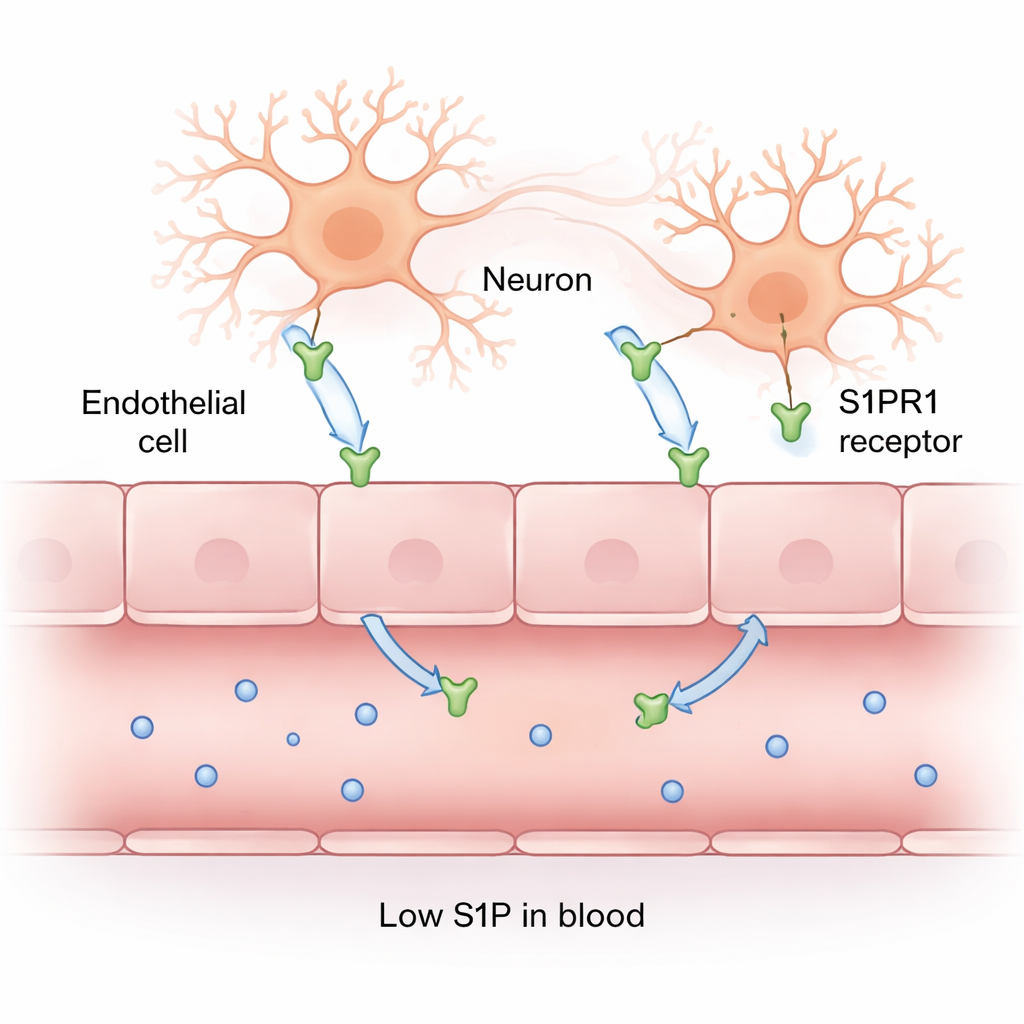
Piecing together a vicious cycle
The pattern points to a possible vicious cycle. As infected cells clog brain vessels, oxygen levels fall and inflammation flares. Blood levels of S1P drop, likely because damaged red cells, platelets, and vessel cells can no longer maintain normal supplies. At the same time, vessel cells and neurons crank up S1PR1 expression, perhaps trying to grab what little S1P is left and restore barrier function. But this heightened receptor activity may further draw S1P out of circulation and may even alter inflammatory signaling inside the brain. The result is worsening vessel leakiness, more bleeding into brain tissue, and deepening neurological damage.
What this could mean for future treatments
To a non-specialist, the key message is that the S1P–S1PR1 system acts like a stabilizing “glue” for brain blood vessels, and this glue appears to fail in cerebral malaria. Patients who die from this condition show very low levels of S1P in their blood and very high levels of its receptor on brain vessels and neurons. This study does not yet prove cause and effect, but it strengthens the idea that drugs which boost S1P signaling or carefully modulate S1PR1—some of which already exist for other diseases—might one day be used alongside antimalarial drugs to keep the brain’s blood vessels intact and reduce deaths from this devastating illness.
Citation: Srisook, C., Nintasen, R., Punsawad, C. et al. Expression of sphingosine-1-phosphate receptor 1 in the brain of fatal cerebral malaria. Sci Rep 16, 5641 (2026). https://doi.org/10.1038/s41598-026-36072-6
Keywords: cerebral malaria, blood-brain barrier, sphingosine-1-phosphate, endothelial cells, brain inflammation