Clear Sky Science · en
Graphene-enhanced non-Hermitian Thue–Morse metamaterial sensor exploiting exceptional point for cancer biomarker detection
Why a Better Cancer Sensor Matters
Finding cancer early can mean the difference between a simple treatment and a life‑threatening disease. Many modern tests rely on labeling blood or tissue with dyes or special molecules, which can be slow, costly, and complicated. This paper describes a new kind of optical sensor – a tiny layered chip that uses light, special materials, and ultra-thin graphene sheets – to detect subtle changes linked to cancer cells and biomarkers without adding any labels. The result is a compact device that could one day help doctors spot cancer earlier and more reliably.
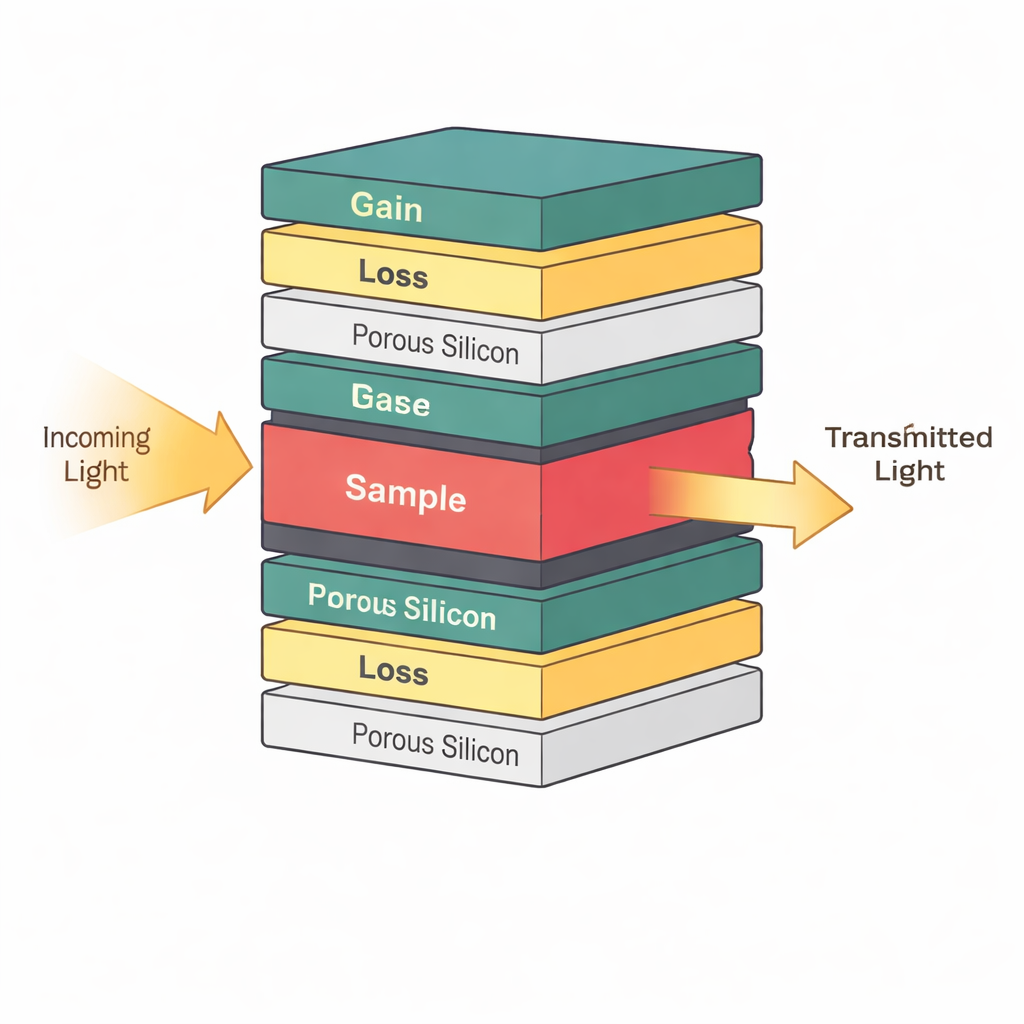
Stacking Light‑Guiding Layers Like a Puzzle
At the heart of the device is a carefully designed stack of ultra-thin layers that guide and trap light. Instead of arranging these layers in a perfectly repeating pattern, the authors use a mathematical recipe called the Thue–Morse sequence, which sits between order and disorder. This quasi‑pattern creates special “sweet spots” where light becomes strongly confined in narrow regions of the stack. In the middle of this structure, they insert a layer that holds the actual sample – for example, a fluid containing healthy cells or cancer cells. Because the light is concentrated around this central layer, even small changes in the sample’s optical properties, such as its refractive index, can noticeably shift how the device transmits light.
Using Gain, Loss, and Exceptional Points to Boost Signals
The sensor also takes advantage of a powerful idea from modern photonics called parity–time symmetry. In simple terms, some layers in the stack slightly amplify light while others slightly absorb it, arranged in a way that balances gain and loss around the center. When this balance is tuned just right, the system reaches what physicists call an exceptional point, where two light modes merge into one. Near this point, the device becomes extraordinarily sensitive: a tiny disturbance in the sample – such as a small change in cancer cell concentration – causes a disproportionately large shift in the transmitted light signal. The authors show that operating close to this special condition makes the resonance peak in the spectrum much sharper, which directly improves how finely the sensor can distinguish different tissues or biomarker levels.
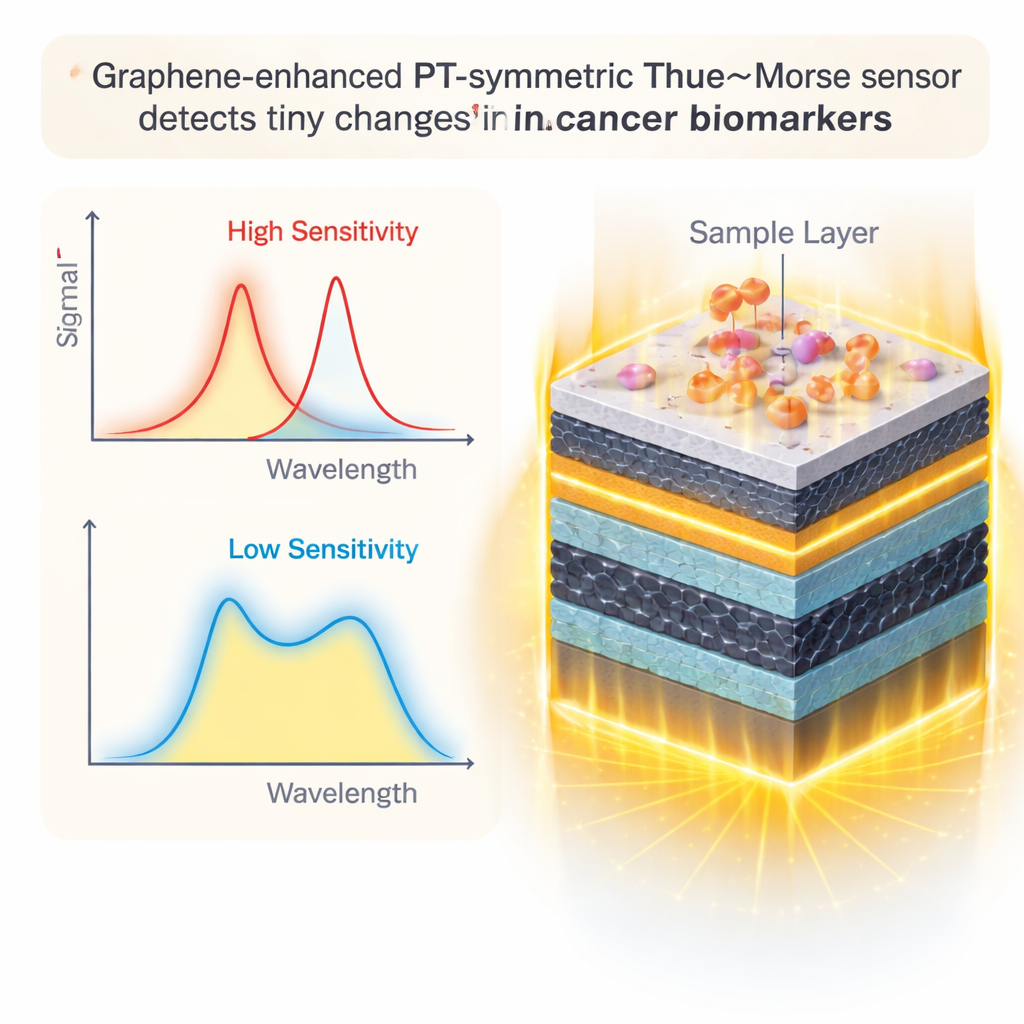
Graphene Layers as a Light‑Anchoring Skin
To enhance performance further, the researchers add sheets of graphene – single‑atom‑thick carbon layers – at key interfaces around the sample. Graphene is famous not only for its strength and conductivity but also for how it interacts with light. By adjusting its electrical properties, the team can make graphene pull light even closer to the sample region and reduce unwanted losses. Careful simulations reveal that when the graphene’s key tuning knobs – its chemical potential and relaxation time – are set to specific values, the resonance becomes narrower and more responsive. Adding up to four graphene layers around the sample provides the best trade‑off: the signal becomes stronger and more precise without being overly damped by extra absorption.
Balancing Design Details and Real‑World Fabrication
The device also uses porous silicon layers, riddled with tiny holes, to host biological material and increase the surface area where cells and biomarkers can bind. The authors systematically vary practical design parameters such as layer thickness, porosity, and angle of the incoming light, and they check how small fabrication errors might impact performance. They find that the sensor remains stable when these parameters vary by about two percent, a range that current nanofabrication techniques can realistically achieve. As the number of graphene layers increases, sensitivity generally improves, but too many layers eventually introduce excessive loss. The study identifies a sweet spot in configuration and operating conditions that could guide future experimental prototypes.
What This Could Mean for Future Diagnostics
In clear numerical terms, the proposed sensor can shift its optical signal by more than 1000 nanometers for a unit change in the sample’s refractive index, with a detection limit fine enough to sense very small differences between healthy and cancerous cells. While some specialized fiber‑based systems can reach even lower limits, they are often bulky or hard to integrate. By contrast, this design is compact, silicon‑compatible, and tailored for on‑chip integration with microfluidic channels and biochemical coatings that target specific cancer markers. In plain language, the work shows how combining an unusual layer pattern, balanced gain and loss, and graphene “skins” around the sensing region can turn a small optical chip into a highly responsive, label‑free cancer detector – a promising step toward faster, more accessible diagnostic tools.
Citation: Mohammadpour, A., Vala, A.S. & Barvestani, J. Graphene-enhanced non-Hermitian Thue–Morse metamaterial sensor exploiting exceptional point for cancer biomarker detection. Sci Rep 16, 6521 (2026). https://doi.org/10.1038/s41598-026-36067-3
Keywords: cancer biosensor, graphene photonics, optical sensor, photonic crystal, biomarker detection