Clear Sky Science · en
ACE2 expression by colonic epithelial cells is associated with viral infection, immunity, and energy metabolism
Why the Gut Matters in a Respiratory Virus
When we think of COVID-19, we picture lungs filling with fluid and patients struggling to breathe. Yet many people with SARS-CoV-2 infection also develop stomach pain, diarrhea, and long‑lasting gut problems. This study explores why the intestines—especially the colon—may be an important target for the virus. By zooming in on individual cells from children’s colons, the researchers show that a key viral doorway, the ACE2 protein, is highly active in specific intestinal cells and is tied not only to viral infection but also to our immune defenses and the way cells manage energy.
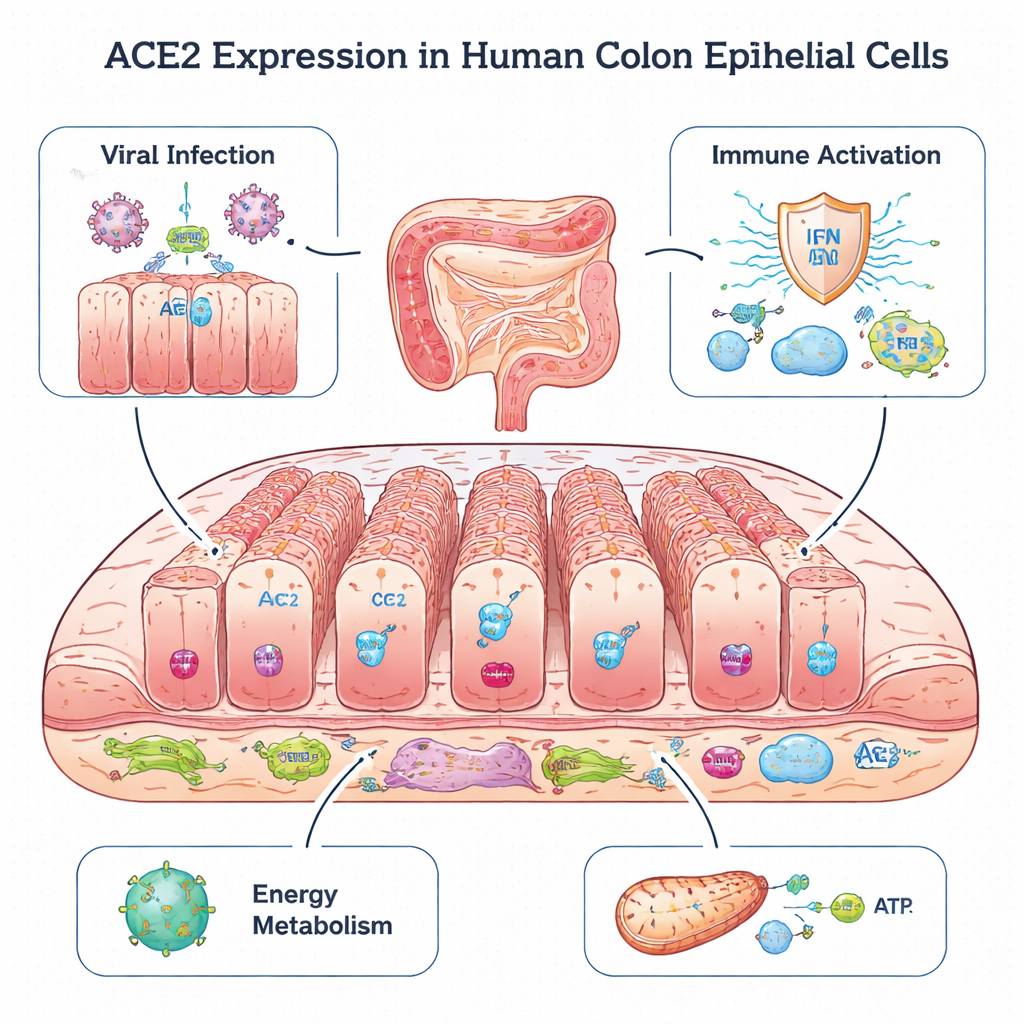
A Cellular Doorway in the Colon
ACE2 is best known as the main entry point that both the original SARS virus and SARS‑CoV‑2 use to infect cells in the lung. Paradoxically, animal studies suggest that ACE2 can also protect tissues from damage by helping keep a hormone system that controls blood pressure in balance. In the gut, ACE2 is already known to support a healthy microbiome and prevent inflammation. In this work, scientists re‑analyzed single‑cell RNA sequencing data from colon biopsies of 17 children: some had healthy intestines, while others had colitis or inflammatory bowel disease, including ulcerative colitis and Crohn’s disease. This technique allowed them to see which genes are active in thousands of individual cells, and to pinpoint exactly where ACE2 is turned on.
Colonocytes: Prime Targets and First Responders
The analysis revealed that ACE2 is concentrated almost exclusively in colonocytes, a major type of epithelial cell that lines the inside of the colon and faces the gut contents. Compared with other epithelial cells, colonocytes also showed higher activity of genes that help viruses enter cells, bud off, and exit. At the same time, these cells expressed many genes involved in sensing viral genetic material and triggering type I and type III interferon responses—key early alarm systems of the immune defense. They also produced inflammatory signals and molecules that present viral fragments to T cells, suggesting that colonocytes are both potential viral targets and important early sentinels that alert the immune system.
Linked Networks of Immunity and Energy Use
The researchers then asked which other genes tend to be turned on or off in lockstep with ACE2 in colonocytes. They found over 3,400 genes that rose with ACE2 levels and more than 2,100 that fell as ACE2 increased. Genes that tracked positively with ACE2 were enriched in processes related to viral entry and release, innate immune signaling, the killing activity of natural killer cells and T cells, and several energy‑producing pathways inside cells, including mitochondrial function and the processing of fats and sugars. In contrast, genes that moved in the opposite direction—high when ACE2 was low—were tied to antibody‑based (humoral) immunity, the cell’s protein‑building machinery, cell‑eating processes such as phagocytosis, and the complement system, another arm of immune defense. This pattern suggests that ACE2 marks a state in which colonocytes are tuned toward rapid antiviral sensing and energy production, while other, slower immune programs are dialed down.
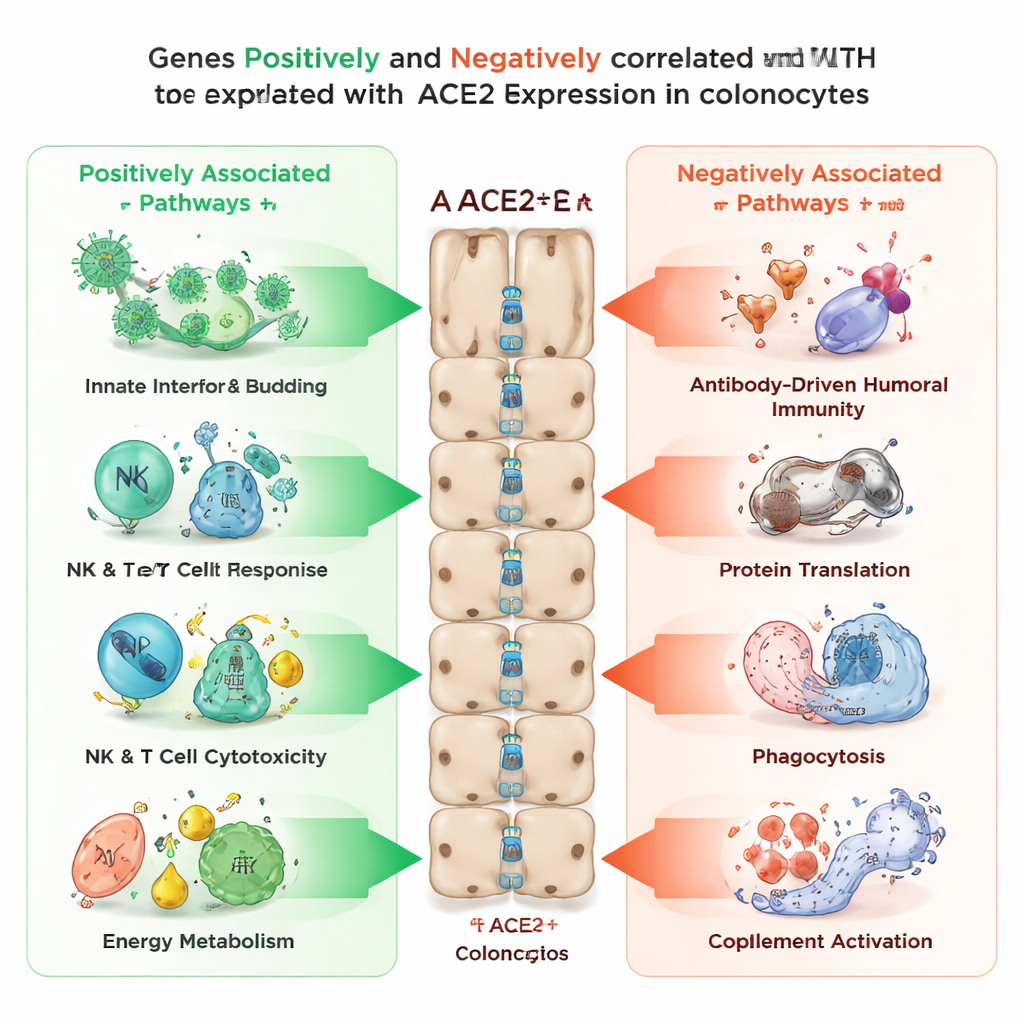
Evidence from Inflamed Intestines
To check that these patterns occur in actual tissues, the team used multiplex immunofluorescence—a technique that lights up specific proteins with different colors—on colon samples from children with ulcerative colitis, Crohn’s disease, and from controls. They confirmed that ACE2 protein sits mainly on epithelial cells marked by EPCAM. In patients with inflammatory bowel disease, ACE2‑positive cells showed higher levels of IFNA4 and RSAD2, two proteins strongly associated with interferon signaling and antiviral activity. This indicates that in the inflamed gut, ACE2‑bearing colon cells are not just potential gateways for viruses; they are also hotspots of antiviral response.
What This Means for Patients and Future Therapies
For non‑specialists, the message is that the colon is not a passive bystander in COVID‑19. The same protein that lets the virus into cells also helps coordinate gut immunity and energy use, especially in children with pre‑existing intestinal inflammation. This dual role may help explain why some patients develop gastrointestinal symptoms and why people with inflammatory bowel disease have complex, but not uniformly worse, outcomes during COVID‑19. The work also feeds into broader discussions about drugs that act on blood‑clotting and blood‑pressure pathways, some of which may lessen lung and vascular damage in severe infection. While the study is based on correlations and cannot prove cause and effect, it highlights ACE2‑rich colon cells as important players at the crossroads of viral entry, immune defense, and metabolism—pointing to the gut as a key, and potentially treatable, arena in coronavirus disease.
Citation: Qi, Y., Huang, Y., Chen, H. et al. ACE2 expression by colonic epithelial cells is associated with viral infection, immunity, and energy metabolism. Sci Rep 16, 5738 (2026). https://doi.org/10.1038/s41598-026-36052-w
Keywords: ACE2, colonocytes, COVID-19 gut, inflammatory bowel disease, antiviral immunity