Clear Sky Science · en
Proteomic analysis of endothelial progenitor cells secretome identifies Serpine 1 as a potent regulator of osteogenesis
Healing Broken Bone Without Donor Grafts
When a bone is badly broken or a piece is missing, surgeons often struggle to restore both strength and shape. Today’s options—taking bone from another part of the body or using synthetic grafts—can be painful, risky, and not always successful. This study explores a different idea: instead of transplanting cells, can we deliver just the right natural proteins to switch on the body’s own bone-building machinery?
Why Some Bone Injuries Refuse to Heal
Most fractures mend on their own, but large gaps in bone—such as those caused by trauma, tumors, or severe jawbone shrinkage—often do not. In these cases, the body simply does not have enough local blood vessel cells and bone-forming stem cells to rebuild the missing segment. Researchers in tissue engineering try to solve this by combining three ingredients: living cells that can form new bone, a scaffold that gives them something to grow on, and signals that tell them what to do. Cell transplants can work, but they are expensive, highly regulated, and not always practical for everyday clinical use. That has pushed scientists to search for “cell-free” solutions built around smart biomaterials and precisely chosen proteins.
Secret Signals from Vessel-Building Cells
Endothelial progenitor cells are rare cells in the blood that help form new blood vessels. Earlier work showed that when these cells are implanted near bone, they encourage healing—largely by sending out soluble signals rather than by turning into bone themselves. In the new study, the authors collected the liquid culture medium in which these cells had been growing and used advanced protein analysis to catalog hundreds of secreted molecules. They focused on eight proteins strongly linked to blood vessel growth and bone formation. Using human bone marrow stem cells and human microvascular endothelial cells in the lab, they systematically tested which of these proteins best boosted cell growth, directed migration, and triggered bone-like mineral deposition.
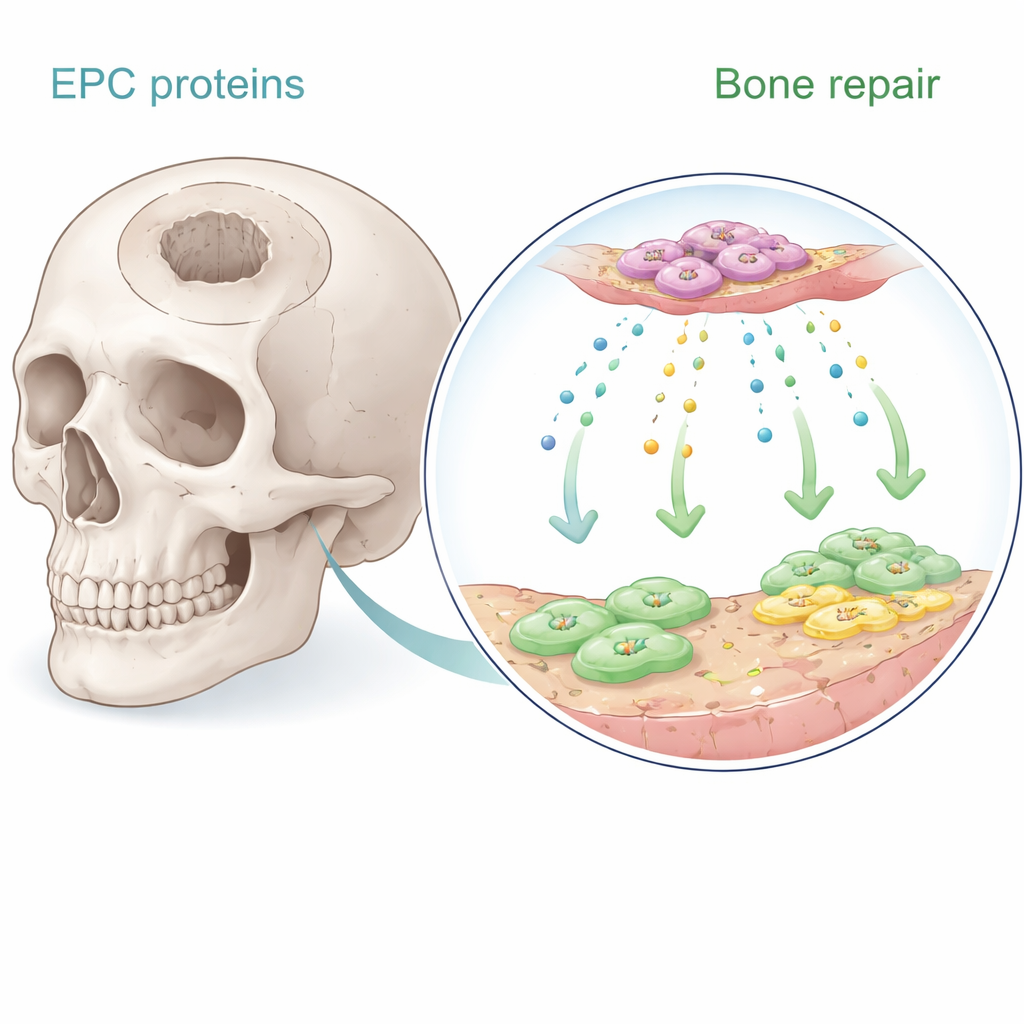
A Surprising Protein Takes the Lead
Among the candidates, one protein stood out: Serpine-1, better known for its role in blood clot regulation and wound healing. In culture dishes, Serpine-1 significantly increased the proliferation of both bone marrow stem cells and endothelial cells in a dose-dependent manner, performing better than well-known growth factors such as BMP-2 and SDF-1 in this respect. It also promoted the maturation of stem cells into bone-forming cells, as shown by classic staining methods that reveal alkaline phosphatase activity and calcium-rich mineral deposits. Other proteins, like platelet-derived growth factor and BMP-2, were more effective at encouraging cells to crawl into a “wound” area in migration assays, but Serpine-1 offered a rare combination of boosting both cell number and bone-building behavior.
Turning a Protein into a Practical Implant
Finding a promising protein is only half the challenge; it must also be delivered to an injury site in a controlled way. The team encapsulated Serpine-1 inside tiny biodegradable spheres made of a medical polymer called PLGA, then mixed these microspheres into a soft collagen gel. This created a scaffold that slowly released the protein over time. They tested it in mice by drilling a 4-millimeter circular hole in the skull—a defect size that does not heal on its own. Some defects were filled with collagen alone, some with collagen plus empty microspheres, and some with the Serpine-1–loaded scaffold. After eight weeks, high-resolution micro-CT scans showed that the Serpine-1 group had significantly higher bone volume, density, and thickness inside the defect. Remarkably, only these animals developed new bone in the center of the gap, rather than just along the edges.
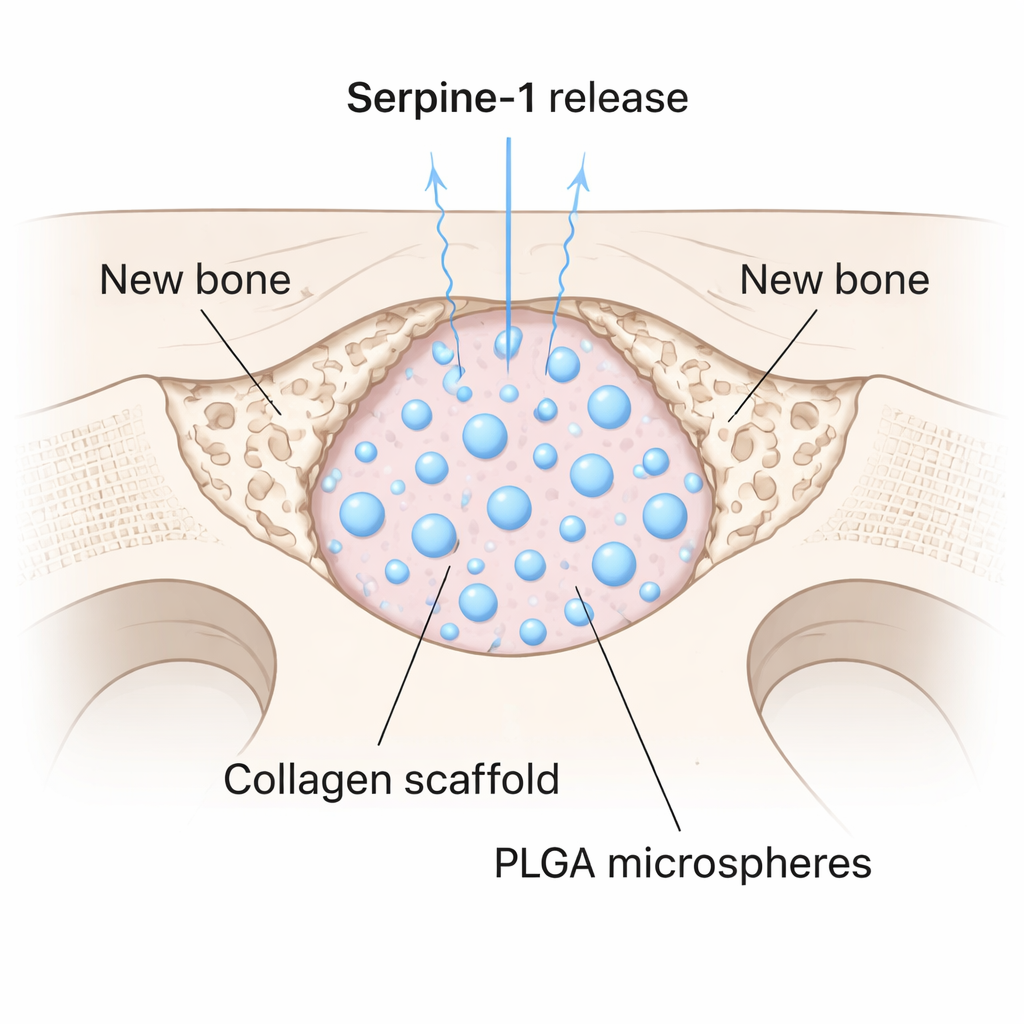
What This Could Mean for Future Bone Care
The study introduces Serpine-1 as a powerful and previously underappreciated signal for bone formation. By pairing this protein with a slow-release scaffold, the researchers achieved meaningful bone regrowth in a defect that would normally remain empty. While Serpine-1 did not draw cells into the defect as strongly as some other factors, its ability to help existing cells multiply and mature suggests it could be combined with migration-promoting proteins for even better results. For patients, such cell-free, protein-based materials could one day reduce the need for harvesting bone from their own body or relying on complex cell therapies, offering a simpler way to coax stubborn bone injuries into finally healing.
Citation: Asbi, T., Tamari, T., Doppelt-Flikshtain, O. et al. Proteomic analysis of endothelial progenitor cells secretome identifies Serpine 1 as a potent regulator of osteogenesis. Sci Rep 16, 5165 (2026). https://doi.org/10.1038/s41598-026-36048-6
Keywords: bone regeneration, Serpine-1, tissue engineering, endothelial progenitor cells, collagen scaffold