Clear Sky Science · en
NiCd/ZnO nanocomposites: novel materials for photocatalytic degradation of Allura Red dye
Why cleaning colored water matters
Bright synthetic dyes make our foods and products look appealing, but once they wash down the drain they can linger in rivers and lakes for years. One such dye, Allura Red, is widely used in drinks, candies, and processed foods and has raised health concerns in recent studies. This paper explores a new type of light-activated material that can break this stubborn red dye down into harmless substances, pointing toward cleaner and safer wastewater treatment.

A stubborn red dye in everyday life
Allura Red is designed to be tough: it resists fading, microbes do not easily eat it, and it can travel long distances in water without breaking apart. That durability is a problem when the dye escapes from factories or sewage systems into natural waterways. Traditional treatment methods—such as filtering, settling, or using chemicals—often only move the dye around or turn it into other waste, instead of fully destroying it. Advanced approaches that rely on powerful oxidizing molecules can do better, but they need efficient materials to trigger these reactions in a practical, low-cost way.
Using light and tiny particles to destroy dye
The authors focus on zinc oxide, a common white powder already used in sunscreens and paints, because it can act as a photocatalyst: under ultraviolet light it creates short-lived reactive forms of oxygen that attack organic molecules. Pure zinc oxide, however, mainly absorbs ultraviolet light and tends to let its excited charges recombine quickly, wasting energy. To overcome this, the team modified zinc oxide by adding tiny amounts of cadmium and nickel, producing three versions: plain ZnO, a cadmium–zinc composite (CdZnO), and a nickel–cadmium–zinc composite (NiCdZnO). Although all three keep the same basic crystal structure, the added metals subtly stretch or squeeze the atomic lattice, change how the particles grow, and increase the surface area available to contact dye molecules.
How co-doping makes light work harder
Detailed measurements showed that adding cadmium and nickel shifts the material’s light absorption from the ultraviolet toward the visible range and narrows the energy gap that electrons must cross when light hits. The particles also become smaller and more porous, offering more sites where dye and oxygen can land. Light-emission tests revealed that the modified particles lose less energy to unwanted recombination of charges: electrons and holes live long enough to react with water and oxygen, producing aggressive species such as hydroxyl radicals and superoxide. These species then attack the complex rings in the Allura Red molecule, cutting them apart step by step until only carbon dioxide, water, and simple salts remain, as confirmed by chemical oxygen demand measurements.
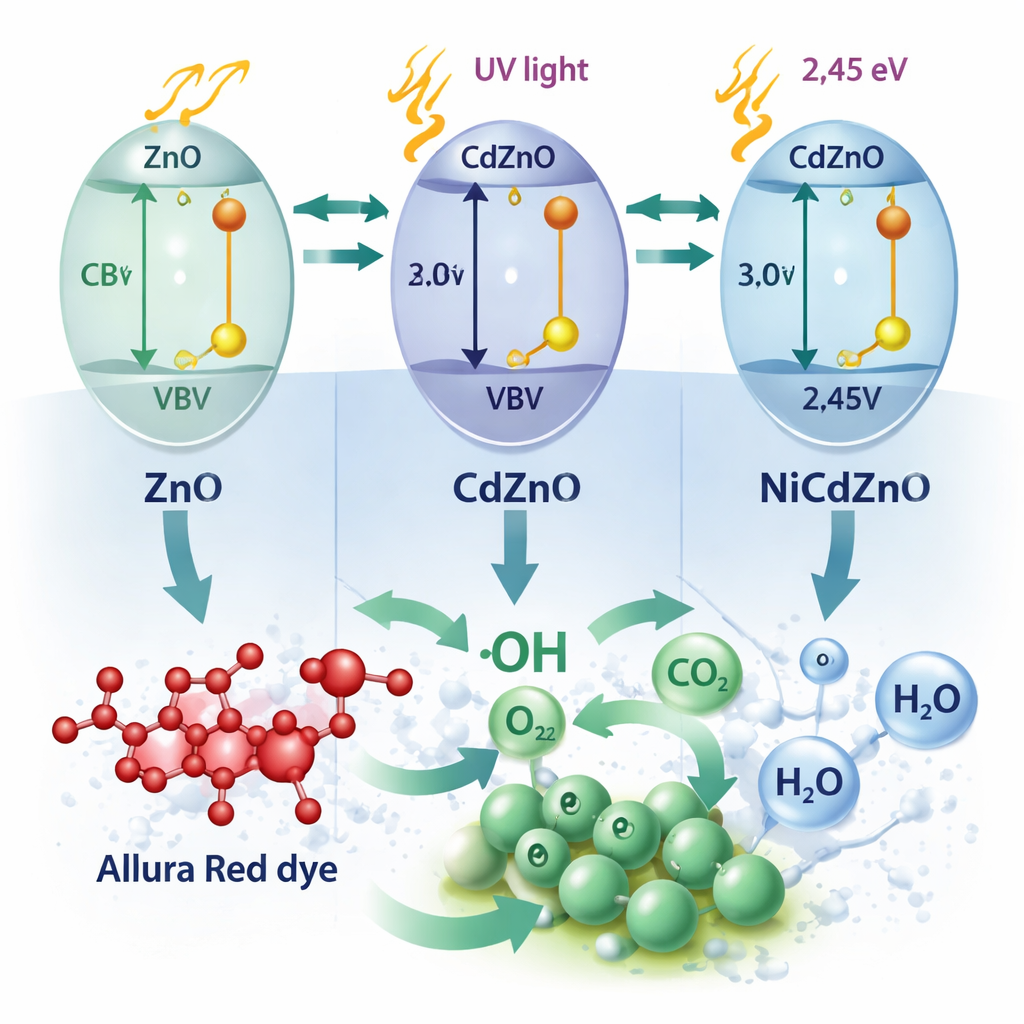
Putting the new materials to the test
When the researchers illuminated dye solutions containing each material, the differences were striking. Under the same UV–visible lamp and at the same catalyst loading, plain zinc oxide removed about half of the dye in 50 minutes. The cadmium–zinc composite reached around 80 percent removal, while the nickel–cadmium–zinc composite eliminated roughly 95–98 percent of the color in that time and showed the fastest reaction rate in kinetic analyses. The co-doped material worked well across a range of dye concentrations and pH values, performed best in mildly alkaline water, and retained most of its activity over several reuse cycles. Experiments that selectively blocked different reactive species showed that holes and hydroxyl radicals were the main agents of destruction, with superoxide playing a supporting role.
What this could mean for cleaner water
For non-specialists, the key message is that very small changes at the atomic level—swapping in traces of cadmium and nickel into zinc oxide—can dramatically boost how efficiently light energy is used to clean contaminated water. The optimized nickel–cadmium–zinc nanoparticles soak up more of the light we already have, keep their charges apart long enough to do useful chemistry, and offer plenty of surface area for dye molecules to land on. While questions of long-term cost, safety, and large-scale deployment remain, this study shows a promising route toward compact, reusable materials that can strip intense food dyes like Allura Red from wastewater before they reach our taps and ecosystems.
Citation: Khan, S., Sadiq, M., Muhammad, N. et al. NiCd/ZnO nanocomposites: novel materials for photocatalytic degradation of Allura Red dye. Sci Rep 16, 5204 (2026). https://doi.org/10.1038/s41598-026-36010-6
Keywords: photocatalysis, wastewater treatment, zinc oxide nanoparticles, Allura Red dye, advanced oxidation processes