Clear Sky Science · en
Molecular crowding effects on protein stability in a bacterial proteome
Why the crowded life of proteins matters
Inside every living cell, proteins do their jobs in a space so packed with other molecules that nearly half the volume can be taken up. Yet most lab experiments study proteins in dilute, almost empty solutions. This paper asks a simple but important question: how does this crowded reality change the stability and behaviour of proteins, and what does that mean for how life works at the molecular scale?
Peeking into a packed cellular world
To explore this, the researchers worked with a bacterium called Cupriavidus necator, a microbe of interest for green technologies such as carbon capture and bioplastic production. They broke open the cells gently, keeping hundreds of different proteins close to their natural forms. Then they added high concentrations of large, water‑loving polymers—common “crowding agents” called PEG, dextran, and Ficoll—that are often used to mimic the dense interior of cells.
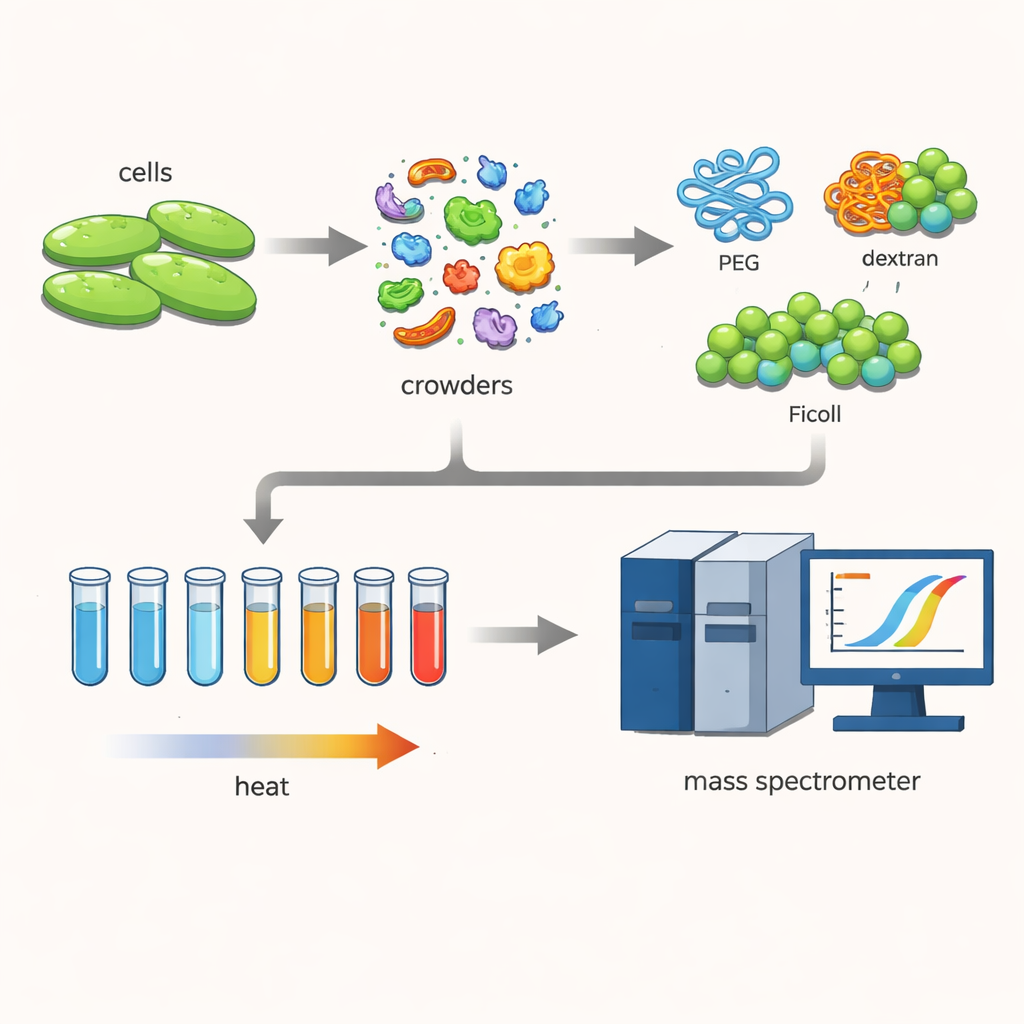
Watching proteins melt
The team used a technique known as thermal proteome profiling. They heated many tiny samples of the protein mixture stepwise from 30 to 70 degrees Celsius. As the temperature rose, less stable proteins unfolded and clumped together, dropping out of solution. By tagging the remaining soluble proteins and measuring them with a sensitive mass spectrometer, the scientists could reconstruct melting curves for each protein and determine its melting temperature—the point where half of it has left the soluble, working state. Comparing these temperatures with and without crowding agents revealed how the crowded environment shifted protein stability.
A mixed picture: some proteins steadier, others shakier
On average, adding any of the six polymer crowders lowered the melting temperatures across the bacterial proteome, implying a slight overall destabilizing effect. But this global trend hid a much more nuanced story. For dozens of individual proteins, the crowding agents either clearly raised or lowered stability, sometimes by several degrees. Most proteins were affected by only one of the six reagents, but a handful responded to several, and almost all of these were either consistently stabilized or consistently destabilized, hinting at shared underlying features in how they interact with crowders.
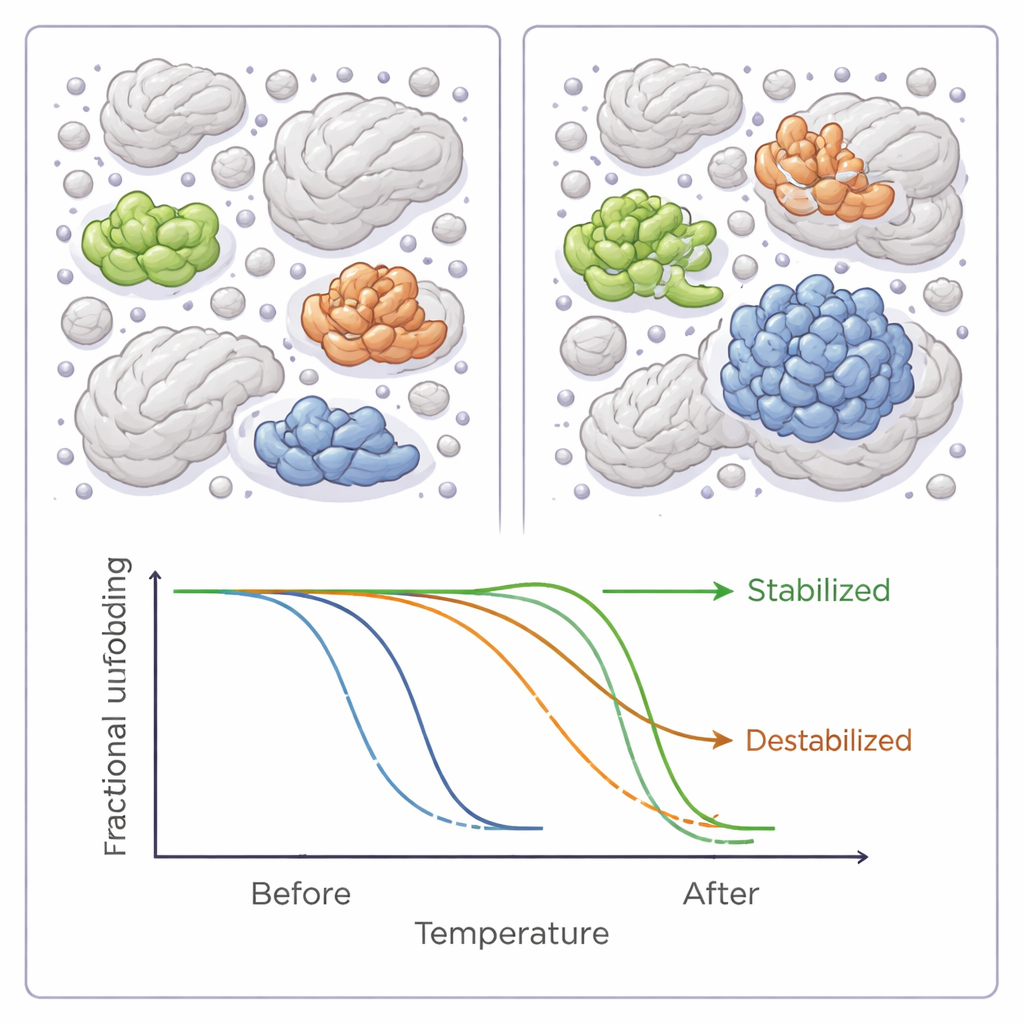
What makes a protein win or lose in a crowd
When the authors looked more closely at the properties of these sensitive proteins, patterns emerged. Proteins that became more stable in crowded conditions tended to be more hydrophobic—that is, they have more water‑repelling surfaces—and were more often classic enzymes with well‑defined active sites. They were also more likely to be involved in protein‑protein interactions or carry chemical modifications that tune their function. Destabilized proteins, in contrast, were on average smaller and less decorated with such modifications. These findings echo computer models and other experiments suggesting that crowding can favour some proteins while making others more vulnerable, depending on size, shape, and how they normally interact with partners.
Beyond simple squeezing: how crowders really work
Traditional explanations for molecular crowding focus on “excluded volume,” the idea that big molecules simply leave less room, nudging proteins to stay folded. If this were the whole story, one would expect crowding agents mainly to stabilize proteins and see strong links to how bulky or viscous the crowders are. Instead, the authors saw only weak connections to size and thickness of the polymers. Their data fit better with a “preferential exclusion” picture: crowders and proteins avoid each other chemically, which indirectly favours certain folded states but can also disturb delicate interactions that keep other proteins stable. In short, it is not just physical squeezing, but subtle chemical push‑and‑pull that shapes protein behaviour.
What this means for understanding life’s chemistry
For non‑specialists, the key message is that proteins inside cells cannot be fully understood in isolation, swimming in clear buffer. The dense molecular jungle around them can make some proteins sturdier and others more fragile, changing how entire networks of reactions behave. This study, by measuring stability changes for hundreds of proteins at once, shows that crowding effects are complex and protein‑specific, and likely driven by direct interactions as much as by simple packing. As researchers design drugs, industrial enzymes, or engineered microbes, recognizing this crowded reality will be crucial to predicting how proteins truly behave in living systems.
Citation: McKeever, K., Dillon, E.T., Wynne, K. et al. Molecular crowding effects on protein stability in a bacterial proteome. Sci Rep 16, 5908 (2026). https://doi.org/10.1038/s41598-026-35990-9
Keywords: molecular crowding, protein stability, thermal proteome profiling, bacterial proteome, cellular environment