Clear Sky Science · en
Visible-light photocatalytic mineralization of 4-Chlorophenol over ZnO-loaded sulfonated carbonaceous bentonite: kinetic analysis, pathway elucidation, and catalyst reusability
Why Cleaning Toxic Water Matters
Many factories release stubborn chemicals into water that are difficult to remove and dangerous even at tiny amounts. One such chemical, 4‑chlorophenol, is linked to cancer and can build up in living things. This study explores a low‑cost, sunlight‑driven way to destroy this pollutant completely, not just hide it, by using a special mixture of natural clay and zinc oxide that turns dirty water into safe, mineralized water.
Turning Common Clay into a Smart Cleaner
The researchers started with bentonite, a cheap, widely available clay already used in environmental cleanup. This clay naturally has stacked layers and many tiny passages that can trap chemicals. They first treated the clay with strong sulfuric acid to create “sulfonated carbonaceous bentonite,” which adds acidic groups and makes the surface more welcoming to pollutants like 4‑chlorophenol. Then they carefully grew zinc oxide nanoparticles on this modified clay, producing a hybrid material they call ZnO@SB. Tests using X‑ray diffraction, electron microscopy, and infrared spectroscopy showed that the clay structure was partly opened up, the acid groups were successfully added, and zinc oxide crystals were evenly spread over the surface at the nanoscale.
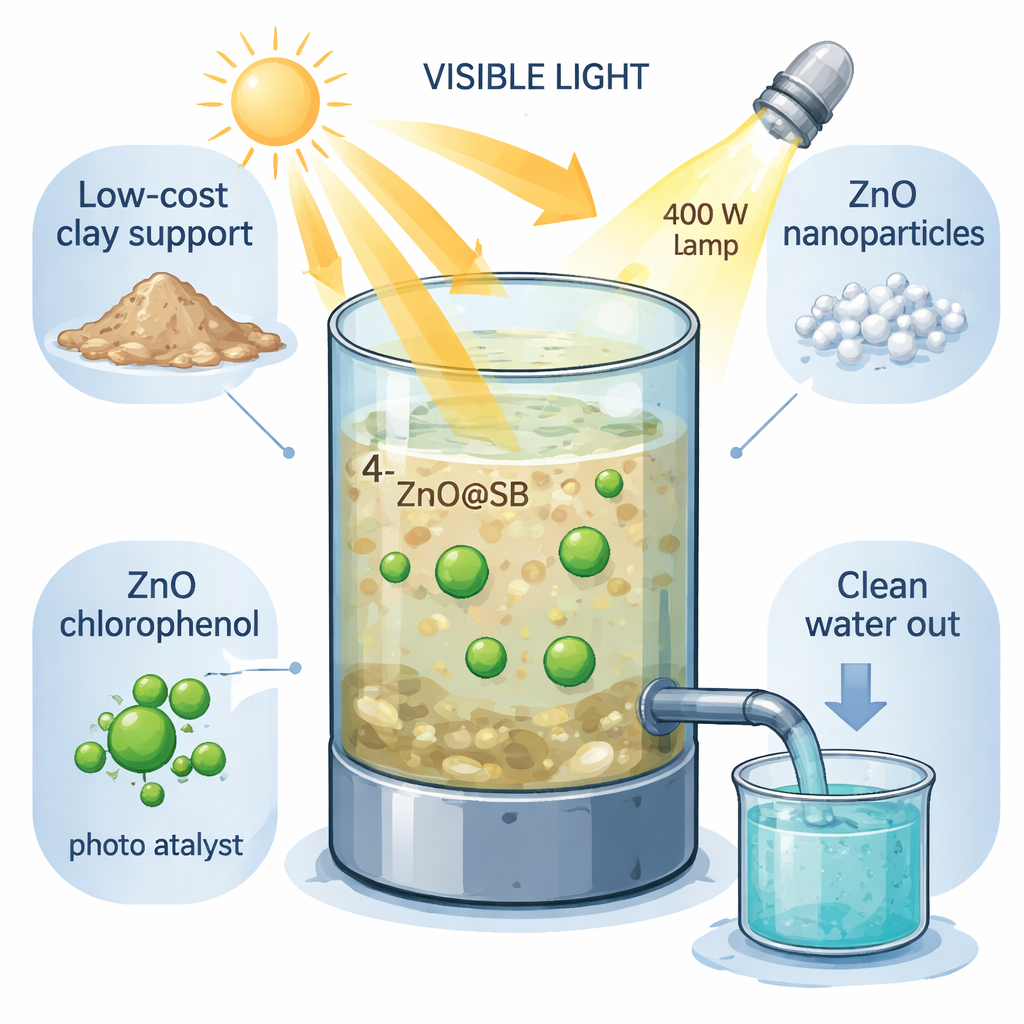
How Light Helps Destroy a Tough Chemical
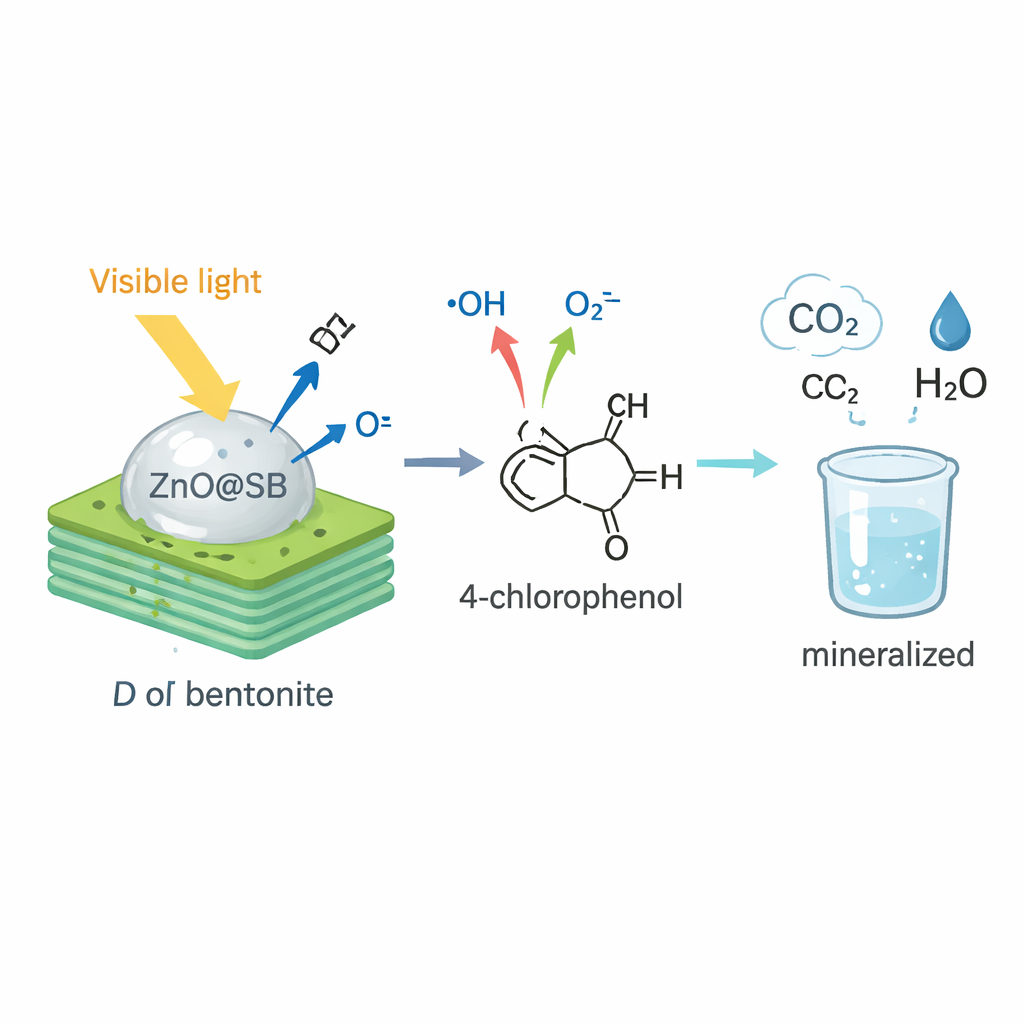
ZnO@SB is designed to use visible light—the same kind of light we get from the sun—to trigger powerful reactions on its surface. When the material is lit, the zinc oxide absorbs light and generates energetic electrons and “holes” that react with water and oxygen to form extremely reactive species called radicals. Two radicals in particular, hydroxyl (•OH) and superoxide (O₂•⁻), attack 4‑chlorophenol molecules that have already been drawn onto the clay’s surface. Step by step, these radicals add oxygen, remove chlorine, split open the ring‑shaped part of the molecule, and finally break it down into simple, harmless products such as carbon dioxide, water, and chloride ions.
Fast and Complete Cleanup in the Lab
In a glass reactor lit by a visible‑light metal‑halide lamp, the team tested how well ZnO@SB could clean water containing 4‑chlorophenol. At a modest pollutant level (5 milligrams per liter) and slightly basic pH 8, a small amount of catalyst (0.5 grams per liter) removed all detectable 4‑chlorophenol in just 30 minutes. Importantly, measurements of total organic carbon showed that all of the organic material was fully converted to carbon dioxide and water in 60 minutes—evidence of complete mineralization rather than partial breakdown. The reaction followed simple first‑order behavior, meaning the speed of cleanup was directly tied to how much pollutant remained. When they used more catalyst, the process became more efficient, and the number of pollutant molecules destroyed per photon of light, known as the quantum yield, increased roughly fourfold.
Built to Be Reused, Not Thrown Away
For a water‑treatment material to be practical, it must work over and over without falling apart or leaking metals back into the water. The ZnO@SB hybrid passed this test well. After five cleaning cycles, it still removed more than 90 percent of the pollutant, showing only a small drop in performance. Measurements of dissolved zinc in the treated water stayed far below international drinking‑water limits, and the material’s infrared “fingerprint” changed very little, meaning the structure remained intact. Because the photocatalyst is based on an abundant natural clay and uses visible light under mild conditions, the authors argue it is both cost‑effective and safer for workers than many high‑temperature or chemical‑intensive methods.
What This Means for Real‑World Water Treatment
To a non‑specialist, the key message is that ZnO@SB acts like a solar‑powered sponge and shredder in one: the clay part grabs a toxic molecule, and the zinc oxide part, activated by light, chops it into harmless pieces. In controlled tests it completely destroyed a priority pollutant faster than many existing systems, while staying stable and releasing almost no metal. Although more work is needed in real, complex wastewaters and at larger scales, this study points toward affordable, reusable, light‑driven materials that could help communities and industries turn hazardous wastewater into safer effluent with far less chemical input and energy demand.
Citation: Ahmed, Z., Allam, A., El-Sayed, M. et al. Visible-light photocatalytic mineralization of 4-Chlorophenol over ZnO-loaded sulfonated carbonaceous bentonite: kinetic analysis, pathway elucidation, and catalyst reusability. Sci Rep 16, 5319 (2026). https://doi.org/10.1038/s41598-026-35956-x
Keywords: photocatalysis, wastewater treatment, zinc oxide, bentonite clay, chlorophenols