Clear Sky Science · en
Evaluation of ventilation at 10 °C as the optimal storage condition for donor lungs in a murine model
Keeping Donor Lungs Alive Longer
Lung transplants can be lifesaving, but there is a race against the clock once a donor lung is removed from the body. Today, most lungs are packed on ice and must be transplanted within a few hours or they become too damaged to use. This study in mice explores a surprisingly simple idea: instead of letting stored lungs sit motionless in the cold, what if we gently keep them breathing at a slightly warmer temperature? The answer could help more donated lungs reach patients in better shape.
Why Storing Lungs Is So Hard
Currently, donor lungs are usually flushed with a preservation solution, inflated once, clamped shut, and placed on ice at about 4 °C. This slows metabolism but also puts the lungs into a kind of deep freeze where they can tolerate only 6–8 hours before damage builds up. More advanced systems called ex vivo lung perfusion can keep lungs warm and supplied with blood and air, but they require expensive machines, specialized teams, and are difficult to move between hospitals. Doctors and engineers have been searching for a simpler approach that keeps lungs healthier for longer without this heavy equipment.
A New Twist: Cool, Gently Breathing Lungs
Recent work has suggested that storing lungs at a slightly warmer 10 °C, instead of 4 °C, can protect their cells and mitochondria—the tiny power plants inside cells. Building on this, the researchers asked whether adding gentle ventilation, so the lungs continue to inflate and deflate with room air, could further improve preservation. Using mouse lungs, they compared three conditions over 24 hours of storage: standard cold static storage at 4 °C, static storage at 10 °C, and storage at 10 °C while the lungs were continuously ventilated with small, protective breaths. All lungs were kept in the same preservation solution to isolate the effect of temperature and motion.
Healthier Cells and Calmer Immune Signals
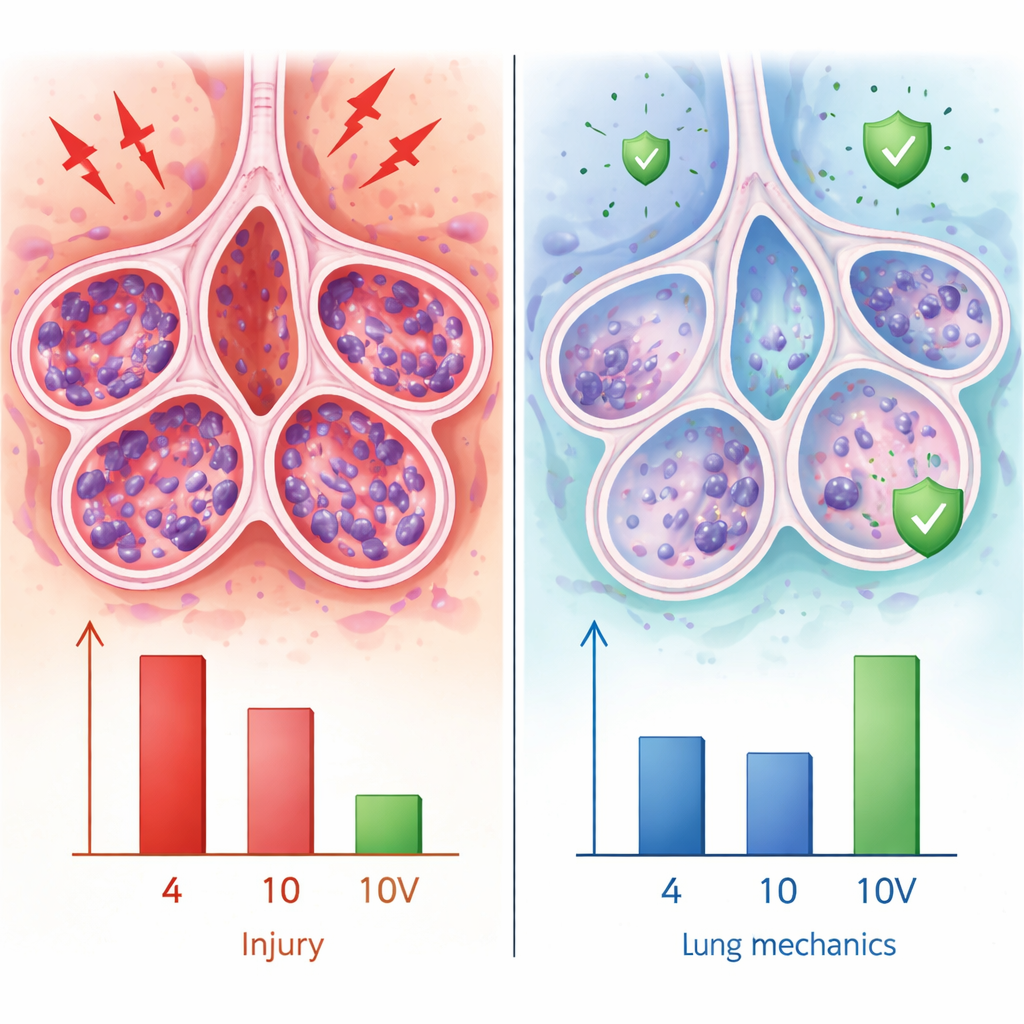
After storage, the team examined the lungs in several ways. Under the microscope, lungs stored with ventilation at 10 °C showed less structural injury—less bleeding, clot-like fibrin, congestion, and fewer invading white blood cells—than lungs kept statically at 4 °C. They also released less complement C3, a blood protein that helps drive inflammation and is linked to early graft failure after transplantation. When the lungs were broken down into individual cells, the ventilated group had fewer cells undergoing programmed cell death (apoptosis) and showed better mitochondrial health, with lower release of cytochrome c and less buildup of damaging reactive oxygen species. Together, these findings suggest that gentle motion and ongoing gas exchange at 10 °C help keep lung cells alive and their internal machinery more stable during storage.
Better Breathing Mechanics After Storage
The ultimate test of a stored lung is how well it functions. Using a specialized ventilator system, the researchers measured how easily air moved through the airways and how stretchy (compliant) the lungs were after 24 hours in each condition. Lungs that had been ventilated at 10 °C had lower airway resistance and higher compliance than lungs stored statically at 10 °C, meaning they opened more easily and required less pressure to fill. Even though not every measure reached statistical significance, the overall pattern favored the ventilated group, pointing to more flexible, less stiff tissue that would likely perform better if transplanted.
What This Could Mean for Future Transplants
This work was done in mice, not humans, and did not include actual transplant surgeries, so it is an early step. Still, the message is clear: keeping donor lungs gently breathing at a modest 10 °C, rather than leaving them motionless on ice, appears to preserve their structure, calm damaging immune signals, and maintain their ability to function. Because this ventilation-only setup is relatively simple and portable compared with full perfusion machines, it could one day be adapted to clinical practice. If confirmed in larger animal and human studies, this approach might extend safe storage times, reduce organ injury, and allow more donor lungs to reach recipients in good condition.
Citation: Hill, M.A., Tennant, M., Watts, B. et al. Evaluation of ventilation at 10 °C as the optimal storage condition for donor lungs in a murine model. Sci Rep 16, 7228 (2026). https://doi.org/10.1038/s41598-026-35943-2
Keywords: lung transplantation, organ preservation, donor lungs, ventilation during storage, cold ischemia