Clear Sky Science · en
APOBEC3B enhances the efficacy of PARP inhibitors in elimination of ovarian cancer stem cell
Why this research matters for ovarian cancer patients
Many women with advanced ovarian cancer initially respond well to modern drugs called PARP inhibitors, which target weaknesses in the tumor’s DNA repair system. Yet over time, the disease often returns as cancer cells learn to survive these treatments. This study explores an unexpected ally inside the cancer cells themselves—a DNA-altering enzyme called APOBEC3B—and shows that, under the right conditions, it can actually make ovarian cancer stem cells easier to eliminate with PARP inhibitors.
A deadly cancer that keeps coming back
High-grade serous ovarian cancer is the most common and most lethal form of ovarian cancer. It is usually diagnosed late, when the disease has already spread in the abdomen, and more than 80% of patients with advanced disease relapse within five years. A key suspect in these relapses is a small population of “cancer stem cells.” These cells can self-renew, survive chemotherapy, and rebuild tumors after treatment. Understanding what makes these stem-like cells resistant—and how to target them—is essential for improving long-term survival.
A mutating enzyme with a double life
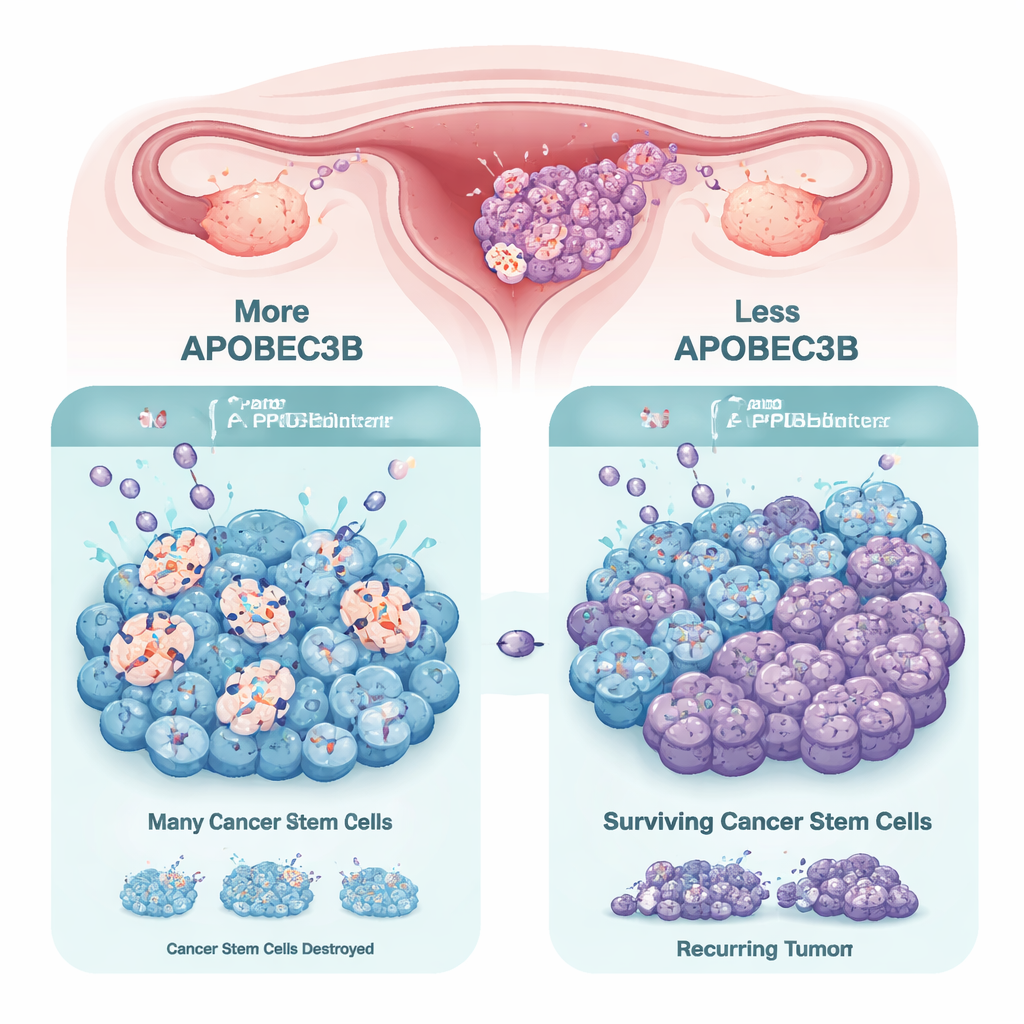
The APOBEC3 family of enzymes normally helps protect our cells from viruses by changing specific DNA letters, which can introduce mutations. In many cancers, one member, APOBEC3B, is overactive and contributes to DNA damage and genetic chaos, which can drive tumor evolution and therapy resistance. In ovarian tumors and patient-derived models, the researchers confirmed that APOBEC3B is usually the most abundant APOBEC3 enzyme. However, when they compared ordinary tumor cells to cancer stem cell–like “tumorspheres” grown in three dimensions, they discovered that the stem-like cells consistently dial down APOBEC3B. This suggests that cancer stem cells may reduce their mutation load to preserve their survival and self-renewal capacity.
Turning down APOBEC3B makes stem cells tougher
To test whether APOBEC3B actually influences stemness, the team used genetic tools to knock down its expression in ovarian cancer models derived from patients and in established cell lines. When APOBEC3B levels were lowered, cancer cells formed tumorspheres more efficiently and showed higher levels of classic stemness markers such as SOX2, OCT4, and NANOG. The fraction of cells with stem cell–like properties increased, indicating that loss of APOBEC3B helps cancer cells shift into a more resilient, stem-like state. In mice, tumors lacking APOBEC3B did not grow faster on their own, but they did behave differently once treated with PARP inhibitors.
More APOBEC3B, more damage—and better drug response
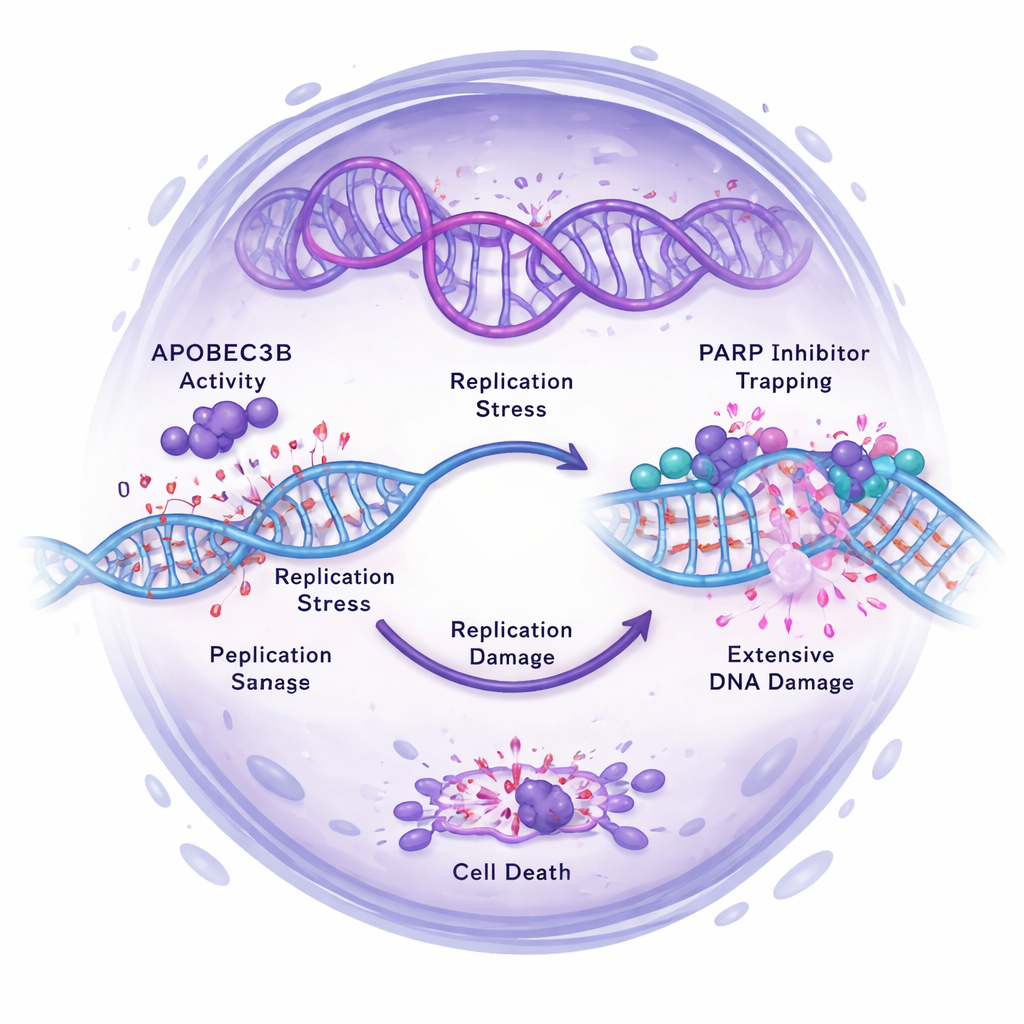
The researchers then asked how APOBEC3B affects the performance of PARP inhibitors such as Olaparib, which work by blocking DNA repair and pushing cancer cells over the edge into lethal DNA damage. In multiple ovarian cancer cell lines, reducing APOBEC3B made cells more resistant to PARP inhibitors, especially in 3D tumorspheres enriched for stem-like cells. Conversely, increasing APOBEC3B sensitized certain cells to PARP inhibitors, greatly lowering their survival. In mouse experiments, tumors with normal APOBEC3B shrank or slowed dramatically with Olaparib, while APOBEC3B-deficient tumors continued to grow despite treatment. Molecular analyses showed why: high APOBEC3B levels heightened DNA replication stress and double-strand breaks, activating DNA damage signaling pathways. When APOBEC3B was knocked down, PARP inhibitor–induced DNA damage, checkpoint activation, and replication stress all decreased, and cells were more likely to stall safely in the cell cycle instead of dying.
A new way to think about resistance and treatment design
Digging deeper with RNA sequencing, the team found that the combination of APOBEC3B loss and PARP inhibition rewired many genes involved in DNA replication and repair, further supporting the idea that APOBEC3B amplifies the harmful effects of PARP inhibitors on cancer cells’ genomes. They also tested whether another repair enzyme, UNG, was required for this effect and found that PARP inhibitor sensitivity in high-APOBEC3B cells was largely independent of UNG. Overall, the data point to APOBEC3B-driven replication stress—not just its mutational activity—as the main partner that helps PARP inhibitors kill ovarian cancer stem cells.
What this means for patients and future therapies
For a layperson, the message is that not all genetic chaos inside a tumor is bad news. In this study, higher levels of the mutating enzyme APOBEC3B actually made ovarian cancer stem cells more vulnerable to PARP inhibitors by overloading their DNA repair capacity. When APOBEC3B was turned down, cancer stem cells became more stem-like and better able to withstand treatment. These findings suggest that measuring APOBEC3B levels could help predict which patients will benefit most from PARP inhibitors, and that timing or combining future APOBEC3B-targeted drugs with PARP inhibitors must be done carefully. Harnessing APOBEC3B’s “self-sabotaging” effect on cancer cells may open new strategies to prevent relapse and more effectively eliminate the roots of ovarian tumors.
Citation: Rivera, M., Liu, L., Enlund, S. et al. APOBEC3B enhances the efficacy of PARP inhibitors in elimination of ovarian cancer stem cell. Sci Rep 16, 5194 (2026). https://doi.org/10.1038/s41598-026-35939-y
Keywords: ovarian cancer, PARP inhibitors, cancer stem cells, APOBEC3B, drug resistance