Clear Sky Science · en
A recyclable dendrimeric Ni nanocatalyst anchored on magnetic nanoparticles for the green reduction of nitroarene pollutants and one-pot synthesis of imines
Turning Toxic Dyes into Safer Chemicals
Many of the colorful compounds used to make dyes, drugs, and pesticides come with an invisible downside: they leave behind stubborn pollutants in water. This study describes a tiny, magnet-based catalyst that can both neutralize a major class of these pollutants, called nitroarenes, and convert them into useful ingredients for medicines and materials—all in water, at room temperature, and in a way that lets the catalyst be fished back out with a simple magnet.
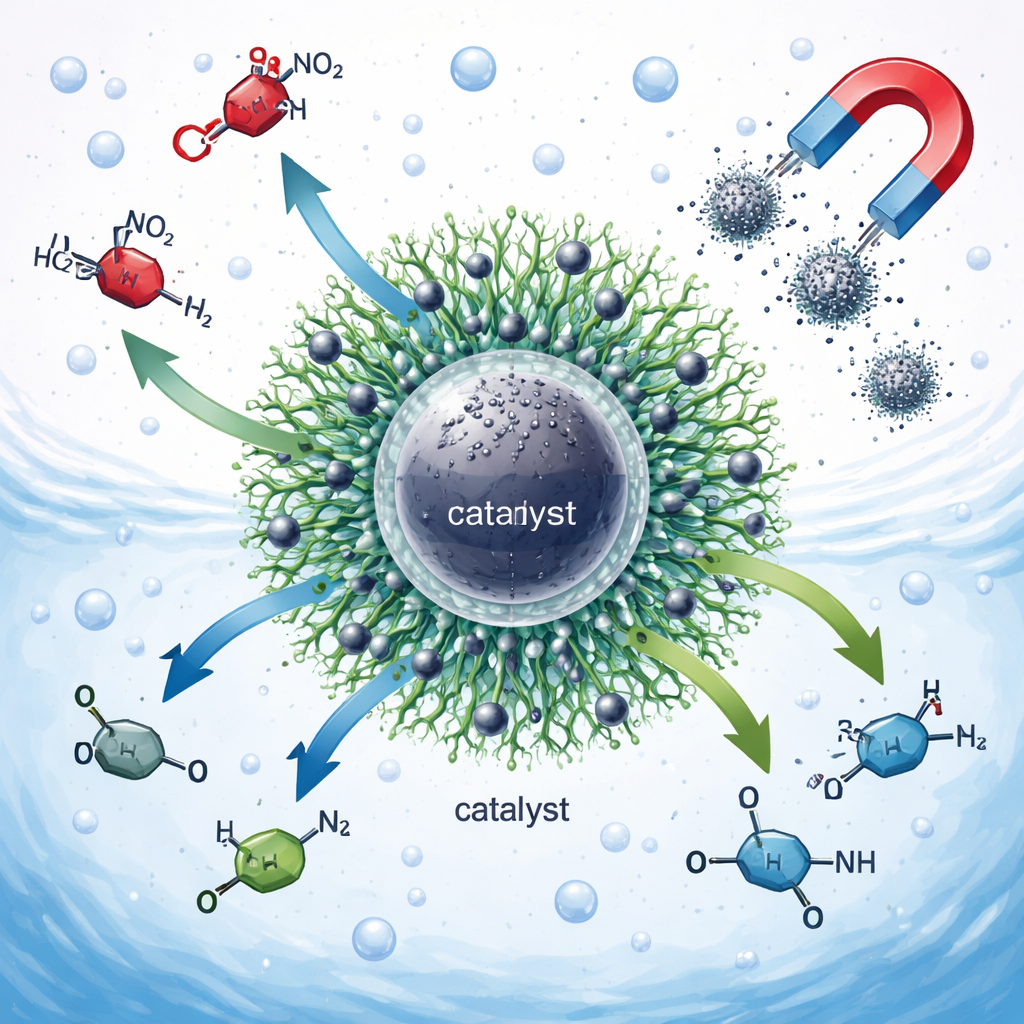
Why Certain Industrial Chemicals Are a Problem
Nitroarenes are ring-shaped organic molecules that contain a nitro group, and they are widely used in chemical manufacturing. Unfortunately, they are also toxic, long-lived in the environment, and frequently found in industrial wastewater. Their safer cousins, aromatic amines, are essential building blocks for dyes, pharmaceuticals, agrochemicals, and special polymers. Converting nitroarenes into amines is therefore a double win: it helps clean water and supplies valuable raw materials. Traditional methods for doing this, however, often rely on expensive precious metals, harsh conditions, or hazardous hydrogen gas, which limit how green and economical they can be.
Building a Tiny Magnetic Workhorse
The researchers designed a nanometer-scale catalyst with several working parts carefully integrated. At its heart is a magnetic iron oxide core coated with a thin layer of silica, which provides chemical stability and an easy way to modify the surface. On this shell they attached a highly branched molecule known as a dendrimer—a tree-like structure with many arms and oxygen-rich end groups. These branches act like a molecular sponge that can tightly hold nickel atoms, an abundant and inexpensive metal known for its ability to promote hydrogen-based reactions. The finished material, called Ni–PAMAM@SMNPs, was thoroughly examined using a suite of techniques to confirm its layered structure, particle size of just a few nanometers, strong magnetism, and uniform dispersion of nickel throughout the dendrimer shell.
Cleaning Up Pollutants in Water
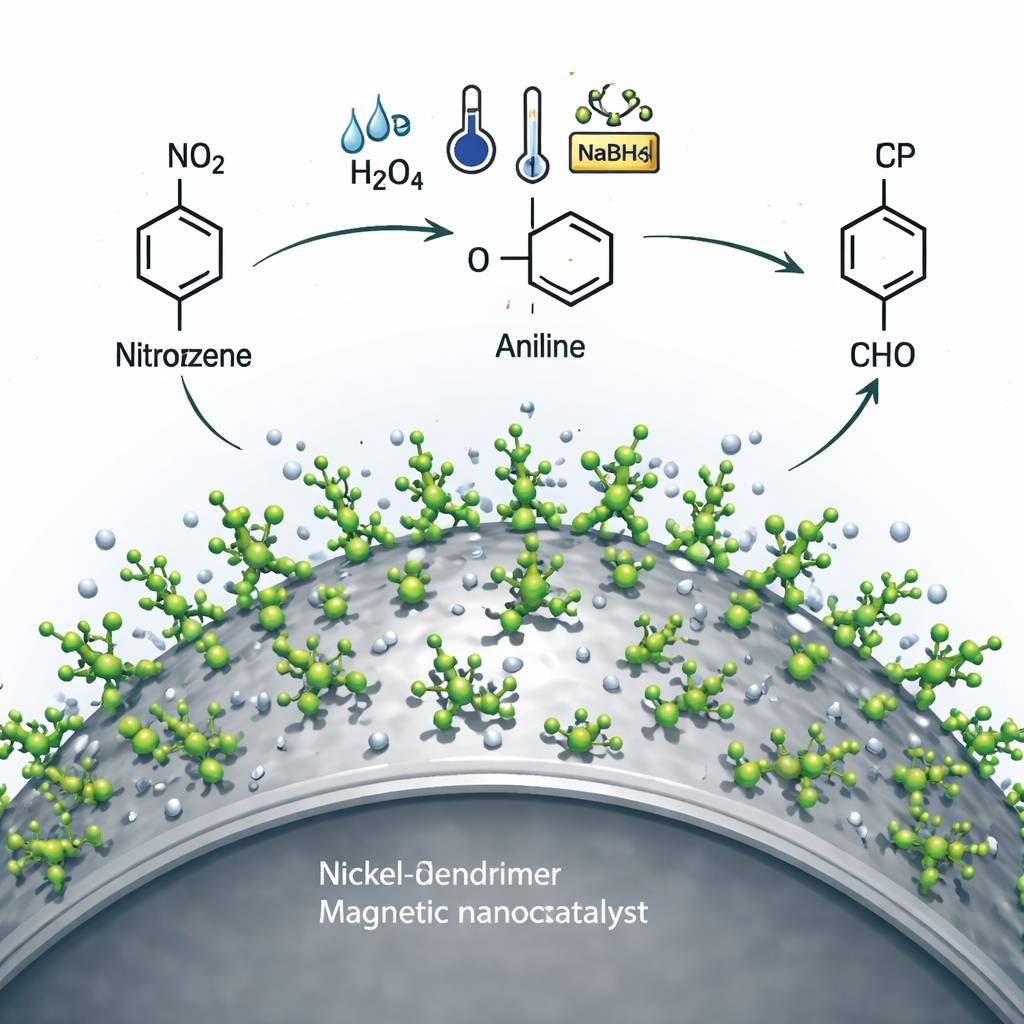
To test performance, the team used sodium borohydride, a common hydrogen donor, to drive the reduction of nitroarenes in water at room temperature. Under optimized conditions, very small amounts of the catalyst rapidly converted a wide range of nitro compounds into their corresponding amines in high to nearly quantitative yields. Importantly, the process showed excellent selectivity: the nitro group was reduced while other sensitive features on the molecules—such as halogens, nitriles, carbonyls, and carboxylic acids—remained intact. This selectivity is crucial when dealing with complex molecules, for example in pharmaceutical intermediates. The authors propose that nickel sites on the dendrimer surface break down the borohydride into highly reactive hydrogen species, which then stepwise transform nitro groups into amines on the catalyst surface.
Making More Complex Molecules in One Pot
Beyond simple cleanup, the catalyst also enables a more sophisticated "one-pot" process. After the nitroarene is reduced to an amine in the same water-based mixture, an aldehyde is added. The freshly formed amine and aldehyde then combine to produce an imine—a versatile class of compounds useful in medicine and materials science—without needing to isolate any intermediates. The dendrimer shell provides acidic and basic sites that help activate both partners, while the nickel centers continue to manage hydrogen transfer. Across many different combinations of nitroarenes and benzaldehyde derivatives, the system delivered imines in high yields under mild conditions, showing that this approach is broadly applicable.
Reusable and Ready for Greener Chemistry
Because the catalyst particles contain a magnetic core, they can be pulled out of the reaction mixture simply by applying an external magnet, washed, and reused. The study shows that the catalyst maintains most of its activity over at least six cycles, with only minimal nickel loss and no detectable structural damage. For a lay reader, the takeaway is that the researchers have built a tiny, reusable "factory" that floats in water, transforms dangerous industrial contaminants into useful chemicals under gentle conditions, and can be collected and used again. This kind of smart, magnetically recoverable nanocatalyst brings industry a step closer to cleaner manufacturing and more sustainable treatment of wastewater.
Citation: Sadeghi, S., Maleki, B. A recyclable dendrimeric Ni nanocatalyst anchored on magnetic nanoparticles for the green reduction of nitroarene pollutants and one-pot synthesis of imines. Sci Rep 16, 6594 (2026). https://doi.org/10.1038/s41598-026-35919-2
Keywords: nitroarene pollutants, magnetic nanocatalyst, nickel catalyst, green chemistry, imine synthesis