Clear Sky Science · en
Supramolecular aggregation of aquaporin-4 shapes astrocyte collective migration and mechanics
How Brain Cells Move Together to Heal
When the brain is injured or inflamed, its support cells—astrocytes—rush in to protect, repair, or wall off damaged areas. This study explores a surprising factor that helps determine how well these cells move together: tiny water channels called aquaporin-4 (AQP4). By looking at how AQP4 molecules clump or spread out in astrocyte membranes, the researchers reveal how the brain’s “water plumbing” helps coordinate collective cell motion, and how chronic inflammation can derail this process.
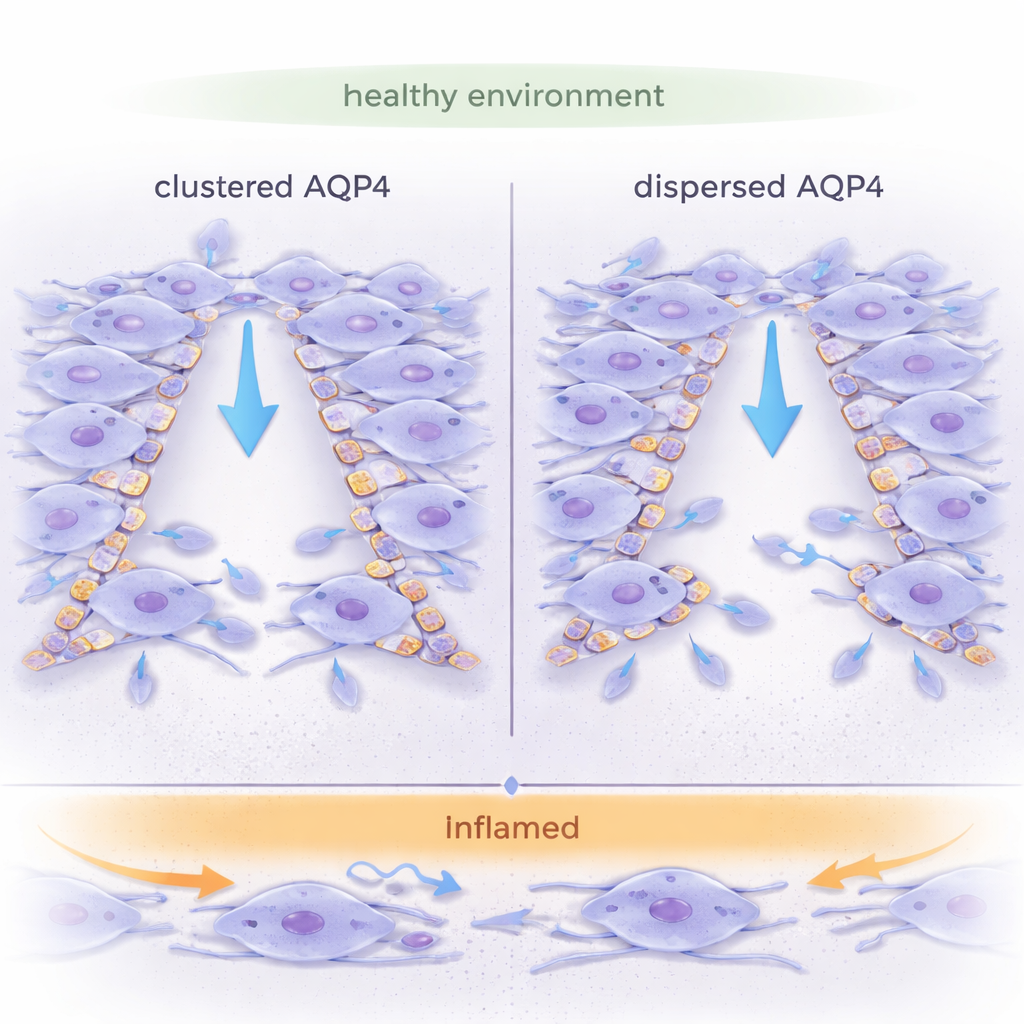
Water Gateways on Brain Cells
Astrocytes are star-shaped cells that help maintain brain balance, guide development, and respond to injury. They are packed with AQP4, a protein that forms pores letting water slip quickly in and out of the cell. Unlike many other water channels, AQP4 can assemble into large crystalline patches called orthogonal arrays of particles (OAPs), or remain as smaller, scattered units (tetramers). The way AQP4 is organized is thought to influence how cells change shape and move, but most earlier work focused on single cells. This study asked a more realistic question: how does AQP4 organization affect the way whole sheets of astrocytes migrate together, as they would when closing a wound in brain tissue?
Testing Cell Movement in Health and Inflammation
The researchers grew two types of mouse astrocytes: normal cells that can form AQP4 arrays, and genetically modified cells (OAP-null) that lack the main array-forming version of AQP4 and therefore carry mostly dispersed tetramers. They then created a “scratch” in a dense layer of cells, mimicking a wound, and watched how quickly and smoothly the cells closed the gap. To mimic a chronically injured brain, they also bathed some cultures in two inflammatory molecules, IL-1β and TNF-α, for a week before testing. Using time-lapse imaging and a computer-vision method called particle image velocimetry, they quantified not only how far and how fast cells moved, but also how straight, coordinated, or strained their motion was across the entire sheet.
Dispersed Channels, Smoother Motion
Under non-inflamed conditions, astrocytes with dispersed AQP4 (OAP-null) were markedly better at collective migration: they closed wounds faster and moved in a more directed, linear fashion than cells with large AQP4 arrays. Their leading front was smooth and continuous, and neighboring cells moved in a cohesive, “sheet-like” way. In contrast, astrocytes with clustered AQP4 (wild type) showed a more irregular front, with many individual protrusions and internal distortions, suggesting that cells were pulling in slightly different directions. Measurements of strain within the cell layer confirmed that OAP-null sheets experienced less internal tug-of-war and more uniform, coordinated motion.
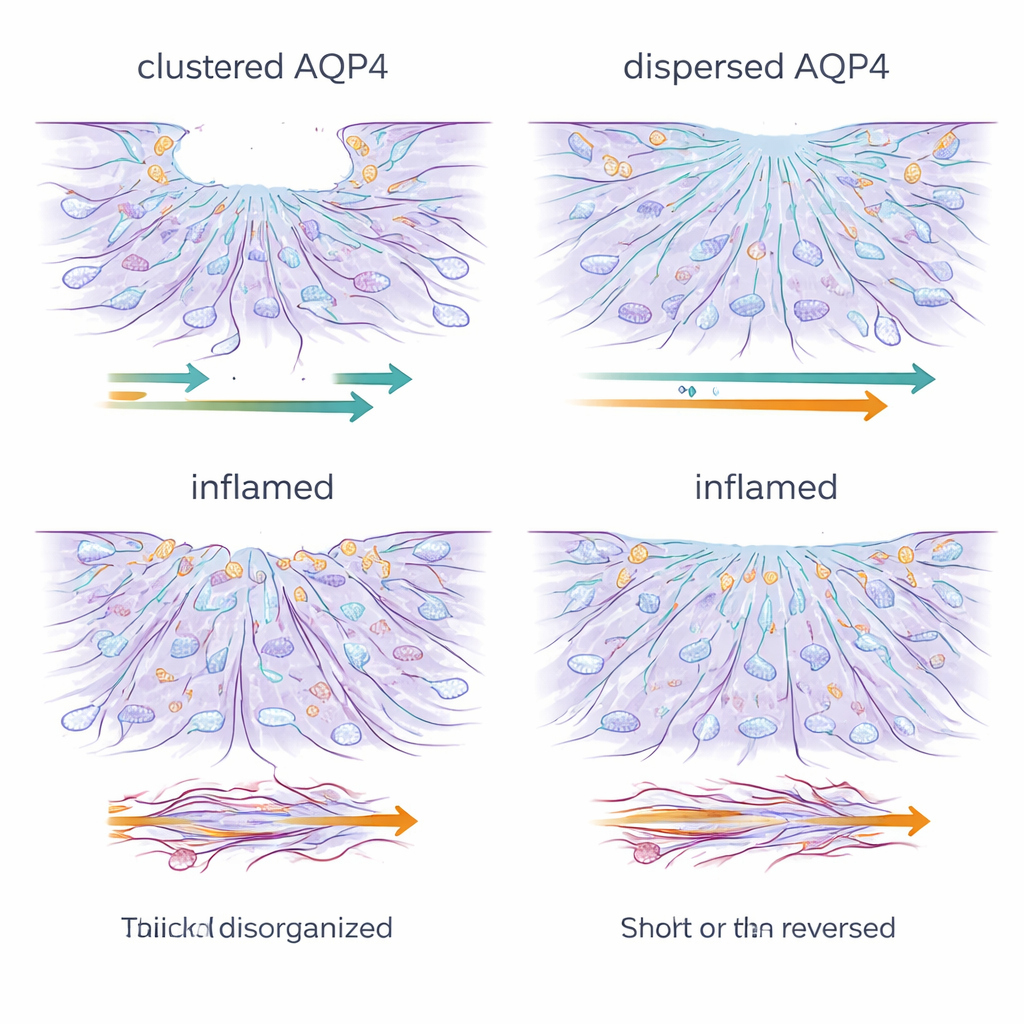
Inflammation Stalls and Scrambles the Repair Crew
Chronic exposure to inflammatory signals profoundly changed this picture. Regardless of AQP4 organization, treated astrocytes became poor wound healers: they barely moved, and in some cases even drifted backward, enlarging the gap. Microscopy showed that the fine, dynamic cell-edge structures needed for forward movement—lamellipodia and filopodia—were lost and replaced by thick, rigid stress fibers of actin, the cell’s main structural protein. At the same time, levels of AQP4 and of connexin-43, a key protein that forms communication channels between astrocytes, dropped sharply. The network of gap junctions that normally helps astrocytes act as a coordinated unit was disrupted, and dye-transfer experiments confirmed that long-range cell–cell communication was severely weakened.
Why This Matters for Brain Health
These findings suggest that it is not just how much AQP4 a cell has, but how it is arranged that helps astrocytes move efficiently as a group. Dispersed AQP4 appears to reduce internal resistance between cells and promote straighter, more coordinated motion, while large clusters are associated with a more erratic, strained style of movement. Chronic inflammation largely overrides these advantages by remodeling the cell skeleton, silencing water and communication channels, and turning an organized repair crew into a rigid, poorly connected sheet. For non-specialists, the takeaway is that the brain’s water channels and cell–cell communication systems do far more than passively support neurons—they actively shape how support cells mobilize after injury. Understanding and eventually tuning AQP4 organization and inflammatory signals could open new avenues to improve brain repair, limit scarring, or even influence how brain tumors spread.
Citation: Barile, B., Mennona, N.J., Mola, M.G. et al. Supramolecular aggregation of aquaporin-4 shapes astrocyte collective migration and mechanics. Sci Rep 16, 6021 (2026). https://doi.org/10.1038/s41598-026-35900-z
Keywords: astrocytes, aquaporin-4, brain inflammation, cell migration, glial scar