Clear Sky Science · en
Inhibiting autophagy enhances anti-cancer properties of sulforaphane
Why broccoli chemicals and an old malaria pill matter
Bladder cancer is common and often deadly once it spreads, and many patients eventually stop responding to treatment. This study explores an unexpected team-up: sulforaphane, a natural compound from broccoli, and chloroquine, a long-used anti-malarial drug. Together, they interfere with cancer cells’ internal recycling system and movement, in ways that could one day help make bladder cancer therapies more effective and longer lasting.
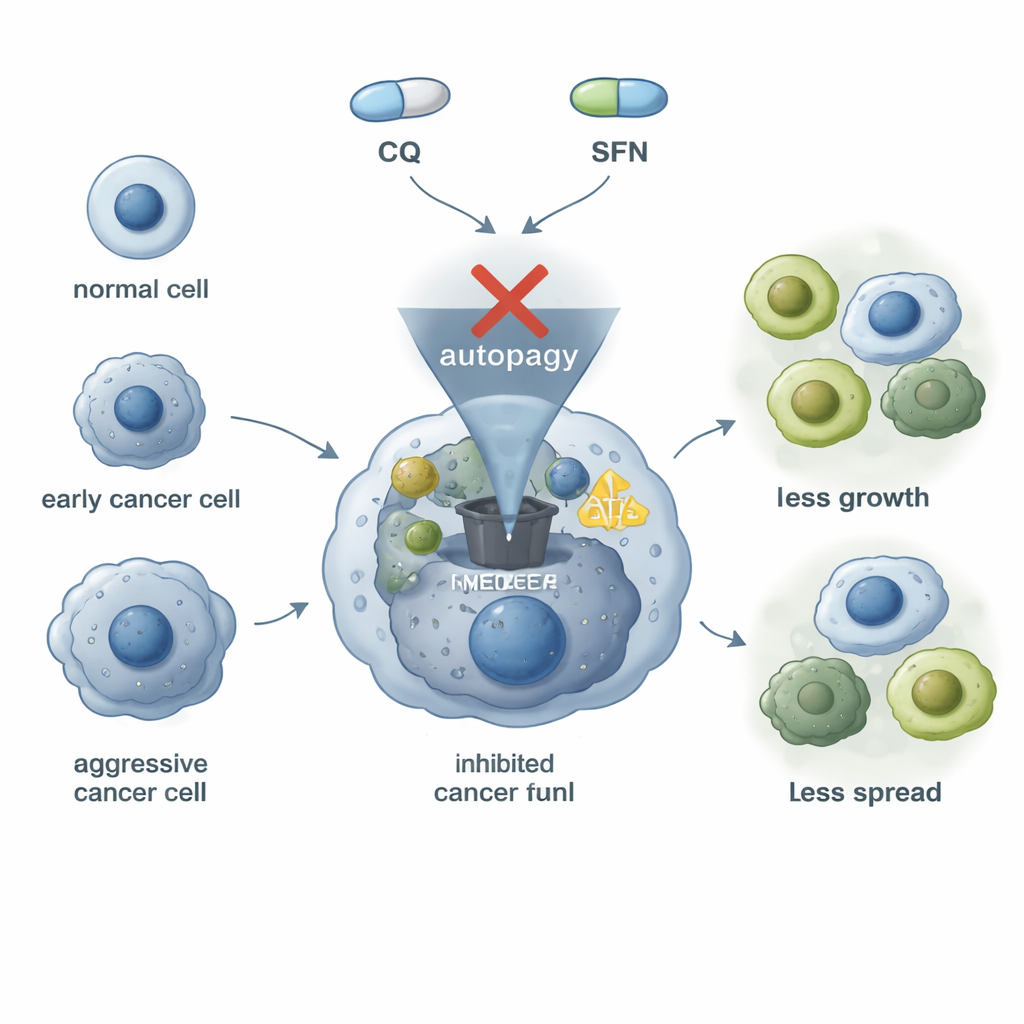
The challenge of stubborn bladder tumors
Bladder cancer is not a single disease; tumors differ in their genes, how aggressive they are, and how they respond to therapy. Many bladder cancer cells rely on a high level of “self-cleaning,” a process called autophagy, to survive stress, repair damage, and resist drugs. The same cells often show high amounts of a surface protein called ICAM-1 and another protein, N-cadherin, both linked to a more mobile, invasive state that makes it easier for cancer to spread. Because standard treatments do not fully address these survival tricks, researchers are searching for ways to shut down both the recycling machinery and the invasive behavior at the same time.
Repurposing chloroquine and tapping into broccoli’s defenses
Chloroquine, best known as an anti-malarial medicine, can block the last step of autophagy: the moment when waste-filled sacs fuse with acidic compartments that break materials down into usable fuel. On its own, chloroquine slowed the growth and movement of three different bladder cancer cell lines in the lab, showing a basic anti-cancer effect. But it also unexpectedly raised ICAM-1 levels in all of these cancer cells, a worrying sign because higher ICAM-1 is associated with more aggressive behavior. Sulforaphane, a compound naturally present in cruciferous vegetables like broccoli, has been reported to slow the growth of many cancer types and, importantly, to lower ICAM-1 levels. This made it an attractive partner for chloroquine: one drug to block the recycling system, and one to dampen a key marker of aggressiveness.
How the drug duo weakens cancer cells
The researchers treated three bladder cancer cell lines with chloroquine, sulforaphane, or both and tracked a network of signaling proteins that control growth, movement, and autophagy. Chloroquine consistently blocked autophagy, leading to a buildup of standard recycling markers inside the cells. When sulforaphane was added, it strengthened several of chloroquine’s desirable effects: it reduced ICAM-1 and N-cadherin levels in two of the three cell lines, pushed beta-catenin (a growth-related protein) toward breakdown, and altered key switches (such as AKT, GSK-3β, mTOR, and ULK) in ways that overall discouraged survival and self-cleaning. At the same time, the combined treatment damaged mitochondria—the cell’s power plants—and, depending on the cell line, changed the balance of reactive oxygen species, small molecules that can tip cells toward self-destruction.
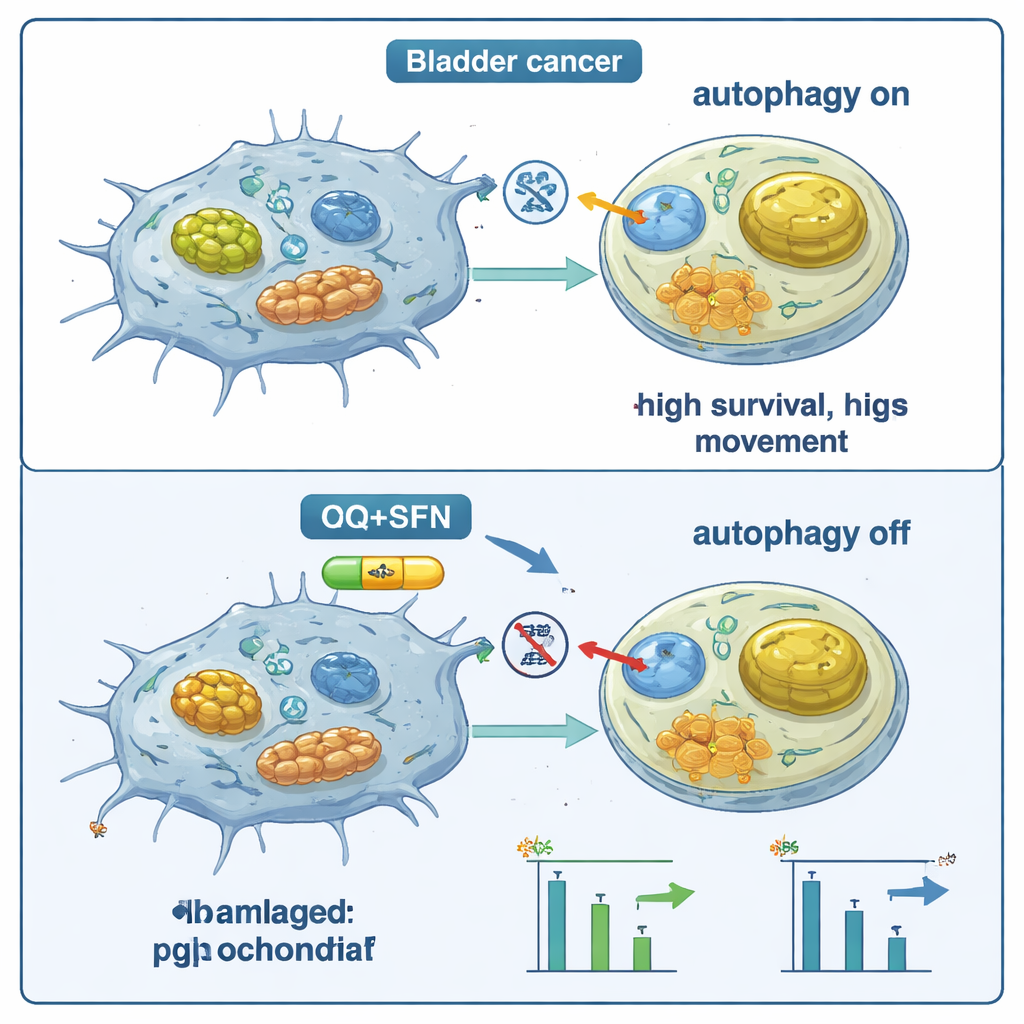
Slower growth and less movement depend on cell type
Beyond these molecular changes, the team asked two practical questions: do the cells divide less, and do they move less? In all three bladder cancer cell lines, the combination of sulforaphane and chloroquine reduced cell proliferation, with especially strong effects in one line that already had high ICAM-1 levels. A scratch “wound” assay showed that treated cells were slower to crawl in and close a gap, indicating reduced migratory potential, again with the strongest effects when both agents were used together. However, not all cell lines responded in the same way. In one line with relatively low ICAM-1, blocking autophagy sometimes appeared to protect the cancer’s aggressive pattern, underscoring that the outcome depends heavily on each tumor’s molecular wiring.
What this could mean for future treatments
For a non-specialist, the main message is that cancer cells use an internal recycling program and certain surface proteins to stay alive, divide, and spread, and that these features can be attacked from more than one angle. In this study, sulforaphane helped counteract an unwanted side effect of chloroquine—its tendency to increase a pro-tumor marker—while boosting the overall anti-cancer impact on bladder cells grown in the lab. The findings do not yet translate directly into a clinic-ready therapy, and they highlight that some tumors may even benefit from autophagy blockage, depending on their makeup. Still, the work supports the idea that carefully chosen combinations of a common drug and a natural dietary compound could be tailored to tumor type to slow bladder cancer growth, limit spread, and improve responses to existing treatments.
Citation: Zarzycka, M., Kotula-Balak, M. & Gil, D. Inhibiting autophagy enhances anti-cancer properties of sulforaphane. Sci Rep 16, 5296 (2026). https://doi.org/10.1038/s41598-026-35891-x
Keywords: bladder cancer, autophagy, sulforaphane, chloroquine, ICAM-1