Clear Sky Science · en
Reduced expression of BIRC2 and BIRC3 associated with longer survival in pediatric high-grade gliomas
Why children’s brain tumors need new clues
Pediatric high-grade gliomas are among the most deadly childhood brain tumors, and current surgery, radiation, and chemotherapy help only a small minority of patients. This study asks a simple but crucial question: are there molecular “survival switches” inside these tumors that help cancer cells resist dying, and can those switches help explain why some children live longer than others?
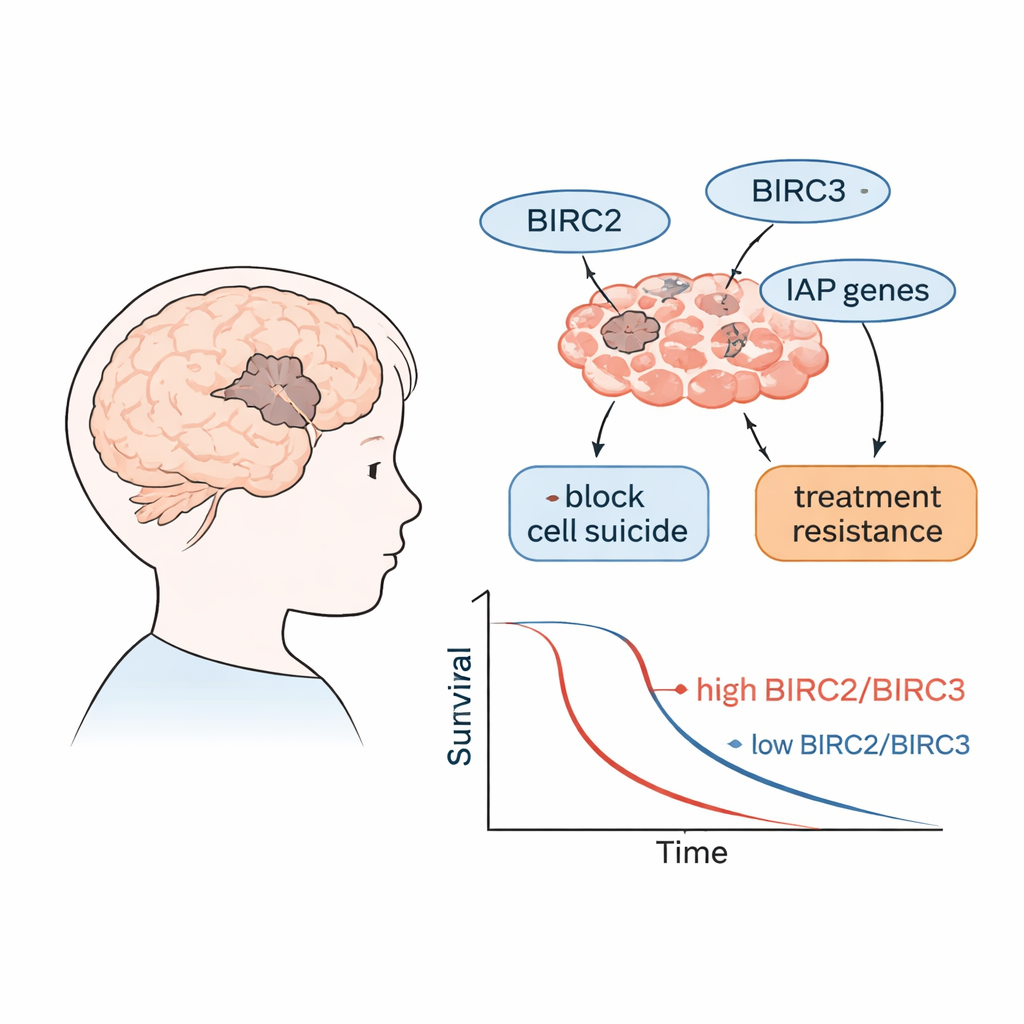
Proteins that won’t let cancer cells die
Our cells are equipped with a built‑in self‑destruct system, often called programmed cell death or apoptosis, that removes damaged or dangerous cells. A family of proteins known as IAPs (inhibitors of apoptosis proteins) can block this self‑destruct machinery. The genes that make these proteins are called BIRC genes. In many cancers, IAPs are turned up too high, helping tumor cells ignore death signals and continue to grow. The researchers focused on several members of this family, including BIRC2 and BIRC3, along with related genes that either promote or oppose cell death, to see how active they are in aggressive brain tumors from children.
Studying real tumors from young patients
The team analyzed tumor samples from 26 young people, all treated at a pediatric neuro‑oncology center and all diagnosed with the most aggressive form of glioma. Using preserved tissue taken during surgery, they measured the activity of multiple genes linked to cell survival and death, such as BIRC2, BIRC3, BIRC5, BIRC6, BIRC7, NAIP, XIAP, DIABLO, XAF1, CASP3, and CASP9. They also compared these molecular readouts with clinical features: how long each patient lived, how long the disease stayed stable before worsening, and the presence of common tumor markers like Ki‑67 (a growth indicator), PD‑1 (an immune checkpoint), Olig2, p53, GFAP, and a small regulatory RNA called miR‑155‑5p.
Survival switches tied to shorter lives
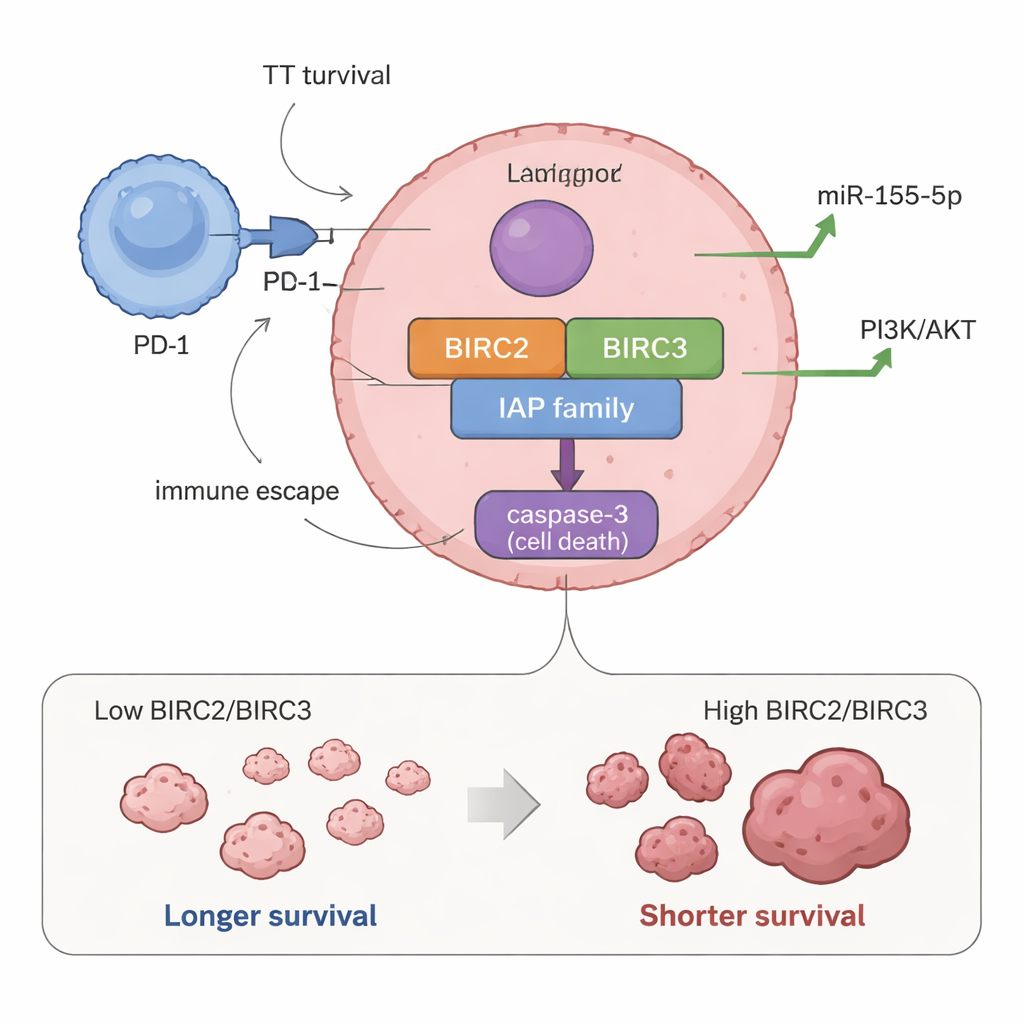
When the researchers compared gene activity with patient outcomes, two genes stood out. Children whose tumors had higher levels of BIRC2 and BIRC3 tended to have shorter overall survival and shorter periods without tumor progression. In other words, when these “don’t‑die” switches were more strongly turned on, the disease behaved more aggressively. Tumors with high BIRC2 and BIRC3 were also linked to higher levels of miR‑155‑5p, a small RNA previously shown to push cells toward growth and away from cell death, suggesting these molecules may work together to harden tumors against treatment.
Links to tumor growth and immune evasion
The study also explored how these survival switches fit into the wider tumor environment. Surprisingly, some genes that can encourage or regulate cell death, such as NAIP, BIRC3, and XAF1, were more active in tumors that lacked the proliferation marker Ki‑67, hinting at a complex balance between growth and death signals. Higher BIRC3 and XAF1 levels tended to appear in tumors with more PD‑1, a key immune “brake” that helps cancer hide from the body’s defenses. In addition, higher activity of CASP3, a core executioner of cell death, was strongly associated with PD‑1 expression, suggesting that interactions between cancer cells and immune cells might reshape how the death machinery is used or blocked within the tumor.
What this could mean for future treatment
For families and clinicians confronting pediatric high‑grade gliomas, these findings do not yet change treatment, but they provide important clues. The work suggests that high activity of the BIRC2 gene in particular, and possibly BIRC3, marks tumors that are more resistant and more likely to return quickly, making these genes potential warning flags for poor prognosis. Because IAP proteins are already being studied as drug targets in adult cancers, the new results raise the possibility that medicines designed to shut off these survival switches might one day be combined with existing brain tumor treatments or with immune therapies that target PD‑1. The study is small and exploratory, so its conclusions must be confirmed in larger groups and at the protein level, but it points toward a future where a child’s tumor could be profiled for these key switches to better predict outcome and, eventually, to guide more tailored and effective therapies.
Citation: Petniak, A., Gil-Kulik, P., Zarychta, J. et al. Reduced expression of BIRC2 and BIRC3 associated with longer survival in pediatric high-grade gliomas. Sci Rep 16, 6665 (2026). https://doi.org/10.1038/s41598-026-35887-7
Keywords: pediatric brain tumor, glioma, apoptosis, BIRC2 BIRC3, immunotherapy