Clear Sky Science · en
A comprehensive study of the crystal structure and dynamics of [N(C3H7)4]2Cd2Cl6
Why this shape-shifting crystal matters
Materials that change their internal structure as they heat up can act like tiny switches inside future electronics, sensors, or optical devices. This study looks at an unusual "hybrid" crystal made of both organic molecules and inorganic metal–salt units, with the long name [N(C3H7)4]2Cd2Cl6. By carefully tracking how this crystal’s structure and atomic motions evolve with temperature, the researchers show how its metal–chlorine units quietly reorganize themselves, while the surrounding organic parts stay largely calm. Understanding this hidden rearrangement is a key step toward designing smarter, more reliable functional materials.
Building a hybrid crystal
The team first grew high-quality single crystals from water, combining a tetrapropylammonium salt with cadmium chloride and letting the solution slowly evaporate. The result is a transparent, square-shaped hybrid crystal where bulky organic ions form a soft framework that separates clusters of inorganic Cd2Cl6 units. In this family of materials, the organic component mainly tunes optical and structural flexibility, while the inorganic metal–halide clusters control thermal stability and mechanical strength. By choosing different metals and halides, scientists can dial in a wide range of electrical, magnetic, and optical behaviors, making this crystal an informative model for a much larger class of functional hybrids.
Watching the crystal change with heat
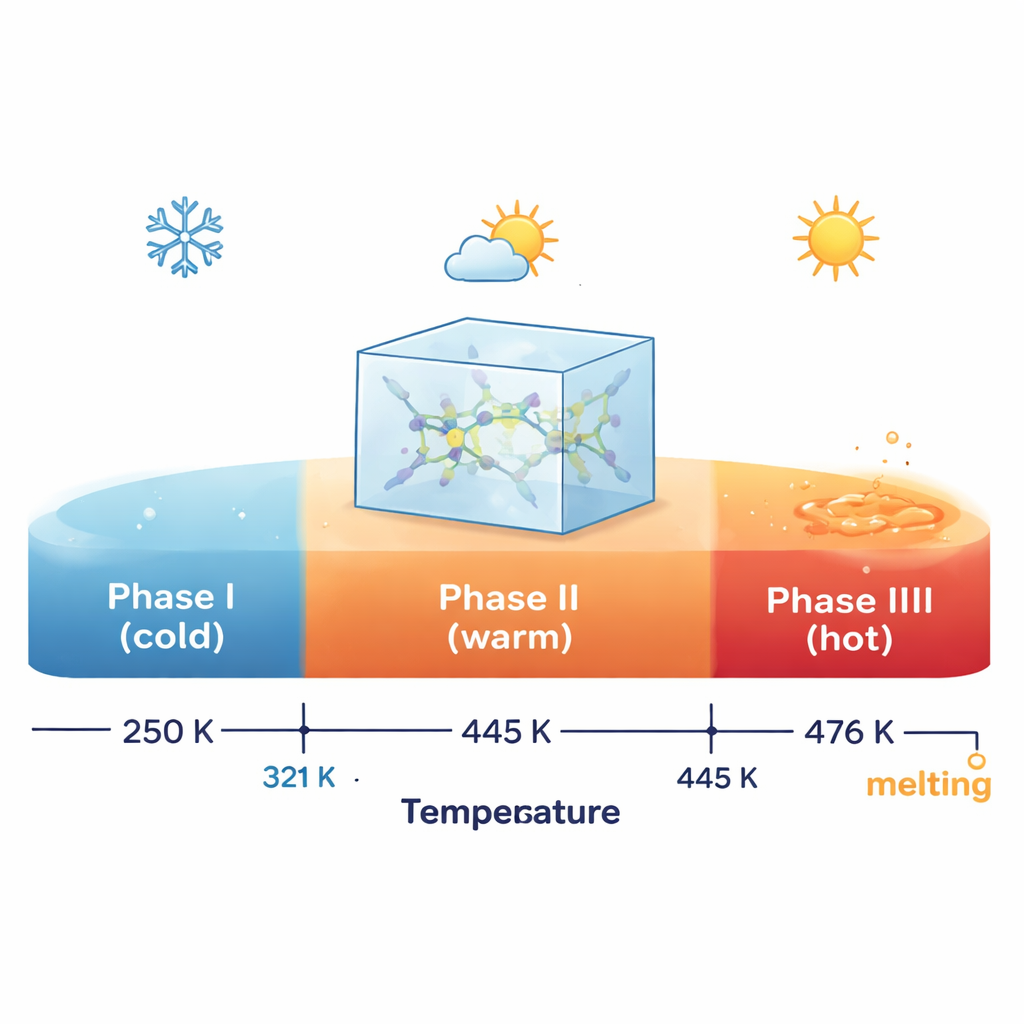
To see how the material responds to heating, the researchers used a suite of thermal measurements. Differential scanning calorimetry and related techniques revealed two distinct internal changes, or phase transitions, at about 321 K and 445 K (roughly 48 °C and 172 °C), followed by melting near 476 K. Under the microscope, the crystal kept its overall shape up to just below the melting point, so these transformations are subtle internal rearrangements rather than cracking or distortion. Thermogravimetric analysis showed that the material remains chemically stable up to around 546 K, only then beginning to decompose in steps as the organic ions and their chlorides break down, eventually leaving a cadmium–chloride residue. Together, these tests map out a clear “thermal life cycle” from solid phases through melting to decomposition.
Shifts in the invisible scaffolding
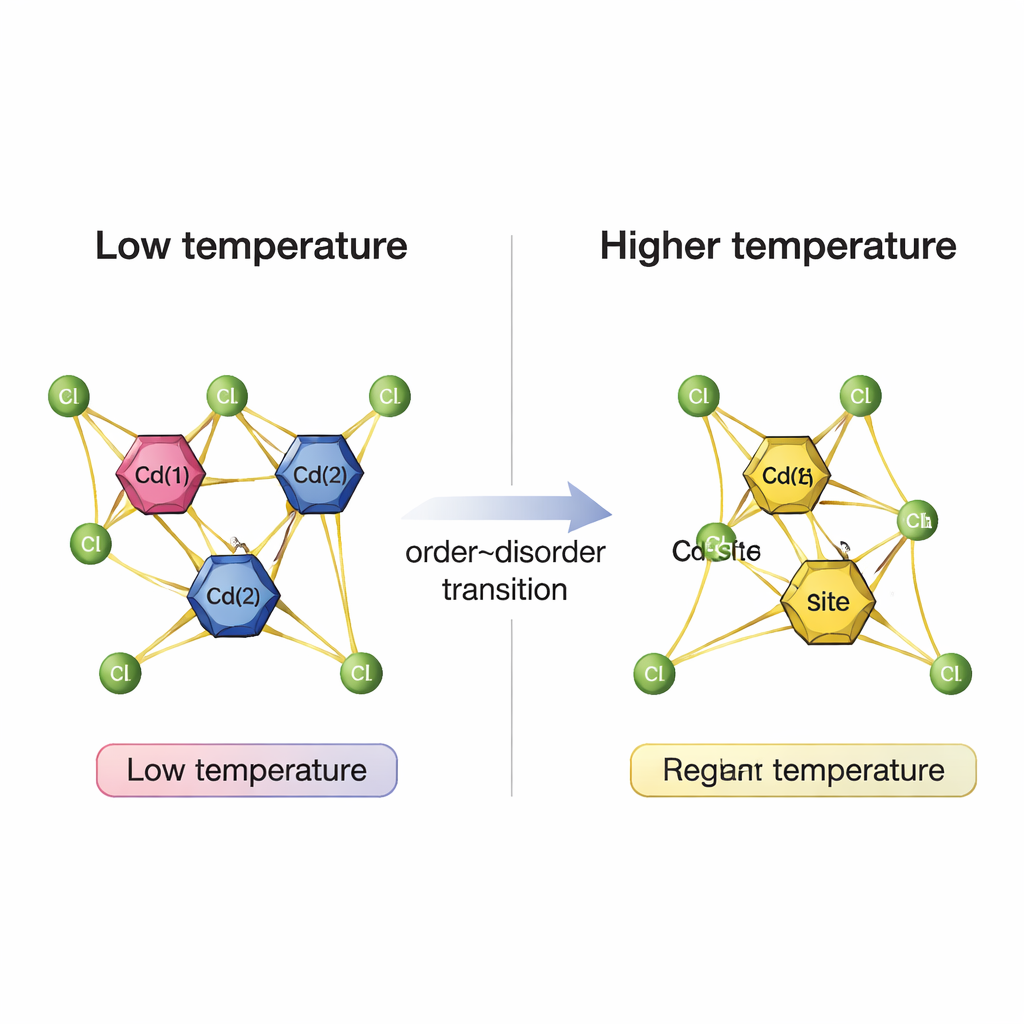
Single-crystal and powder X-ray diffraction provided a detailed picture of how the atomic lattice responds across the first transition. At room temperature, the crystal has a low-symmetry triclinic arrangement with two formula units per unit cell and two distinct cadmium–chloride clusters. When heated above 321 K, the material keeps the same overall symmetry but its lattice dimensions jump, and the unit cell now contains only one formula unit. This implies a structural simplification: the two previously distinct Cd2Cl6 clusters become equivalent, even though the surrounding organic ions maintain a similar average arrangement. Powder diffraction patterns confirm that the change between the first and second solid phases is modest, while the jump to the highest-temperature solid phase is more dramatic, hinting at a more symmetric structure before melting.
Listening to atoms in motion
To probe what the atoms themselves are doing, the team turned to magic-angle spinning nuclear magnetic resonance (MAS NMR), which is sensitive to the local environment and motion of specific nuclei. Signals from hydrogen, carbon, and nitrogen in the organic tetrapropylammonium ions changed only slightly near the first transition, although their line widths narrowed steadily with increasing temperature. This narrowing indicates that these ions gradually move and reorient more freely as the crystal warms, but without undergoing a sharp rearrangement at 321 K. In striking contrast, the NMR signal from cadmium in the Cd2Cl6 units showed a clear fingerprint of the phase change: at low temperature there are two cadmium environments, but above 321 K these merge into one, and the lines become narrower as motion increases.
What the crystal is really doing
Putting all the measurements together, the researchers conclude that the first phase transition in [N(C3H7)4]2Cd2Cl6 is driven mainly by an order–disorder change in the cadmium–chloride clusters, not by the organic ions. As temperature rises, the two distinct cadmium sites become dynamically and structurally equivalent, while the soft organic framework simply becomes more mobile. The crystal thus acts like a quiet internal switch that reorganizes its inorganic backbone without visibly changing shape. This detailed understanding of how structure and motion are linked in a hybrid crystal lays groundwork for designing new materials whose internal rearrangements can be harnessed for future electronic, optical, or sensing applications.
Citation: Ju, H., Shin, Y.S. & Lim, A.R. A comprehensive study of the crystal structure and dynamics of [N(C3H7)4]2Cd2Cl6. Sci Rep 16, 5309 (2026). https://doi.org/10.1038/s41598-026-35886-8
Keywords: organic–inorganic hybrid crystals, phase transitions, cadmium chloride complexes, solid-state NMR, crystal structure