Clear Sky Science · en
In vitro characterization and in ovo embryotoxicity assessment of a triazol-5-one derivative in broiler embryos
Why a new lab-made chemical matters for baby chicks
Modern poultry farms are looking for ways to keep chickens healthy without relying heavily on antibiotics. One promising group of lab-made chemicals, called triazoles, can fight germs and affect the body’s defenses against harmful molecules. Before such compounds can be used around food animals, however, scientists must be sure they are safe—especially for fragile developing embryos inside eggs. This study asked a simple but crucial question: what happens to broiler chicken embryos when a powerful triazole-based compound is placed directly into the egg shortly before hatching?
A new chemical with strong germ-fighting power
The research team first created a specific triazol-5-one compound using standard organic chemistry steps, then confirmed its structure using laboratory techniques that read the positions of atoms in the molecule. Once they were sure they had the right substance, they tested how it behaved in test-tube experiments. The compound turned out to be a weak defender against free radicals—unstable molecules that can damage cells—but it showed very strong ability to grab and hold on to metal ions such as iron, zinc, and copper. It also blocked the growth of several important bacteria, including both common “Gram-positive” and “Gram-negative” species, with an effect similar to some older antibiotics in certain cases. 
From test tube to egg: trying the compound in developing embryos
To see how this promising lab result would translate to living animals, the scientists used a method known as in ovo injection: delivering substances into the egg before the chick hatches. They worked with fertilized eggs from a common meat chicken line (Ross 308) and divided 120 eggs into three groups. One group was left untouched as a control. A second group received only the solvent mixture, a small amount of dimethyl sulfoxide diluted in a salt solution, to check whether the injection process or carrier liquid harmed the embryos. The third group received the same injection volume but contained 15 milligrams of the new triazole compound suspended in the salt solution. All eggs were handled under clean conditions and incubated using standard temperatures and humidity so that any differences in outcome would most likely reflect the compound itself.
When a promising molecule becomes deadly in the egg
At the end of the normal 21-day incubation period, the differences between groups were stark. In the untreated control eggs, nearly nine out of ten embryos hatched into live chicks. In the solvent-only group, hatchability was slightly lower but still high, at 80 percent. In sharp contrast, none of the embryos that received the triazole compound survived to hatching—hatchability in that group was 0 percent. Statistical checks showed that the starting weights of the eggs were similar across all groups, ruling out egg size as a cause. All embryo losses in the treated group occurred after the injection on day 17, a time when the embryo’s oxygen needs and metabolism are rapidly rising, suggesting that the compound’s effects were tightly linked to this sensitive late stage of development. 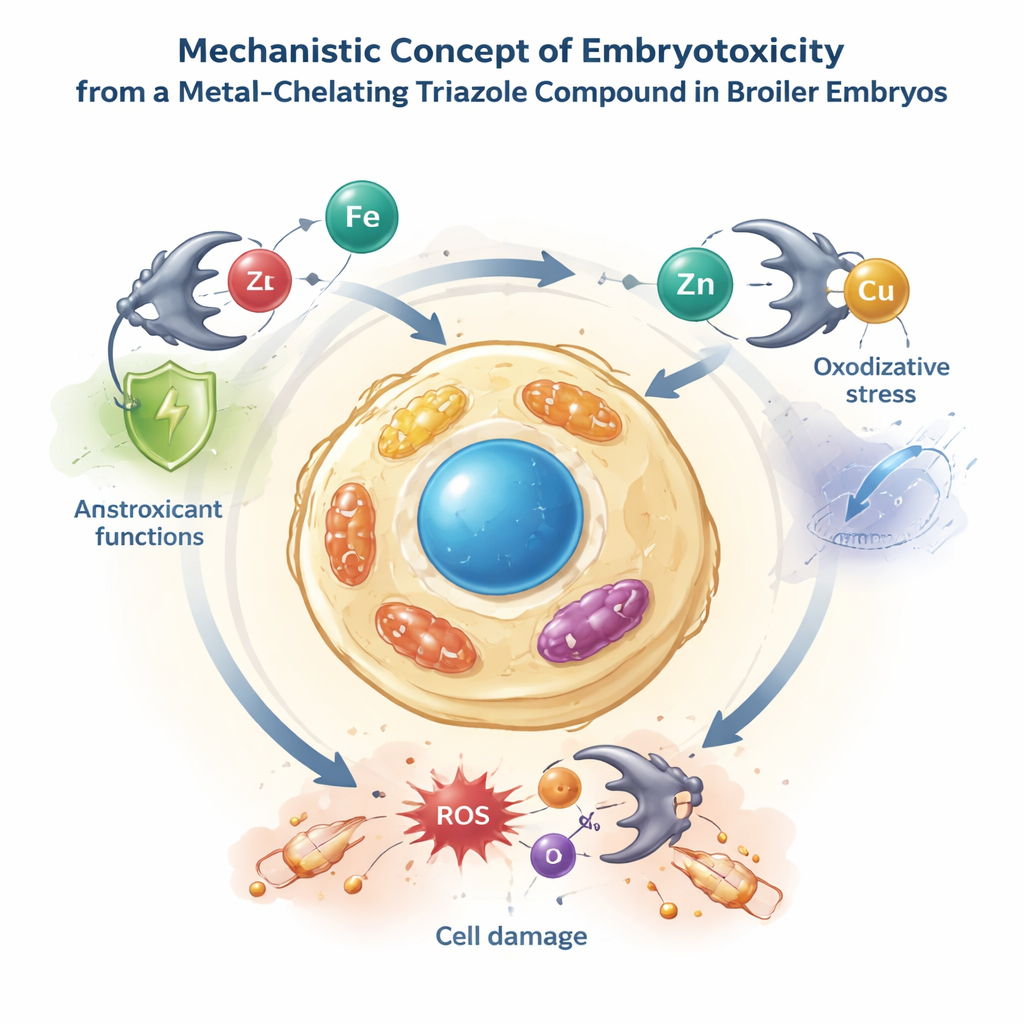
Possible reasons behind the embryo losses
Why would a compound that looks useful in a petri dish kill embryos in an egg? The authors point to a combination of its laboratory-measured properties. Because the molecule strongly binds metal ions but offers little direct antioxidant protection, it may strip essential metals away from enzymes that protect the embryo against oxidative stress, while failing to neutralize damaging free radicals. Its antimicrobial action, which often involves disturbing cell membranes and basic energy processes, could also accidentally harm the embryo’s own cells. The fact that the compound was delivered as a suspension, rather than fully dissolved, might have created tiny pockets of high local concentration around developing tissues. Together, these factors may have overwhelmed the embryo’s ability to cope, leading to complete embryo death in the treated group.
What this means for future poultry health tools
For non-specialists, the key message is that not every germ-fighting chemical that works well in the lab is safe for use in living animals—especially at early life stages. This particular triazole compound combined strong metal-binding and antibacterial properties with unexpectedly severe toxicity to broiler embryos when given directly into the egg at the tested dose. The study does not rule out safer uses of related compounds, but it highlights the need to carefully test dose, formulation, and timing before any such molecule is considered for on-farm use. In practical terms, the work draws a clear boundary: under these conditions, this triazole derivative is not suitable for in-egg applications, and more cautious, stepwise studies will be required to find any members of this chemical family that can support poultry health without putting developing embryos at risk.
Citation: Durna, Ö., Ulufer Bulut, S., Boy, S. et al. In vitro characterization and in ovo embryotoxicity assessment of a triazol-5-one derivative in broiler embryos. Sci Rep 16, 6450 (2026). https://doi.org/10.1038/s41598-026-35880-0
Keywords: broiler embryos, in ovo injection, embryotoxicity, triazole compound, poultry health