Clear Sky Science · en
KIN17 facilitates the initiation and progression of renal tumor progression through the PI3K-AKT-mTOR pathway
Why this kidney cancer study matters
Kidney cancer is being detected more often, but for many patients the disease is still hard to control once it spreads. This study looks under the microscope—down to the level of individual molecules inside cancer cells—to understand what drives kidney tumors to grow and invade, and whether an experimental drug can put the brakes on that process. The findings point to a little‑known protein, KIN17, as a new troublemaker in kidney cancer and suggest a way to target it indirectly with a pill‑type treatment.
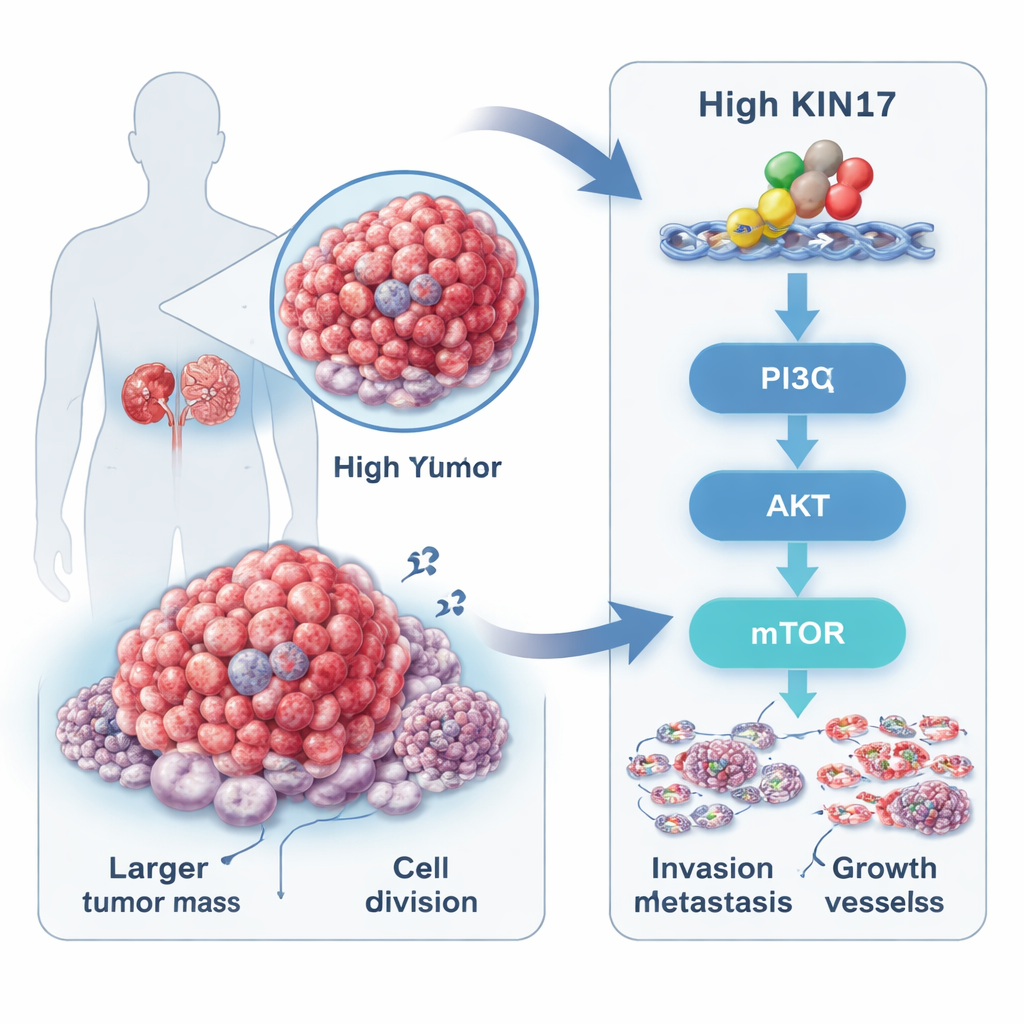
A hidden driver inside kidney tumors
Renal cell carcinoma, the most common form of kidney cancer, often begins without symptoms and is frequently discovered late. The researchers focused on a protein called KIN17, long known for its role in protecting and copying DNA, but only recently linked to several other cancers. By analyzing public cancer databases and tumor samples from 88 patients, they found that KIN17 levels were much higher in kidney tumors than in nearby non‑cancerous tissue. Patients whose tumors contained more KIN17 tended to have larger cancers, higher grade disease, more advanced stage, and more distant spread, and they lived for a shorter time than those with low KIN17. This pattern suggested that KIN17 is not just a bystander, but closely tied to how aggressive a tumor becomes.
Testing KIN17 in cancer cells
To find out what KIN17 actually does inside kidney cancer cells, the team engineered laboratory cell lines to either dial KIN17 down or crank it up. When they reduced KIN17, the cells grew more slowly, formed fewer colonies, and were less able to move and invade through artificial membranes. More of these cells got stuck in the resting phase of the cell cycle and went on to die, showing higher levels of programmed cell death. The opposite happened when KIN17 was overproduced: cells multiplied faster, copied their DNA more readily, closed scratch‑like gaps in cell layers more quickly, and invaded more aggressively. These experiments showed that KIN17 acts like a gas pedal for kidney cancer cell growth and spread.
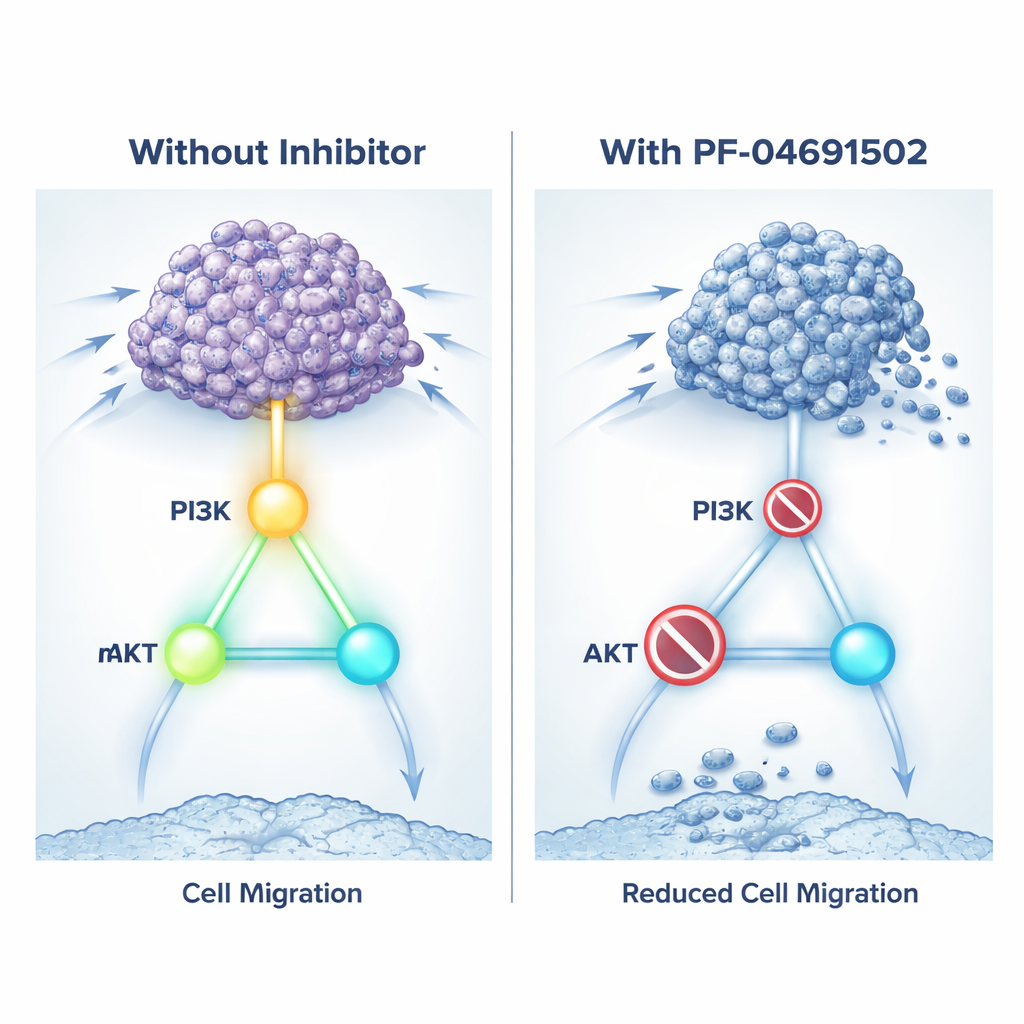
A growth pathway caught in the act
The researchers next asked how KIN17 exerts this power. Their gene‑activity surveys and protein tests pointed to a well‑known growth and survival network inside cells called the PI3K–AKT–mTOR pathway. This pathway is often compared to a central control hub that tells cells when to grow, divide, and avoid death. Kidney cancer cells with high KIN17 had higher "on" signals—chemical tags called phosphates—on key components of this pathway, even though the total amount of these proteins did not change. When KIN17 was knocked down, these activation tags fell, and molecules linked with cell movement and invasion also shifted. In mouse experiments, tumors built from KIN17‑rich cells grew larger and faster, and tissue staining showed that the same growth pathway was strongly activated in those tumors.
Turning down the signal with a dual‑target drug
Because directly blocking KIN17 is not yet possible with existing medicines, the team tested a different tactic: shutting off the growth pathway it seems to control. They used PF‑04691502, an experimental drug that blocks both PI3K and mTOR, two key switches in the pathway. In dishes, treating KIN17‑high kidney cancer cells with this inhibitor slowed their growth, reduced their ability to migrate and form colonies, and pushed more cells into death. The drug also cut the activation tags on PI3K, AKT, and mTOR and lowered levels of invasion‑related proteins. In mice carrying kidney tumors, daily doses of PF‑04691502 shrank tumors that overproduced KIN17 and reduced markers of cell division while increasing signals of cell death. In essence, the drug was able to mute the harmful effects of KIN17 by silencing its favored growth route.
What this could mean for patients
For people facing kidney cancer, these findings suggest two hopeful ideas. First, measuring KIN17 in tumor samples might help doctors judge how aggressive a cancer is and how likely it is to spread. Second, tumors that rely heavily on KIN17 may be especially vulnerable to drugs that hit the PI3K–AKT–mTOR pathway, such as PF‑04691502 or similar dual‑target inhibitors already in clinical trials for other cancers. While much work remains before any new treatment reaches the clinic, this study maps out a clear chain of events—from KIN17, through a key growth pathway, to uncontrolled tumor expansion—and shows that breaking that chain can slow kidney cancer in the lab and in animals.
Citation: Wen, Y., Lyu, L., Zhang, H. et al. KIN17 facilitates the initiation and progression of renal tumor progression through the PI3K-AKT-mTOR pathway. Sci Rep 16, 5721 (2026). https://doi.org/10.1038/s41598-026-35851-5
Keywords: renal cell carcinoma, KIN17, PI3K AKT mTOR, targeted therapy, PF-04691502