Clear Sky Science · en
Protein content of extracellular vesicles from patients with advanced melanoma changes upon progression to anti-PD1 therapy
Why tiny messengers in blood may predict cancer treatment success
For people with advanced melanoma, powerful new immunotherapy drugs can sometimes make tumors melt away—but in many others, the cancer quickly pushes back. This study looks at microscopic “packages” that tumors release into the blood to see whether they can reveal, early on and without a biopsy, who is likely to benefit from anti-PD1 treatment and who may need a different strategy.
Small bubbles with big clues
Our blood carries countless nanosized bubbles called extracellular vesicles. These are tiny, membrane-wrapped packets that cells use to send molecular messages. Cancer cells also shed them, filling them with proteins that reflect what is happening inside the tumor and its surroundings. Because these vesicles circulate freely, they can be fished out of a simple blood sample instead of cutting into a tumor. The researchers wondered whether the protein cargo of these vesicles in patients with advanced melanoma could signal, in advance, how well a person would respond to anti-PD1 immunotherapy, a widely used drug class that “releases the brakes” on immune cells.
Following patients through treatment
The team studied nine people with advanced melanoma who all received anti-PD1 drugs. Blood samples were taken at three points: before treatment began, shortly after the first dose, and either when the disease progressed or nine months later if it had not. From each sample, the scientists isolated extracellular vesicles and used high-end mass spectrometry to catalog their protein content. After careful data filtering, they focused on 969 reliably measured proteins. They then grouped these proteins into networks representing major biological processes—such as immune activity, cell adhesion, blood clotting, and energy metabolism—and asked how these patterns differed between patients whose disease remained controlled and those whose tumors continued to grow.
Early signs of response hidden in protein patterns
Even before treatment started, the vesicles looked different in eventual responders versus non-responders. Patients who later fared better tended to have vesicles enriched in proteins linked to cell–cell contacts and certain protective responses to ultraviolet light, a key factor in skin cancer. In contrast, some immune-related proteins and inflammatory markers were more abundant in those who went on to progress, hinting that a “hot but misdirected” immune environment might blunt the benefit of therapy. By zeroing in on individual molecules, the researchers built an eight-protein signature—essentially a weighted combination of protein levels—that separated patients into high- and low-risk groups for earlier disease worsening. In this small group, this signature was able to distinguish these groups with promising accuracy.
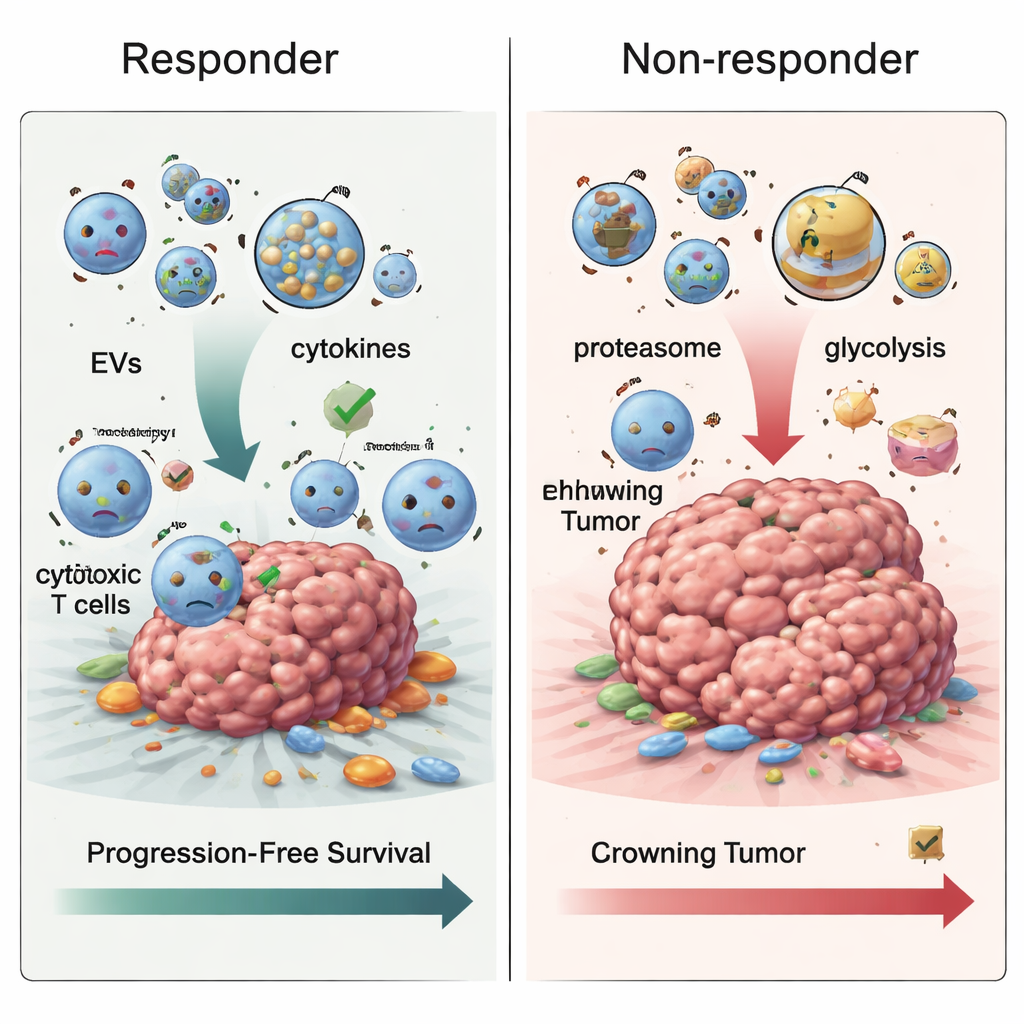
How resistance may develop over time
When the team examined samples taken after treatment had begun, another pattern emerged. In patients whose melanoma progressed, extracellular vesicles became enriched in proteins involved in the cell’s waste-disposal machinery, called the proteasome, and in energy-generating pathways such as glycolysis, the sugar-burning process many tumors rely on. These changes fit with the idea that resistant tumors rewire their metabolism and protein handling to survive immune attack. Tracking all three time points together, the researchers also saw that proteins tied to blood clotting tended to drop over time in non-responders, while proteins involved in presenting tumor fragments to the immune system went up—yet, paradoxically, this did not translate into better control of the cancer, suggesting that the way these signals are packaged into vesicles may actually hinder effective immune killing.
What this could mean for patients
In plain terms, the study suggests that tiny bubbles in the bloodstream carry a detailed molecular fingerprint of how melanoma tumors and the immune system are interacting with anti-PD1 therapy. Specific protein patterns in these vesicles—measured before and during treatment—could help doctors predict who is likely to enjoy longer control of their disease and who might be developing resistance driven by inflammation and altered metabolism. While the work is based on only nine patients and needs to be confirmed in larger groups, it points toward a future in which a routine blood draw could guide and adapt immunotherapy for melanoma, making these powerful drugs more precise and effective.
Citation: Trilla-Fuertes, L., Gámez-Pozo, A., Laso-García, F. et al. Protein content of extracellular vesicles from patients with advanced melanoma changes upon progression to anti-PD1 therapy. Sci Rep 16, 5891 (2026). https://doi.org/10.1038/s41598-026-35848-0
Keywords: melanoma immunotherapy, extracellular vesicles, blood biomarkers, anti-PD1 resistance, proteomics