Clear Sky Science · en
Ag-decorated Cu-doped ZnO nanomaterial for enhanced antibacterial application
Why tiny particles matter for big infections
Antibiotic-resistant infections are rising worldwide, and new drugs are slow and expensive to develop. This study explores a different tactic: using carefully engineered tiny particles made of zinc oxide mixed with copper and silver to physically and chemically attack bacteria, including hard-to-treat strains, even in the dark. By understanding how these particles are built and how they injure microbes, researchers hope to create coatings, dressings, and surfaces that quietly kill germs before they can cause serious disease.
Building better germ-fighting particles
The researchers set out to improve zinc oxide, a material already known to harm bacteria but mostly when exposed to light. They used a “bottom-up combustion” method, where metal salts and a common polymer are heated so they foam up, burn, and leave behind a rigid, highly porous network of tiny crystals. Into the zinc oxide framework they introduced copper and silver, creating a mixed material called a heterostructure, in which several metals and metal oxides touch each other very closely. 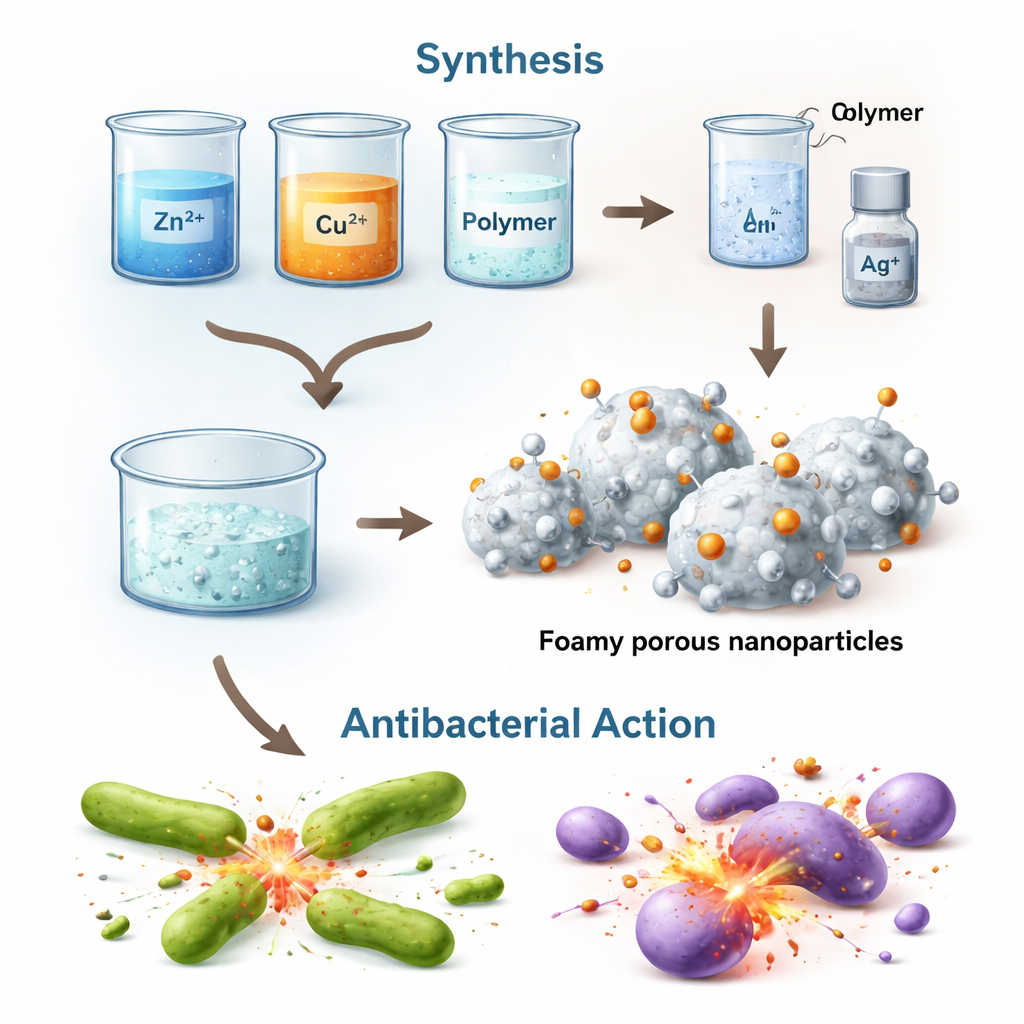
Peering inside the new material
To see what they had made, the team used a battery of structural and optical tests. X-ray measurements showed that copper atoms slipped into the zinc oxide crystal grid, slightly squeezing it, while silver mostly formed its own tiny crystals on the surface. High-resolution electron microscopy revealed these different components packed together within porous, foam-like structures. Light-based measurements confirmed that adding copper and silver narrowed the energy gap of zinc oxide and improved how charges move through the material. In practical terms, this means it can generate harmful, short-lived oxygen-based chemicals more easily and keep reactive charges from canceling each other out, both of which are useful for killing bacteria.
Turning structure into antibacterial power
The key test was whether these designer particles could actually stop bacteria from growing. The scientists compared plain zinc oxide with copper‑doped, silver‑decorated, and fully combined copper–silver–zinc oxide particles against both “Gram-positive” and “Gram-negative” bacteria, which differ in cell wall structure. They also studied versions made before and after an extra heating step called calcination. Plain zinc oxide worked modestly, mainly before final heating, but largely lost its effect afterward. In contrast, the fully combined material—containing zinc oxide, copper oxide, and silver—became more powerful after calcination, achieving an inhibition zone of up to 22 millimeters against Streptococcus pyogenes, a Gram-positive bacterium, at the highest tested dose. Overall, the new mixed particles outperformed single-metal particles, especially against Gram-positive strains.
How the particles attack bacteria in the dark
Unlike many light-activated materials, these particles were designed to work without illumination. The study proposes that the mixed metal particles kill bacteria through a multi-pronged attack. First, zinc, copper, and silver ions slowly dissolve from the particle surface and bind to bacterial membranes, enzymes, and DNA, disrupting vital processes and making the cell envelope leaky. Second, the close contact between the different metals helps generate reactive oxygen species—highly aggressive forms of oxygen—even in the dark. These species damage proteins, fats, and genetic material. Third, the porous, rough texture of the particles increases contact with bacterial cells and can physically injure their outer layers. Together, these effects overwhelm bacterial defenses and make it harder for resistance to develop. 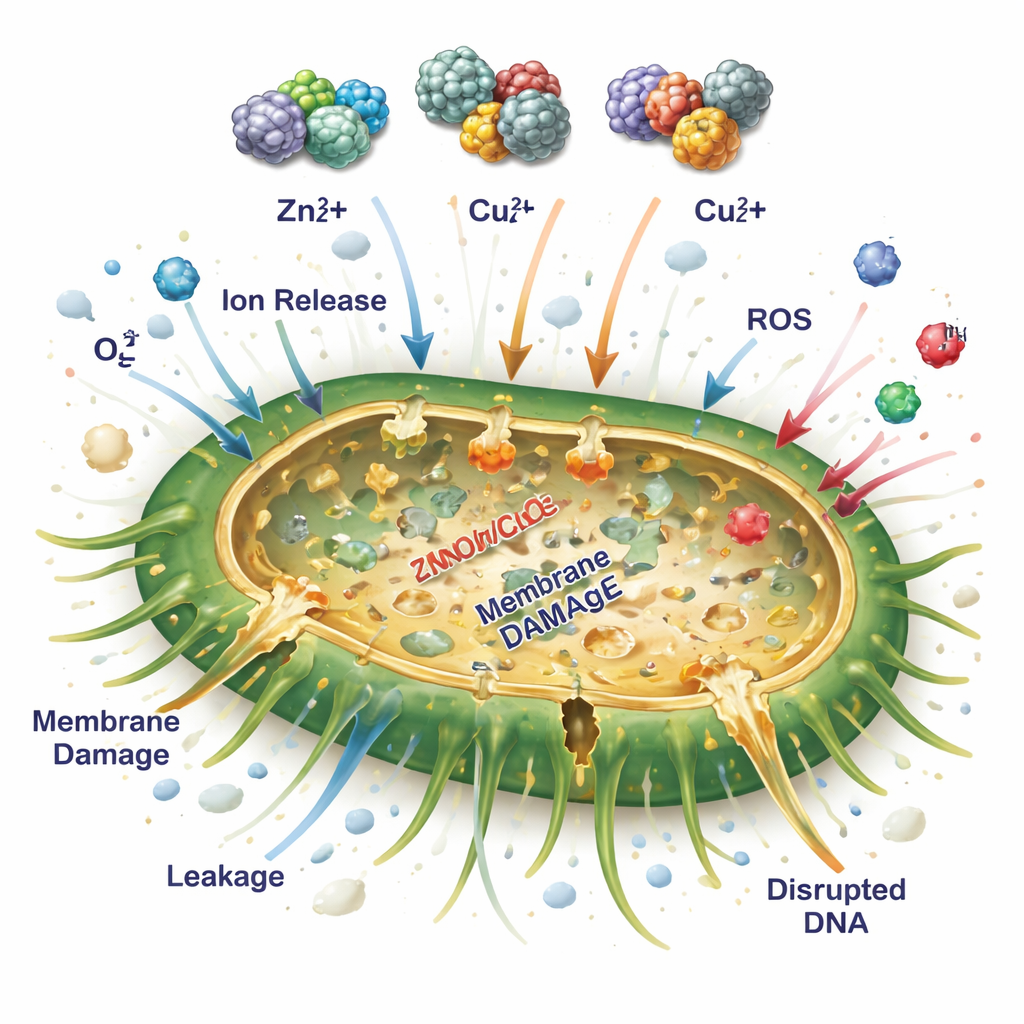
From lab dish to real-world protection
For non-specialists, the main message is that carefully combining familiar metals like zinc, copper, and silver into a single, well-structured nanoparticle can turn an ordinary ingredient into a potent, broad-spectrum antibacterial tool. The most effective material in this work stopped certain bacteria almost as well as a standard antibiotic, without relying on light exposure. Because these particles can be made as porous foams in a relatively simple process, they could eventually be scaled up for use in wound dressings, coatings for medical implants, or surfaces in hospitals that passively suppress bacterial growth. While more work is needed to confirm safety and performance in real tissues, this study shows a promising path toward physical–chemical antibacterials that complement, rather than replace, traditional antibiotics.
Citation: Gebretsadik, A., Reddy, S.G., Gonfa, B.A. et al. Ag-decorated Cu-doped ZnO nanomaterial for enhanced antibacterial application. Sci Rep 16, 5552 (2026). https://doi.org/10.1038/s41598-026-35838-2
Keywords: antibacterial nanomaterials, zinc oxide nanoparticles, copper and silver doping, antibiotic resistance, heterostructured nanocomposites