Clear Sky Science · en
Sickle cell disease detection in low-resource conditions using transfer-learning and contrastive-learning coupled with XAI
Why smarter blood tests matter
Sickle cell disease is a lifelong blood disorder that can cause severe pain, infections, and early death, especially in parts of Africa and India where medical resources are scarce. Early diagnosis can save lives, but traditional tests require skilled staff, specialized machines, and time that many clinics simply do not have. This paper explores how artificial intelligence (AI) can turn simple microscope images of blood into fast, reliable screening tools, making it easier to spot sickle cell disease even in low-resource settings.
A closer look at a dangerous blood disorder
In sickle cell disease, a small change in the gene for hemoglobin—the protein that carries oxygen—causes red blood cells to bend into stiff, crescent or “sickle” shapes instead of staying soft and round. These misshapen cells can clog tiny blood vessels, blocking blood flow and damaging organs. 
From overloaded labs to AI helpers
Traditional ways to confirm sickle cell disease—such as hemoglobin electrophoresis or genetic testing—are accurate but often expensive, equipment-heavy, and slow. In contrast, many clinics already have basic microscopes, and modern cameras can capture high-resolution images of blood smears. The authors build on this reality: instead of changing how blood is collected, they change how images are analyzed. They feed digitized blood-smear images into AI models that have already learned to recognize patterns from millions of everyday photographs, then fine-tune these models to tell normal red blood cells from sickled ones. This reuse of prior knowledge, called transfer learning, is crucial when only a few hundred medical images are available for training.
Teaching machines to tell subtle shapes apart
Not all AI training methods are equal, especially when data are limited. The researchers compare three popular image-recognition networks—ResNet-50, DenseNet-121, and EfficientNet-B0—and three ways of training them. Two of the training methods treat the problem as a simple yes-or-no question (sickle or not) and try to improve the model’s raw classification accuracy. The third, called triplet loss, instead teaches the network to arrange images in a “shape space” where images of sickle cells cluster together and pull away from images of normal cells. This contrast-focused training turns the model into a specialist in spotting small, shape-based differences, which is exactly what matters in sickle cell microscopy.
Making the decision process visible
Doctors and lab workers need to trust any automated system that influences patient care. To open the AI “black box,” the authors use an explainable AI method called Grad-CAM, which overlays a heatmap on the original microscope image to show which regions influenced the decision most. 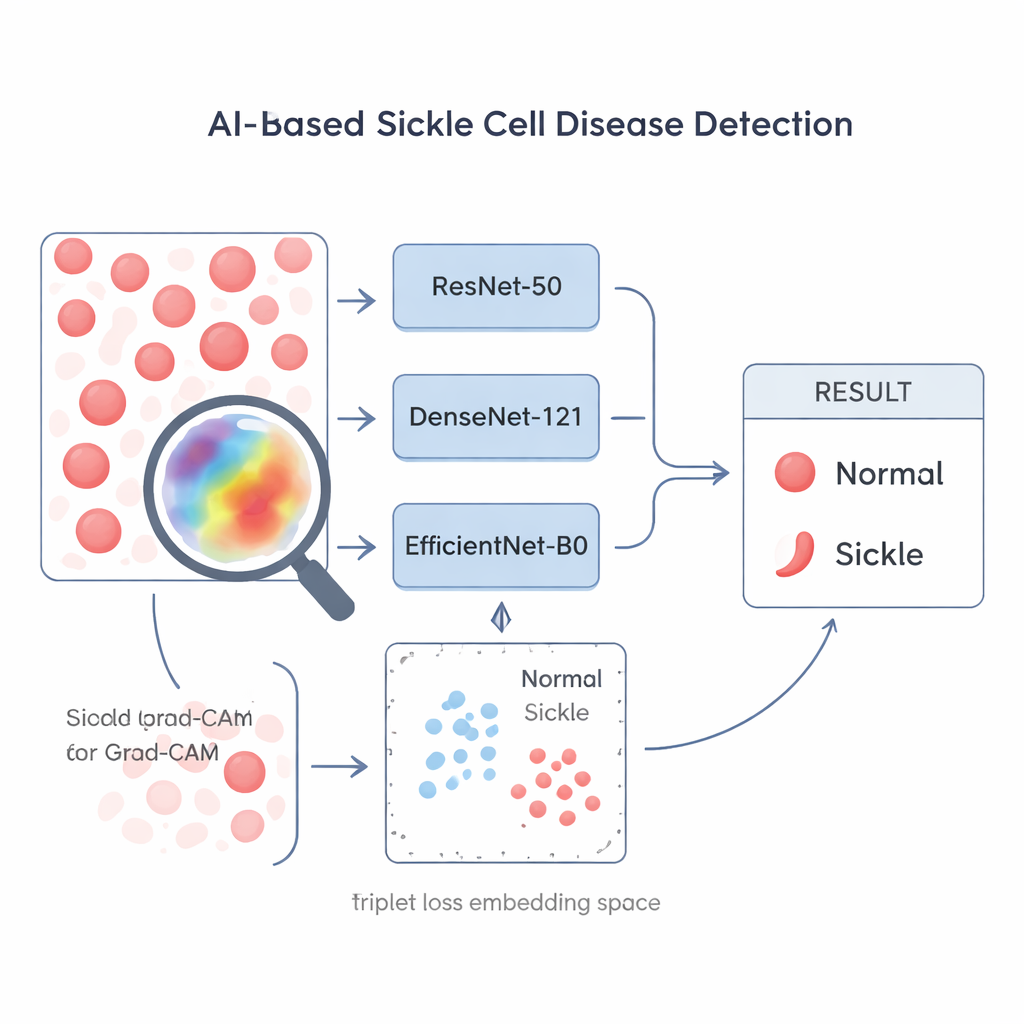
From research to real-world clinics
The study concludes that smart reuse of existing image-recognition networks, combined with triplet-loss training and visual explanations, can deliver accurate and transparent sickle cell detection from relatively few images. In plain terms, a standard microscope plus a camera and laptop could help front-line clinics rapidly flag patients who likely have sickle cell disease, even without advanced lab tests. While larger and more diverse datasets will still be needed before such tools are widely deployed, this work shows a clear path toward low-cost, AI-assisted screening that could make a real difference in regions where sickle cell disease is common but diagnostic resources are limited.
Citation: Patel, J., Muralikrishna, H., Chadaga, K. et al. Sickle cell disease detection in low-resource conditions using transfer-learning and contrastive-learning coupled with XAI. Sci Rep 16, 6104 (2026). https://doi.org/10.1038/s41598-026-35831-9
Keywords: sickle cell disease, medical imaging, deep learning, low-resource diagnostics, explainable AI