Clear Sky Science · en
Synergistic effects of hard anodizing parameters on the microstructural, mechanical, and tribological properties of 6061 aluminum alloy
Making Everyday Metals Last Longer
From airplanes and cars to laptops and window frames, aluminum alloys are everywhere because they are strong yet light. But there is a catch: bare aluminum surfaces can wear down and scratch more easily than we would like, especially in harsh or high‑friction environments. This study explores how to turn a common aluminum alloy, known as 6061, into a tougher, longer‑lasting material by carefully growing a very hard, ceramic‑like skin on its surface.
Growing a Protective Skin on Aluminum
The researchers focused on a process called hard anodizing, in which an aluminum part is placed in an acid bath and used as an electrical anode so that a thick oxide layer grows on its surface. Unlike the thin, naturally formed oxide that appears in air, this engineered layer can be much thicker and harder. The team systematically adjusted four key knobs on the process—acid concentration, bath temperature, electrical current density, and treatment time—to see how they work together. Their goal was to find a recipe that makes the protective skin as thick, hard, and wear‑resistant as possible without damaging it.
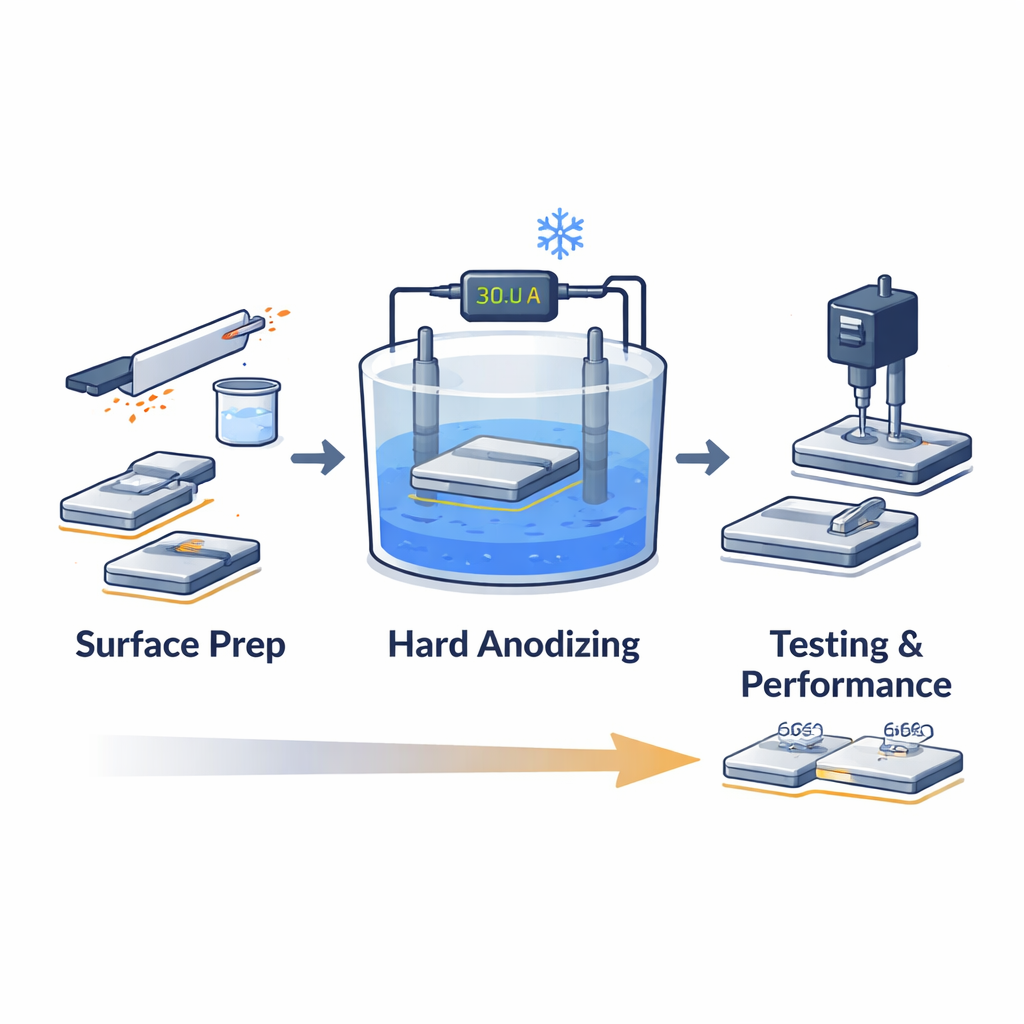
Finding the Sweet Spot in Processing Conditions
Surprisingly, simply “more” or “less” of any one setting did not always give better results. When the sulfuric acid solution was too weak, the oxide grew slowly and the protective layer stayed thin. When it was too strong, the aggressive liquid started to dissolve the very film it had just helped form. A similar balancing act appeared with temperature: cooling the bath from 10 °C down to just below freezing (−2 °C) produced a thicker, denser film, because the cold slowed chemical attack. But going even colder made the liquid less conductive, so the electrical reactions that build the film stalled and the coating quality dropped. The best combination of thickness and hardness occurred at a moderate acid concentration (about 190 g per liter) and an electrolyte temperature of −2 °C.
Electricity, Time, and Hidden Heat
The strength of the electric current and how long it was applied also played a crucial role. Higher current and longer times generally made the oxide layer thicker, because more aluminum and oxygen ions were driven to react. Up to a point, this also increased hardness: the film became denser, with a fine internal structure and good adhesion to the metal underneath. However, as the coating thickened it resisted the flow of current, which caused extra heating at the interface. That hidden heat began to eat away at the internal walls of the film, coarsening its structure and reducing hardness. The best trade‑off was found at a relatively high current density for one hour, which produced a coating about 59 micrometers thick—roughly the width of a human hair—and close to six times harder than the bare 6061 aluminum.
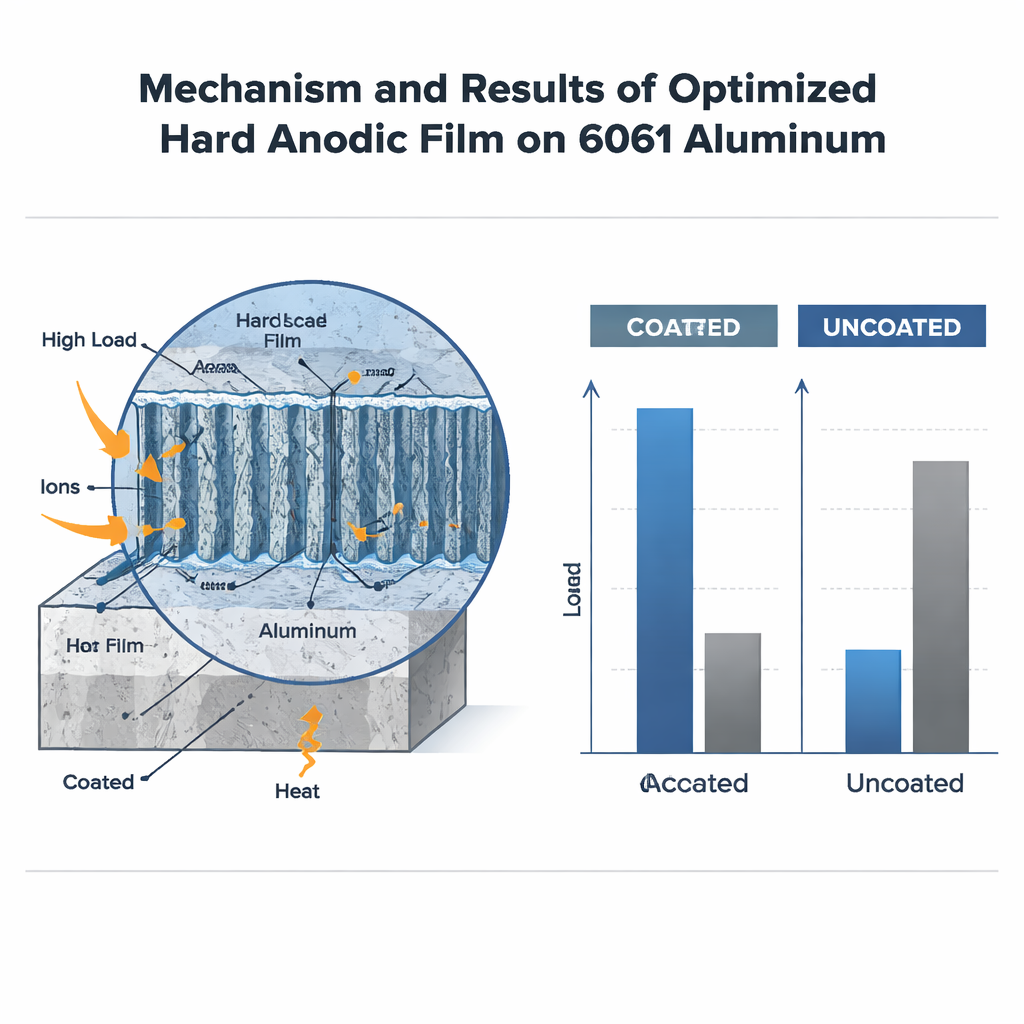
From Sticky Wear to Gentle Sliding
To see whether this hard skin actually protects components in motion, the team rubbed coated and uncoated samples against a tungsten carbide pin under different loads. The untreated aluminum suffered heavy damage: its soft surface stuck, tore, and deformed, and it lost much more material. In contrast, the hard‑anodized samples showed far less mass loss and a smoother, more stable friction behavior. At low and medium loads, the coating turned severe “grabby” wear into mild abrasion, where tiny hard bumps simply scratch the surface lightly. At the highest load, the brittle ceramic layer began to crack and chip, and the broken fragments acted like abrasive grits, increasing wear—but even then, the coated alloy still outperformed the bare metal.
What This Means for Real‑World Parts
In plain terms, the study shows that 6061 aluminum can be given an armor‑like shell if hard anodizing is tuned with care. The right combination of acid strength, cold temperature, current, and time creates a dense, uniform oxide layer that is much harder than the original metal and dramatically slows wear and friction. For designers of aircraft parts, automotive components, or consumer products where lightness matters, this optimized process offers a practical way to extend service life without switching to heavier materials. The key message is that surface engineering—getting the process details right—can unlock far better durability from an already familiar alloy.
Citation: Behzadifar, J., Najafi, Y. & Nazarizade, B. Synergistic effects of hard anodizing parameters on the microstructural, mechanical, and tribological properties of 6061 aluminum alloy. Sci Rep 16, 5021 (2026). https://doi.org/10.1038/s41598-026-35825-7
Keywords: hard anodizing, 6061 aluminum, surface coating, wear resistance, tribology