Clear Sky Science · en
Mechano-stress endorsing heterogeneous lung cancer cells migration into confined channels and investigating tumor spheroids growth of confined space migrating cells
Why squeezing cancer cells matters
Cancer does not spread in open space. As tumor cells leave their original site, they must squeeze through tiny gaps in tissues, blood vessel walls, and other crowded structures. This study asks a simple but powerful question: within a mixed crowd of lung cancer cells, which ones actually make it through these tight spots, and what kind of tumors do they later build? The answers may help explain why some cancer cells are more dangerous than others and could guide new ways to predict or slow metastasis.
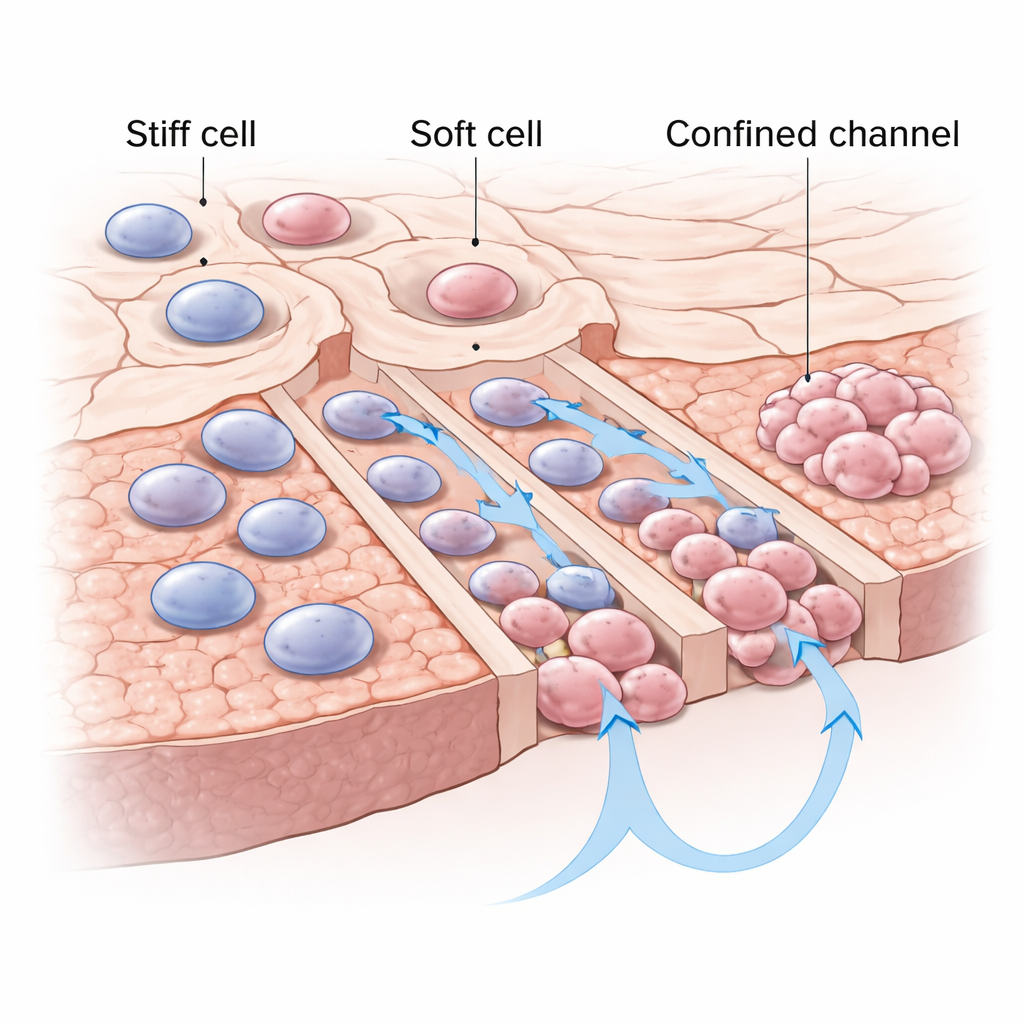
Softer cells win in tight spaces
The authors focused on a physical trait of cells that most people never hear about: stiffness. Some cancer cells are relatively rigid, while others are soft and easily deformed. Using specially designed microfluidic chips—tiny plastic devices etched with channels narrower than a typical cell—they trapped individual lung cancer cells at the mouths of narrow (confined) and tall (unconfined) channels. After 24 hours, more single cells chose and successfully traversed the narrow, confined channels than the more open ones, and they moved faster inside these tight passages. When the researchers later probed the cells’ mechanics with an atomic-force microscope (a kind of nanoscale “finger”), they found that the travelers in confined channels were consistently softer than their counterparts that stayed behind or moved in unconfined channels.
A molecular signature of softness
To understand what made some cells softer, the team examined a structural protein called vimentin and a nuclear support protein encoded by the LMNA gene. Vimentin is part of the internal scaffold that helps a cell resist deformation. Cells that had squeezed through confined channels showed lower levels of vimentin compared with cells in roomy channels or in standard dishes. High-resolution imaging revealed that, in confined migrants, the vimentin network was more loosely arranged both around the cell body and near the nucleus, rather than densely bundled. Gene-expression tests at the single-cell level confirmed that both the vimentin gene (VIM) and LMNA, which helps keep the nucleus mechanically robust, were dialed down in cells that preferred confined routes.
Turning cells softer boosts confined migration
The researchers then asked whether they could intentionally soften cells and see if that changed their behavior. They used two approaches: a biochemical treatment (TGF-β1) known to remodel the cell skeleton, and gentle mechanical compression using soft silicone slabs pressing on the cells. Both methods reduced stiffness and lowered vimentin levels without killing the cells. When these pre-softened cells were introduced into the microfluidic device, an even larger fraction migrated into the tight channels compared with untreated cells. This suggests that softness is not just a side effect of confinement; it is a property that can be tuned and that strongly influences whether a cell can enter and traverse small openings.
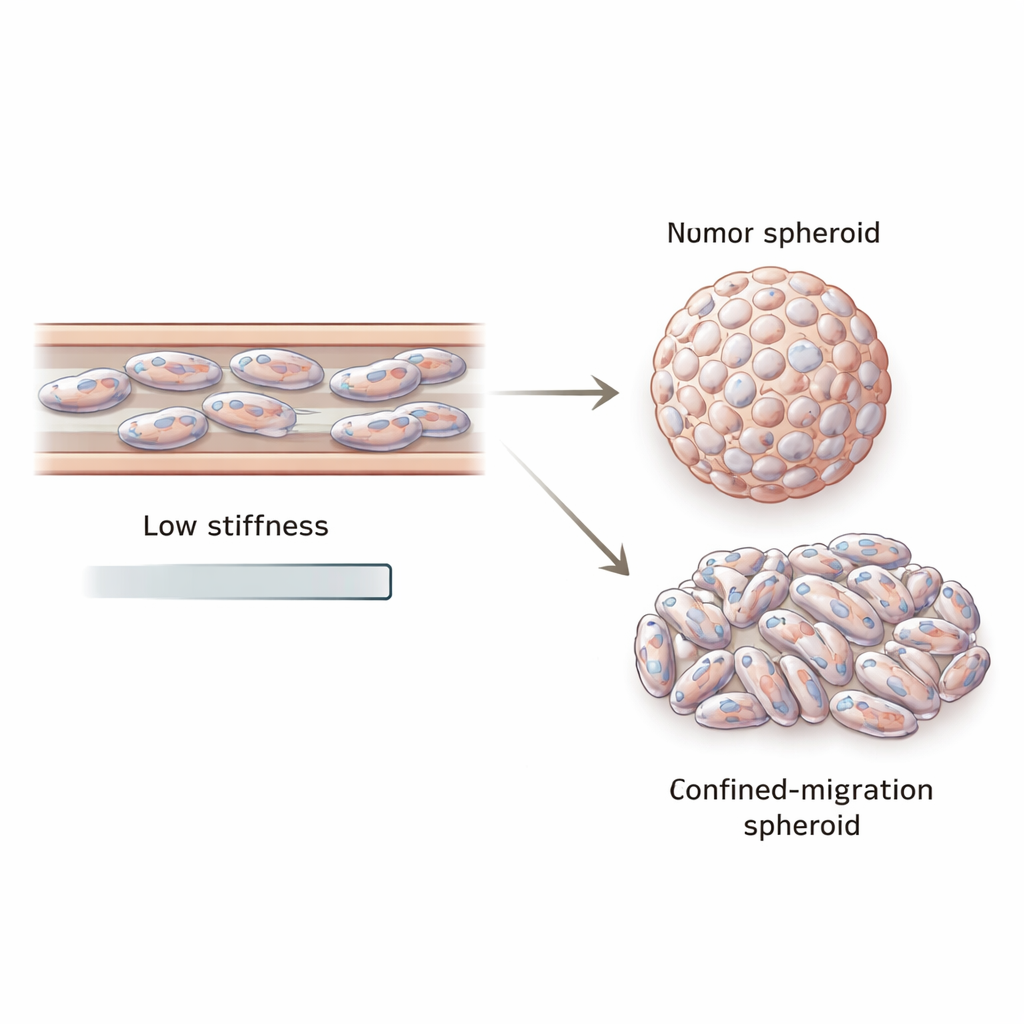
From squeezed cells to misshapen tumor balls
Metastasis, however, is not only about movement. Escaped cells must also regrow into new tumors. To mimic this step, the team used a second confined system called a trans-well, where cells migrate through tiny pores before being collected and grown in non-stick wells to form three-dimensional tumor “spheroids.” Spheroids grown from confined-migration cells were smaller and noticeably less round than spheroids grown from the same cell lines without prior squeezing. Their cell nuclei were more elongated and deformed, and these spheroids continued to show reduced VIM and LMNA expression. Even within individual spheroids, vimentin levels varied from center to edge, hinting at mechanical and genetic diversity in the daughter tumor.
What this means for understanding metastasis
Put plainly, this study shows that among many lung cancer cells, the softer ones are more likely to slip through tight spaces, survive the journey, and later build oddly shaped, mechanically fragile tumor clusters with distorted nuclei. Those cells carry and maintain a distinct molecular signature—low levels of key structural genes—that links how they move to how they grow. While the work was done in carefully controlled lab devices rather than in patients, it offers a physical and genetic blueprint of a “confined-migrating” cancer cell. In the long run, measuring or targeting cell softness and its molecular markers could become part of strategies to understand, track, or eventually disrupt the most invasive cancer cell subpopulations.
Citation: Alam, M.K., Ma, Y., Zhai, J. et al. Mechano-stress endorsing heterogeneous lung cancer cells migration into confined channels and investigating tumor spheroids growth of confined space migrating cells. Sci Rep 16, 6649 (2026). https://doi.org/10.1038/s41598-026-35818-6
Keywords: cancer cell stiffness, confined cell migration, lung cancer spheroids, vimentin and LMNA, tumor mechanics