Clear Sky Science · en
Development of robust dual functioning PPy-based photothermal membranes for simultaneous freshwater and salt harvesting
Turning Sunlight into Drinking Water and Useful Salt
Billions of people live in regions where clean water is scarce, even as oceans and salty wastewaters are all around us. This study explores a new kind of fabric-based membrane that uses only sunlight to make fresh water from salty or polluted sources, while also recovering valuable salts instead of throwing them away. The approach aims to cut energy use, lower costs, and reduce liquid waste, offering a practical route toward cleaner water and smarter resource use.
A Simple Fabric that Drinks in Sunlight
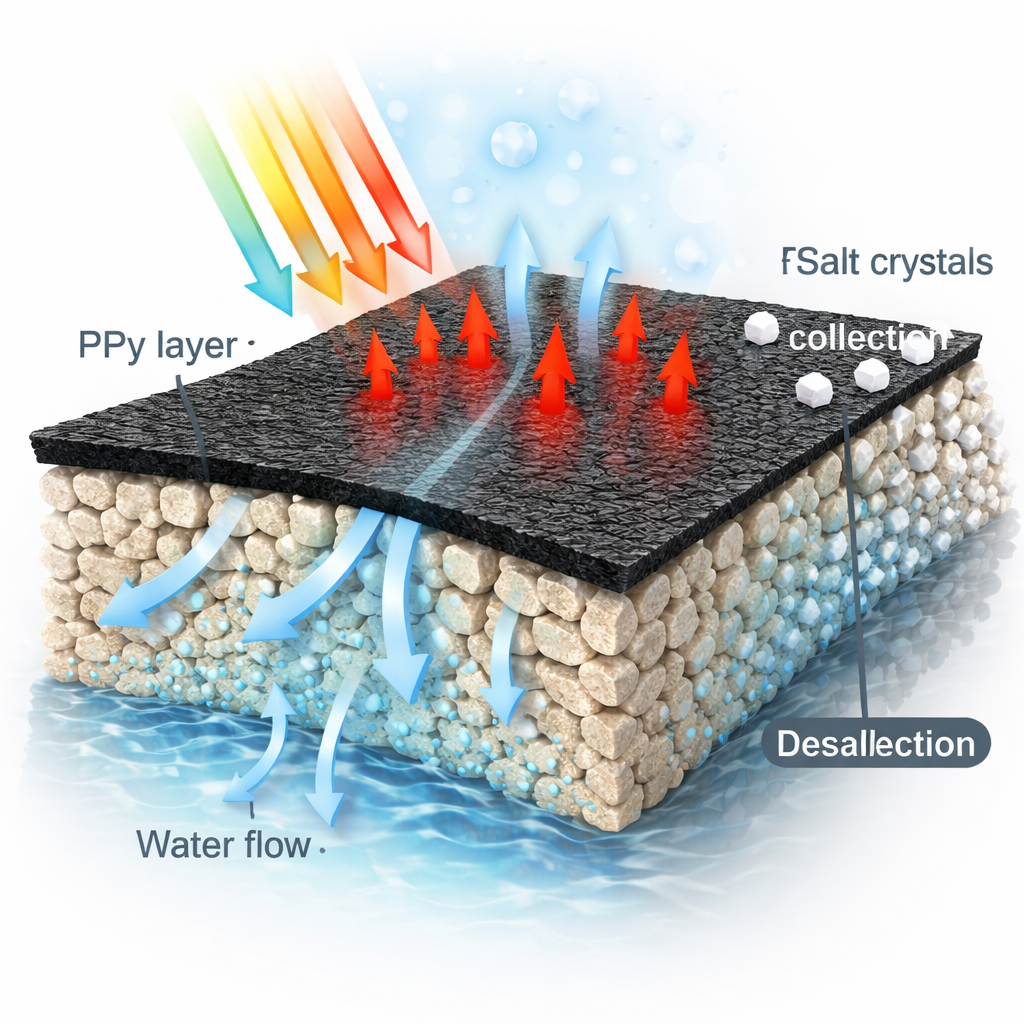
The heart of the work is a thin, dark coating made from a conductive plastic called polypyrrole (PPy), placed on common polyester fabrics. When sunlight hits PPy, it absorbs light strongly across a wide range of colors and turns it into heat very efficiently. The researchers used a solvent-free method called chemical vapor deposition polymerization to form a uniform PPy layer on either woven or nonwoven fabric. Cleverly, only the top side of the fabric is coated, leaving the bottom side hydrophilic so it can pull up water from below while the dark upper side faces the sun and heats up. This design keeps water flowing steadily to the hot surface while minimizing material and chemical use.
Tuning the Recipe for Maximum Heating
To build the PPy layer, the team tested several oxidizing agents—chemicals that trigger the polymer to form—including ferric chloride, copper chloride, ammonium persulfate, potassium permanganate, and sodium dichromate. By varying their concentration and the tiny amount of pyrrole monomer used, they found conditions that created a continuous, jet-black PPy skin on the fibers. Microscopy showed that the coated fibers developed a rough, finely textured surface, which reduces light reflection and helps trap more solar energy. Light measurements confirmed that the best membranes absorbed more than 94% of incoming light from ultraviolet through infrared, far higher than the bare fabrics. Under one-sun illumination (the strength of normal noon sunlight), these optimized membranes quickly heated to about 60–65 °C, much hotter than the uncoated fabric.
Boosting Evaporation with a Thin Hot Skin
When these PPy-coated fabrics were floated on water and exposed to simulated sunlight, they significantly increased the rate at which water evaporated. Plain water without a membrane evaporated slowly, at about 0.22 kilograms per square meter per hour. Adding bare fabric already tripled this rate, but the PPy coating pushed it higher still: up to 0.95 kg m−2 h−1 for nonwoven fabric treated with copper chloride and 0.93 kg m−2 h−1 for woven fabric treated with ammonium persulfate. Even though only very small amounts of pyrrole were used, the membranes achieved a solar-to-heat conversion efficiency of about 57%. Under stronger, three-sun illumination—similar to concentrated sunlight—the best membrane drove evaporation as high as 2.91 kg m−2 h−1 while remaining stable over repeated heating cycles.
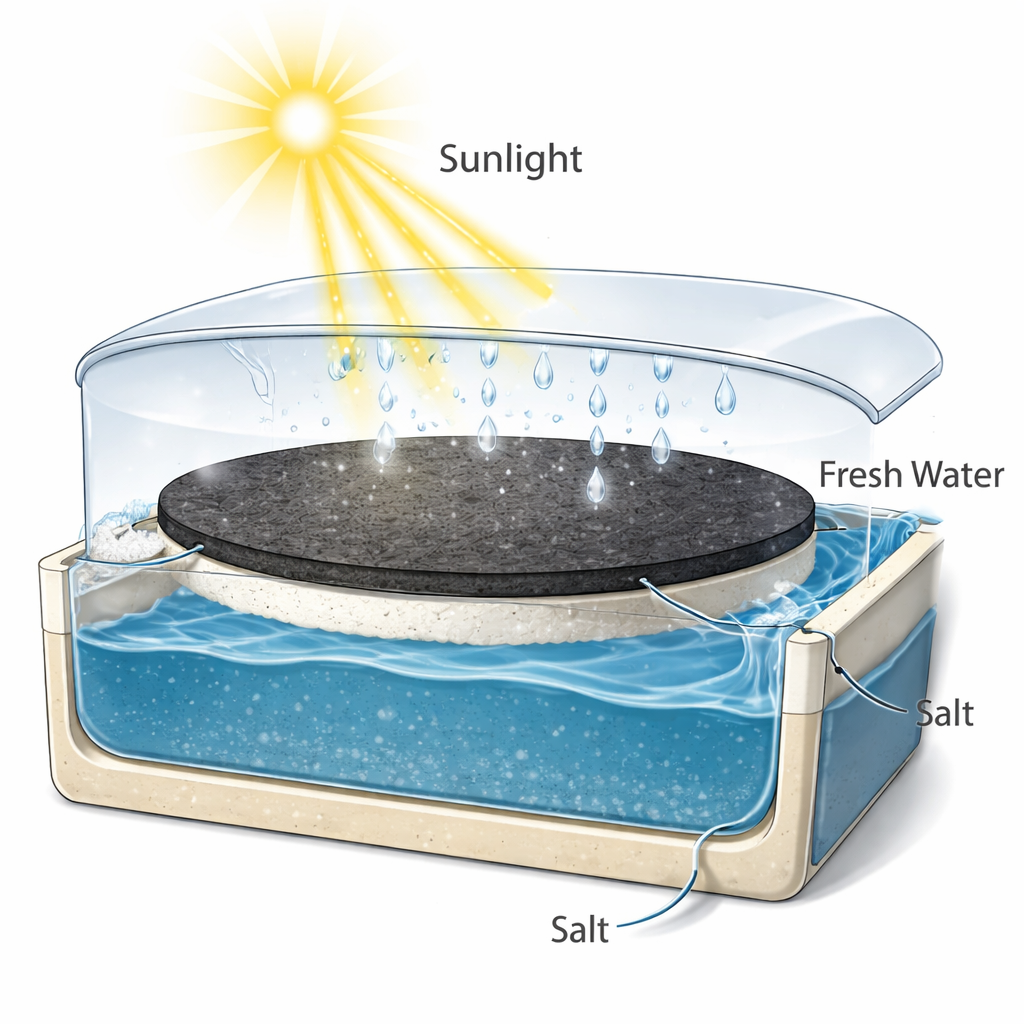
Freshwater on Top, Crystals at the Edge
Beyond making clean water, these membranes are designed to help recover the salts that would otherwise become waste. As the surface heats and water turns to vapor, the rising steam is collected and condensed as nearly pure water, while the remaining solution becomes more concentrated. Because the PPy surface is hydrophobic and rough, salt crystals preferentially form at the edges of the evaporation zone rather than clogging the central hot area. In tests with sodium chloride, copper sulfate, and iron chloride at realistic seawater and brine concentrations, the system maintained high evaporation rates and produced visible rings of salt that could be easily harvested. In one demonstration, a membrane operating on a 7% salt solution recovered 100% of the dissolved salt, at a salt collection rate of about 58.6 grams per square meter per hour, all while continuing to generate freshwater.
Toward Cleaner Water with No Liquid Left Behind
In everyday terms, this research shows that a low-cost, cloth-like material can turn sunlight into both drinkable water and reusable salt, without needing high-pressure pumps, complex electronics, or large amounts of chemicals. The PPy-coated membranes are robust, washable, and compatible with real-world brines and wastewaters, including acidic or alkaline streams. Because they can concentrate salty waste until virtually no liquid is left, they support the ambitious goal of "zero liquid discharge," where water is recycled and solids are recovered instead of dumped. With further engineering and scale-up, such sun-driven membranes could play a key role in small, decentralized systems that help coastal communities, farms, and industries secure fresh water while cutting pollution and waste.
Citation: Mahmoud, M.T., Abdel-Ghafar, H.M., El-Sherif, A.A. et al. Development of robust dual functioning PPy-based photothermal membranes for simultaneous freshwater and salt harvesting. Sci Rep 16, 5945 (2026). https://doi.org/10.1038/s41598-026-35812-y
Keywords: solar desalination, photothermal membrane, freshwater harvesting, salt recovery, zero liquid discharge