Clear Sky Science · en
Metabolomic-driven prediction of the mutational status of healthy individuals with a family history of hereditary breast and ovarian cancer syndrome: the HRRmet study
Why Hidden Clues in Blood Matter
Many people know breast and ovarian cancer can run in families, often because of inherited changes in genes like BRCA1 and BRCA2. Today, genetic testing can reveal who carries these risky mutations—but it is expensive, not always available, and some people are reluctant to take a DNA test. This study asks a bold question: could a simple blood test that measures small molecules made by our bodies hint at whether a healthy person carries such inherited cancer-related mutations, long before any tumor appears?
Tiny Molecules as Early Messengers
Our cells constantly produce and break down thousands of small chemicals, known as metabolites, as part of everyday metabolism. Together, these chemicals form a “metabolomic” fingerprint that reflects both our genes and our environment. The researchers reasoned that if certain DNA repair genes are faulty—specifically those involved in a process called homologous recombination repair, which fixes dangerous breaks in DNA—then subtle shifts in metabolism might appear in the blood. Detecting those shifts could offer a new way to flag people who silently carry harmful variants in genes such as BRCA1, BRCA2, PALB2, ATM, CHEK2, and RAD51.
Studying Healthy Relatives at Risk
The team enrolled 260 healthy volunteers from families with hereditary breast and ovarian cancer. Half of them were known carriers of pathogenic variants in one of the DNA repair genes, while the other half were closely matched relatives who did not carry such variants. Crucially, none of the participants had cancer, so any differences in blood chemistry would be linked to inherited genetic status rather than to a tumor. Using a sensitive technique that couples liquid chromatography with high-resolution mass spectrometry, the scientists measured 285 metabolites in plasma and subjected 169 high-quality signals to detailed analysis.
Patterns that Separate Carriers from Noncarriers
Initial statistical tests showed that dozens of metabolites differed between carriers and noncarriers in at least one comparison. The strongest signals often came from lipids, especially fat-related molecules such as lysophosphatidylcholines and carnitine derivatives, and from certain amino acids like proline, tyrosine, and valine. These molecules are already known to be involved in how cancer cells rewire their energy use, suggesting that the metabolic “groundwork” for cancer-like behavior may be detectable even in people who are currently healthy. When the researchers built a single, broad model using 31 metabolites to distinguish all carriers from all noncarriers, it correctly classified about 62 percent of individuals—better than chance but not yet suitable as a stand‑alone test.
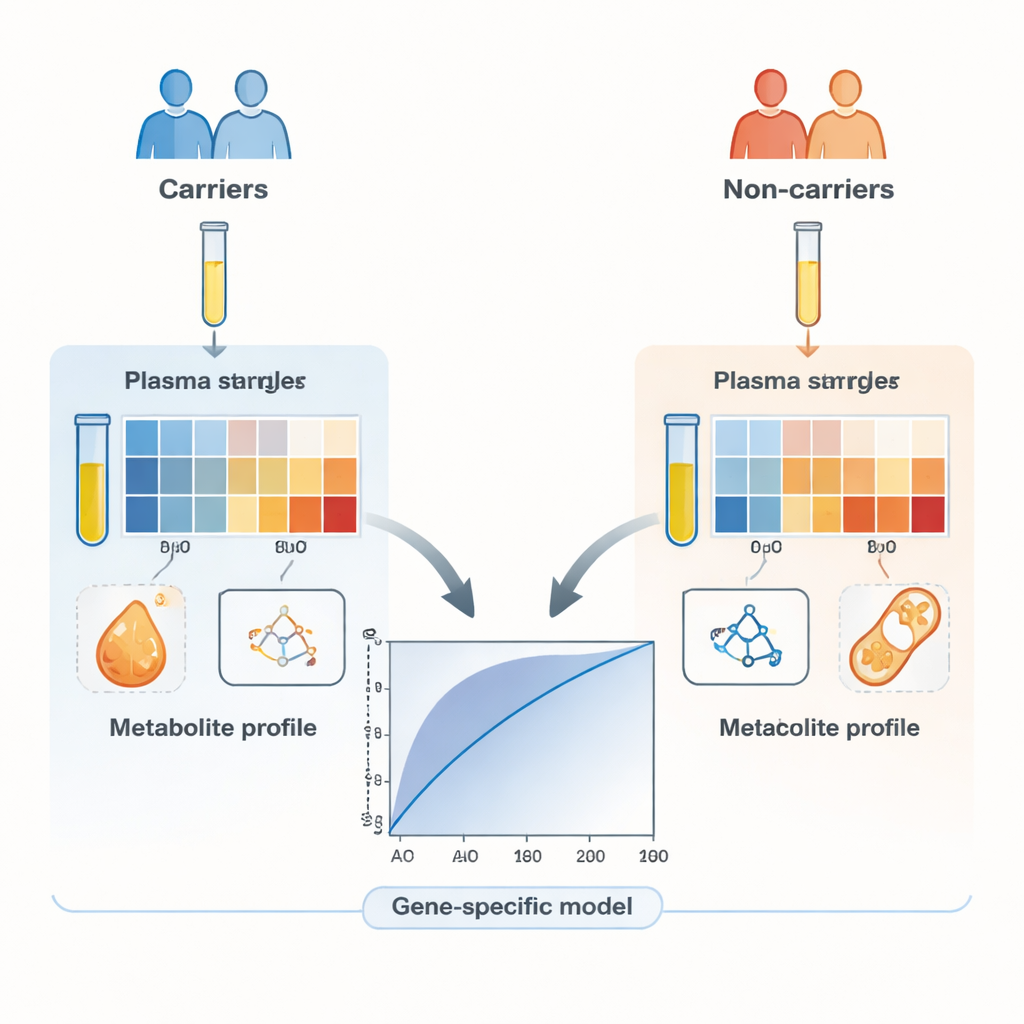
Sharper Signals When Focusing on Individual Genes
The picture improved dramatically when the team built separate prediction models for specific genes. By letting a machine-learning algorithm choose the most informative subset of metabolites for each gene, they produced compact signatures—sometimes just a handful of molecules—that identified carriers of BRCA1, BRCA2, PALB2, ATM, or CHEK2 with more than 80 percent accuracy, and often much higher. Models built around genes with so‑called moderate risk, such as ATM and CHEK2, performed particularly well despite using relatively few metabolites. Heatmaps of the data showed that results grouped more clearly by the type of metabolite (for example, lipids versus amino acids) than by which gene was mutated, underscoring that different gene defects can disturb overlapping metabolic pathways.
What This Could Mean for Families
In plain terms, this study shows that the blood chemistry of healthy people with inherited mutations linked to breast and ovarian cancer is measurably different from that of their noncarrier relatives. Those differences are subtle and invisible to standard blood tests, but they emerge when hundreds of metabolites are examined together with advanced statistical tools. While this approach is not ready to replace genetic testing, it points toward a future in which a metabolomic blood test could help flag high‑risk individuals, refine screening strategies, or prioritize who should receive full DNA testing. For families living with hereditary cancer, such tools could offer an extra, noninvasive window into inherited risk long before disease develops.
Citation: Roig, B., Fernández-Castillejo, S., Gumà, J. et al. Metabolomic-driven prediction of the mutational status of healthy individuals with a family history of hereditary breast and ovarian cancer syndrome: the HRRmet study. Sci Rep 16, 6844 (2026). https://doi.org/10.1038/s41598-026-35789-8
Keywords: hereditary breast cancer, ovarian cancer risk, metabolomics, genetic mutations, blood biomarkers