Clear Sky Science · en
Preliminary exploration of radiomic mammographic analysis in triple negative breast cancer related to BRCA profile
Why breast scans can say more than meets the eye
When most people think of a mammogram, they picture a doctor looking for obvious lumps or suspicious spots. This study asks a deeper question: could the subtle patterns in a standard breast X‑ray also reveal whether a woman carries high‑risk hereditary mutations, such as in the BRCA genes, even before a pathologist or genetic test weighs in? If so, the everyday images used to detect cancer might also help flag women at greater inherited risk, guiding closer follow‑up or genetic counseling.
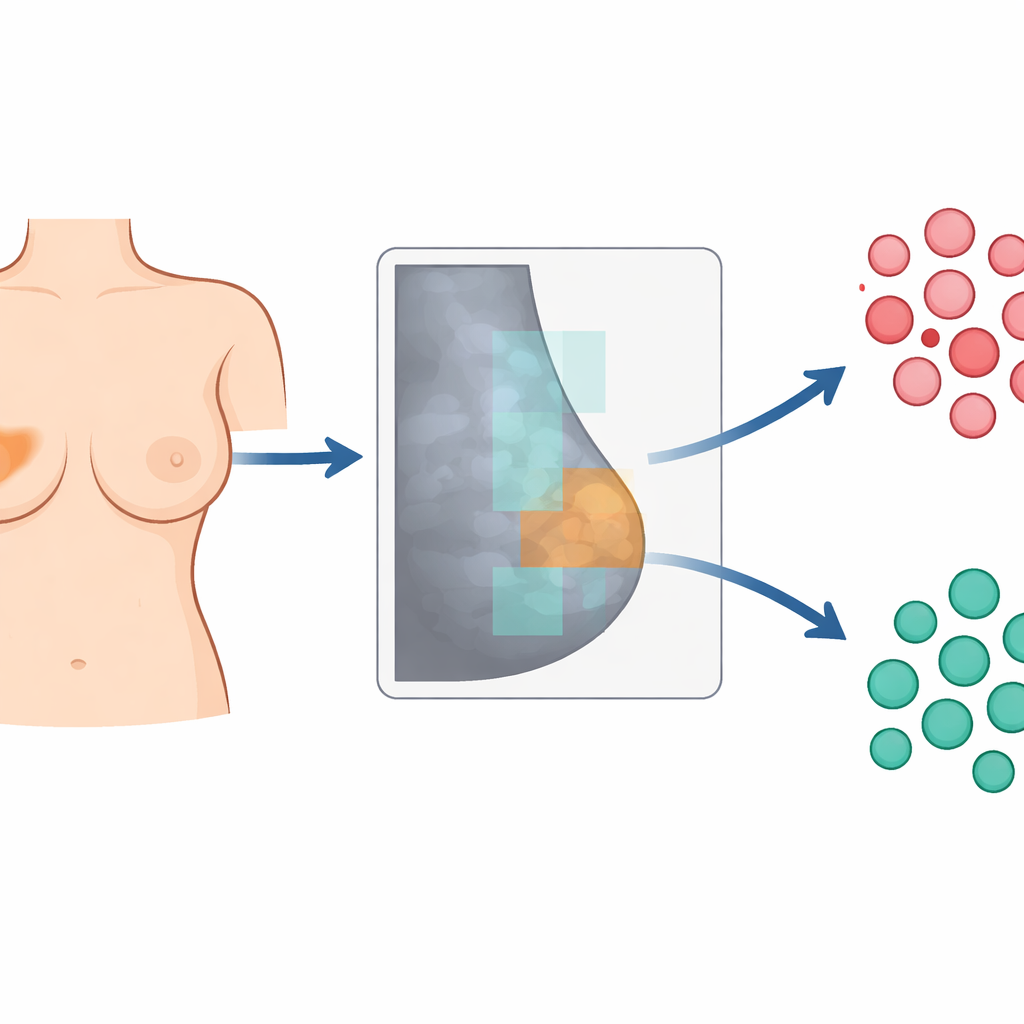
A tough kind of breast cancer and inherited risk
The researchers focused on triple‑negative breast cancer, a particularly aggressive form that lacks three common hormone and growth‑factor markers and currently has fewer tailored treatment options. Triple‑negative tumors are more common in women who carry harmful changes in the BRCA1 or BRCA2 genes, which are involved in repairing damaged DNA. Yet today, confirming those mutations still requires genetic testing. The team wondered whether mammograms themselves might contain hidden clues—too subtle for the human eye—that distinguish women with and without BRCA mutations among those already diagnosed with triple‑negative disease.
Turning mammograms into numbers
To explore this, the scientists carried out a retrospective review of 52 women with triple‑negative breast cancer who had undergone digital mammography before any treatment and had known BRCA status. Thirteen women carried BRCA mutations and 39 did not. Radiologists manually outlined each visible tumor on the mammogram and also drew a standard‑sized oval region in the most uniform area of the opposite, apparently healthy breast. Using an open‑source software package, they converted each outlined area into 195 numerical descriptors, or “features,” that capture brightness, contrast and fine‑scale texture—patterns of light and dark that hint at how tissue is organized on a microscopic level.
Letting algorithms hunt for meaningful patterns
Because hundreds of measurements can be redundant, the team used statistical tools to whittle the list down to a few features that were most informative yet not strongly overlapping. They then built three types of models: one based only on features from the healthy breast tissue, one using only the tumor features and one combining both. Several off‑the‑shelf machine‑learning classifiers—including logistic regression, support vector machines and decision trees—were trained and tested repeatedly on shuffled subsets of the data to estimate how well they could tell BRCA‑mutated from non‑mutated patients.
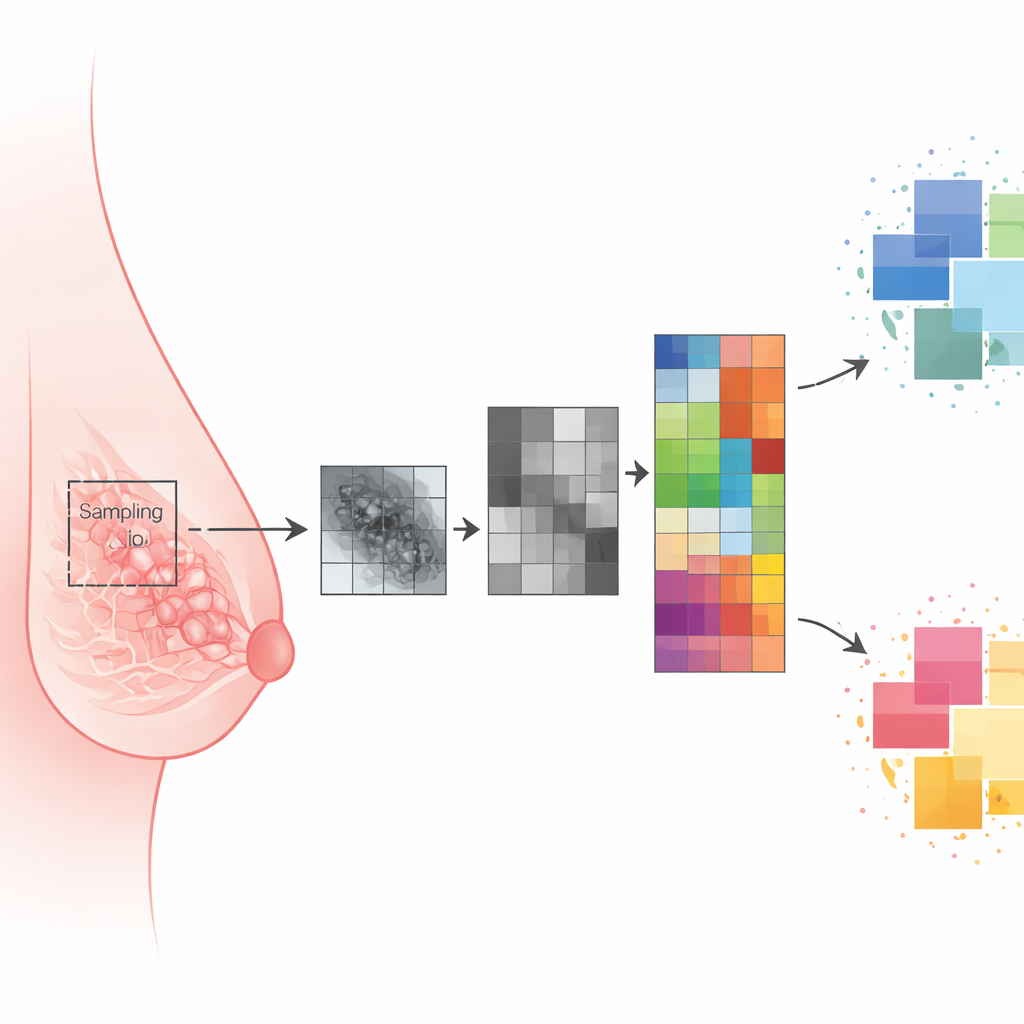
The quiet breast tissue speaks the loudest
Surprisingly, the most accurate models did not come from the tumors themselves but from the normal‑appearing glandular tissue in the opposite breast. A simple linear model based solely on three features from this healthy tissue achieved good discrimination between mutation carriers and non‑carriers, with high specificity—meaning few false alarms. One texture measure, known as “sum entropy,” which reflects how random or complex the pixel patterns are, was consistently higher in women with BRCA mutations. The authors suggest that inherited defects in DNA repair may subtly alter the microscopic architecture of breast tissue long before or beyond what can be seen as a distinct mass, and that this altered architecture shows up as a more irregular texture in the mammogram.
What this could mean for future checkups
For a layperson, the key takeaway is that standard mammograms may hold much richer information than radiologists currently use. In this preliminary study, computer analysis of the “background” breast tissue—not just the tumor—helped distinguish women with high‑risk BRCA mutations from those without, within a group already facing triple‑negative breast cancer. If larger, multi‑center studies confirm these findings and integrate them with clinical data, routine screening images could one day support non‑invasive tools that estimate inherited risk and help decide who should be offered genetic testing or closer monitoring, all without changing how the mammogram itself is performed.
Citation: Pecchi, A., Sessa, G., Nocetti, L. et al. Preliminary exploration of radiomic mammographic analysis in triple negative breast cancer related to BRCA profile. Sci Rep 16, 8765 (2026). https://doi.org/10.1038/s41598-026-35774-1
Keywords: triple negative breast cancer, BRCA mutation, mammography, radiomics, radiogenomics