Clear Sky Science · en
KChIP1 splice variants modulate Kv4 channels by promoting P/C-type inactivation features
How Tiny Pores Shape the Brain’s Electrical Rhythm
Every thought, memory and movement in the brain depends on rapid electrical signals in nerve cells. These signals are finely tuned by microscopic pores—ion channels—that let charged particles flow in and out. This paper explores how subtle variations of a helper protein, KChIP1, can dramatically change the behavior of one such channel family (Kv4 channels), shifting how easily neurons can fire repeated bursts of electrical activity.
Potassium Channels as the Brain’s Brake Pedal
Kv4 channels carry a so‑called A-type potassium current that turns on and off quickly in the cell body and branches of neurons. This current helps set how easily a neuron fires and how faithfully it can follow incoming signals, especially at low firing rates. Kv4 channels do not act alone: they assemble with helper proteins in a three-part complex that includes DPP proteins and KChIP proteins. These partners influence how fast the channels shut off and how quickly they become ready to open again. Until now, most work suggested that KChIPs generally make Kv4 channels recover more quickly after they switch off, allowing neurons to respond rapidly to repeated inputs.
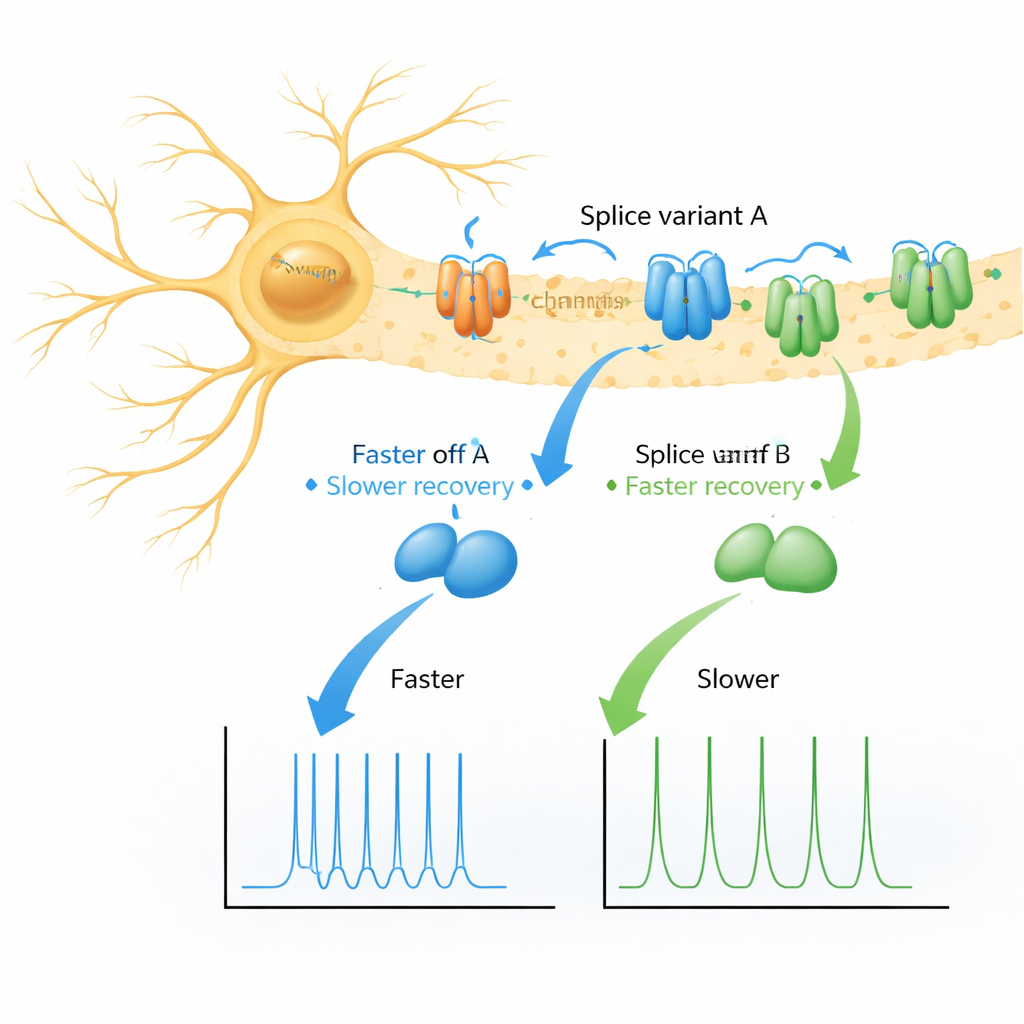
Two Protein Variants, Two Flavors of Recovery
The authors focused on two nearly identical versions (splice variants) of KChIP1, called 1a and 1b, which differ only by a short aromatic “tail” at the beginning of 1b. Using frog egg cells as a controlled testbed, they expressed several Kv4 channel types alone, with each KChIP1 variant, with DPP, or with both helpers together. As expected, both 1a and 1b gently reshaped how the channels switched off during a voltage step, without doing anything dramatic. The surprise came when they examined how channels recovered after being switched off: instead of a single, smooth return to readiness, channels paired with either 1a or 1b showed two distinct recovery phases—a fast one and a much slower one, with the slow route far more pronounced for 1b.
A Hidden Slow Pathway Revealed
When Kv4 channels were expressed without KChIP1, they recovered along a simple, fast pathway, and DPP made this even quicker. Adding KChIP1 changed that pattern. With 1a, most channels still recovered quickly, but a small fraction took a slow detour back to the ready state. With 1b, a much larger fraction entered this slow route, stretching recovery into seconds. This effect appeared in all Kv4 subtypes tested and persisted even when DPP was present, indicating that the slow pathway is a built-in feature of KChIP1-containing complexes, not an artifact of a particular channel or experimental setup. The authors also found that 1b shifted the “turn-off” voltage range of the channels to more negative values, further biasing them toward being unavailable during ongoing activity.
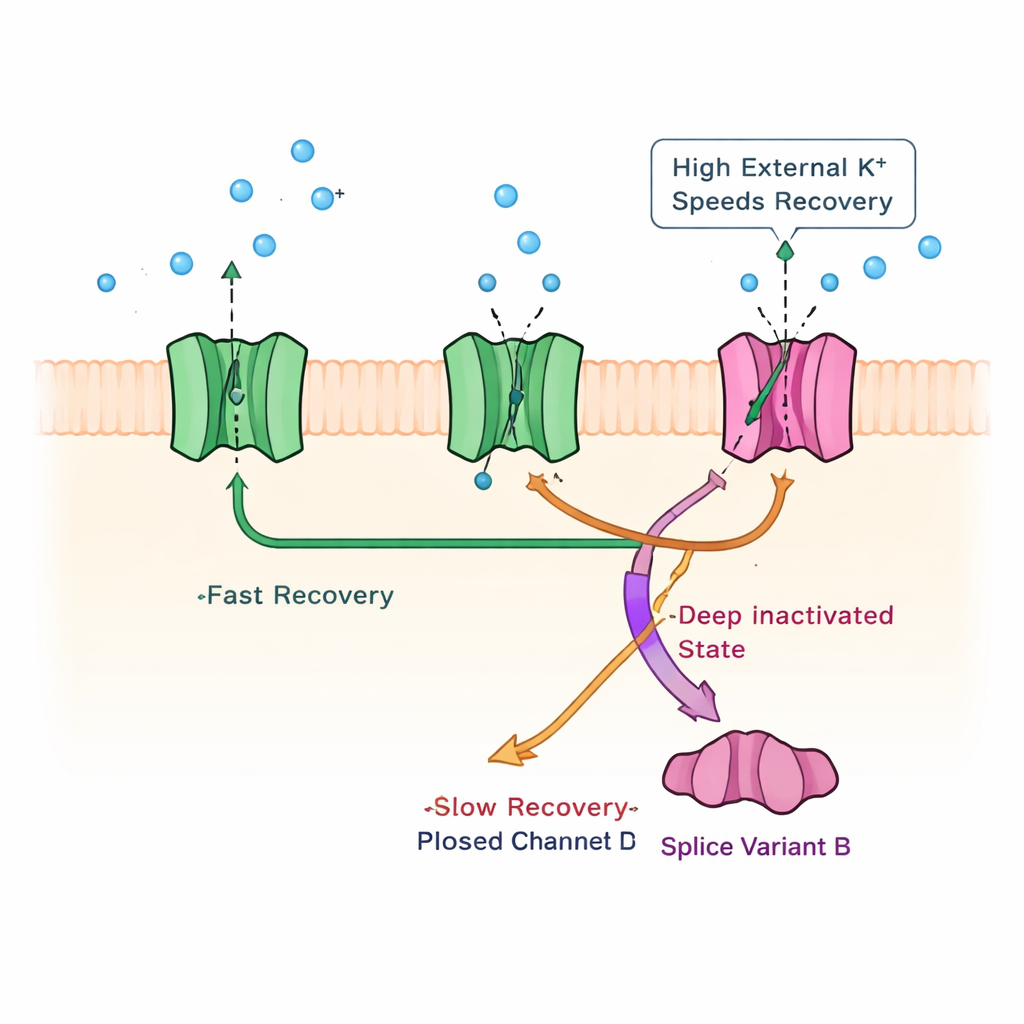
Uncovering a Dormant Inactivation Mechanism
To understand what physical change inside the channel underlies this slow recovery, the team probed known inactivation mechanisms from other potassium channels. They ruled out a classic “ball-and-chain” block at the channel’s inner mouth by cutting off part of the channel’s internal tail; the slow phase persisted. They then used high levels of external potassium, a trick known to alter a pore-based inactivation process in other channels. Under these conditions, current shutoff became faster, but, crucially, the slow recovery phase itself sped up specifically, as if the high potassium were releasing channels from a deep, long-lasting blocked state. Strategic mutations in a gating segment of the channel further supported the idea that KChIP1b promotes a pore-centered inactivation route—normally weak in Kv4 channels—that coexists with their usual, faster closed-state inactivation.
Why This Matters for Brain Activity
The findings suggest that by choosing between KChIP1a, KChIP1b, or mixtures of both, neurons can finely tune how quickly A-type currents recover between spikes. In cells rich in KChIP1b, a significant fraction of Kv4 channels get “parked” in a deep, slowly reversing inactivated state, limiting their availability during rapid firing. This may allow certain interneurons—cells that coordinate and restrain activity in brain circuits—to fire at higher frequencies or with distinct timing patterns. In essence, a small splice difference in one helper protein unlocks a hidden braking mode in potassium channels, adding a new layer of flexibility to how the brain controls its own electrical rhythms.
Citation: Cao, W., Tachtsidis, G. & Bähring, R. KChIP1 splice variants modulate Kv4 channels by promoting P/C-type inactivation features. Sci Rep 16, 2632 (2026). https://doi.org/10.1038/s41598-026-35770-5
Keywords: Kv4 potassium channels, KChIP1 splice variants, A-type current, neuronal excitability, channel inactivation