Clear Sky Science · en
Device-driven cyclic compression of the superior vena cava as a preload reduction strategy to improve cardiac function in heart failure: a pilot feasibility study
Why easing the heart’s workload matters
Heart failure is a common and serious condition in which the heart cannot pump enough blood to meet the body’s needs. Many patients land in the hospital with fluid-filled lungs, swollen legs, and crushing fatigue because blood is backing up into their circulation. Current drugs can help remove excess fluid and relax blood vessels, but they often stop working well over time or cause side effects such as low blood pressure and kidney problems. This study explores a very different idea: using a small device to gently squeeze a large vein that feeds blood into the heart, giving the failing heart a rhythmic break from constant overload.
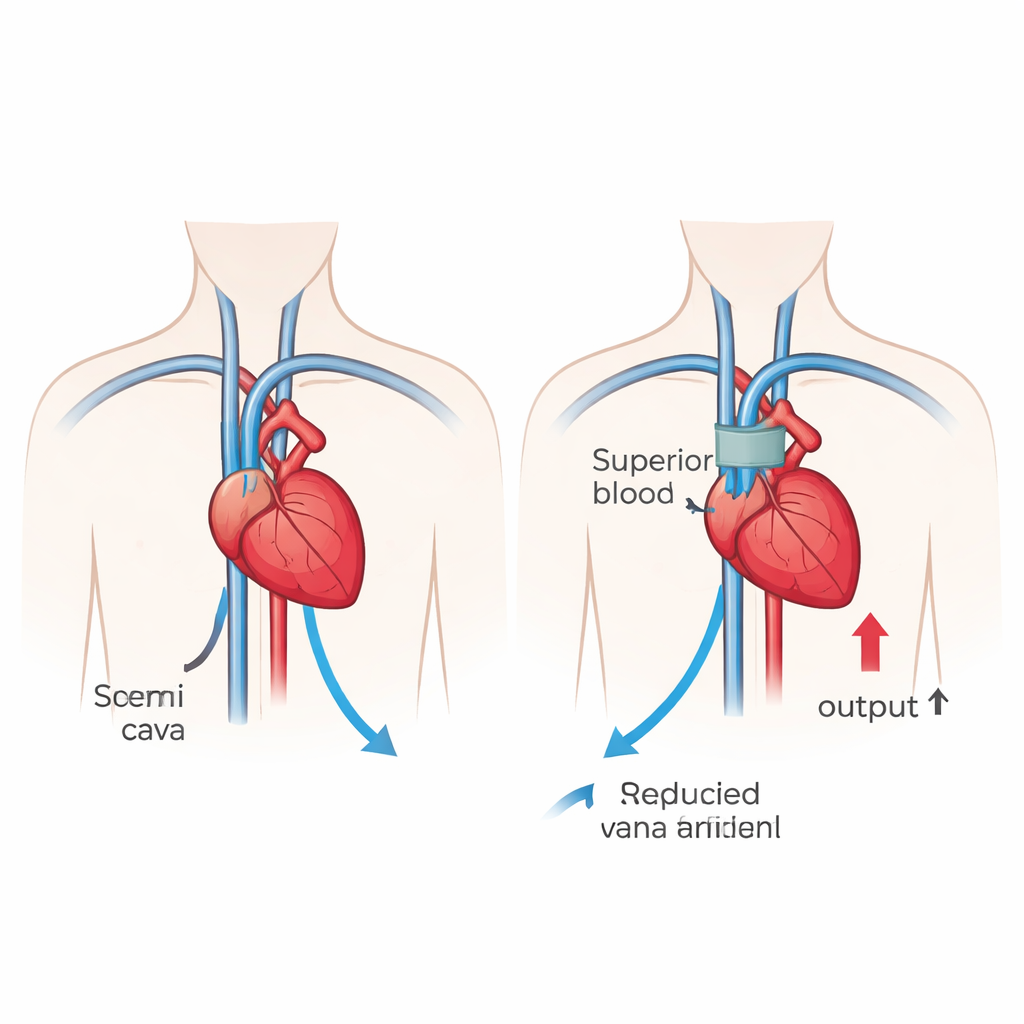
A new way to help a tired heart
The researchers focused on the superior vena cava, a large vein that returns blood from the upper body to the heart. In heart failure, that returning blood—known as preload—can overwhelm a weakened heart, worsening congestion in the lungs and body. Instead of relying on drugs to lower this load, the team created a 3D-printed clip that sits outside the vein and can be opened and closed. When closed, it narrows the vein and temporarily slows blood flow into the heart; when opened, it lets blood pass freely again. By cycling between these two states, the device aims to reduce stress on the heart without permanently blocking blood flow or entering the bloodstream.
Testing the idea in a living heart
To see whether this approach could really help, the team tested it in pigs, whose hearts and circulation are similar in size and function to those of humans. First, they induced heart failure by briefly blocking and then restoring blood flow in a coronary artery, mimicking a heart attack followed by early healing. After one to three weeks—long enough for the hearts to weaken and remodel—they surgically placed the compression device around the superior vena cava. Thin catheters and specialized sensors measured blood pressure, blood flow, and the heart’s pumping performance before, during, and after compression cycles.
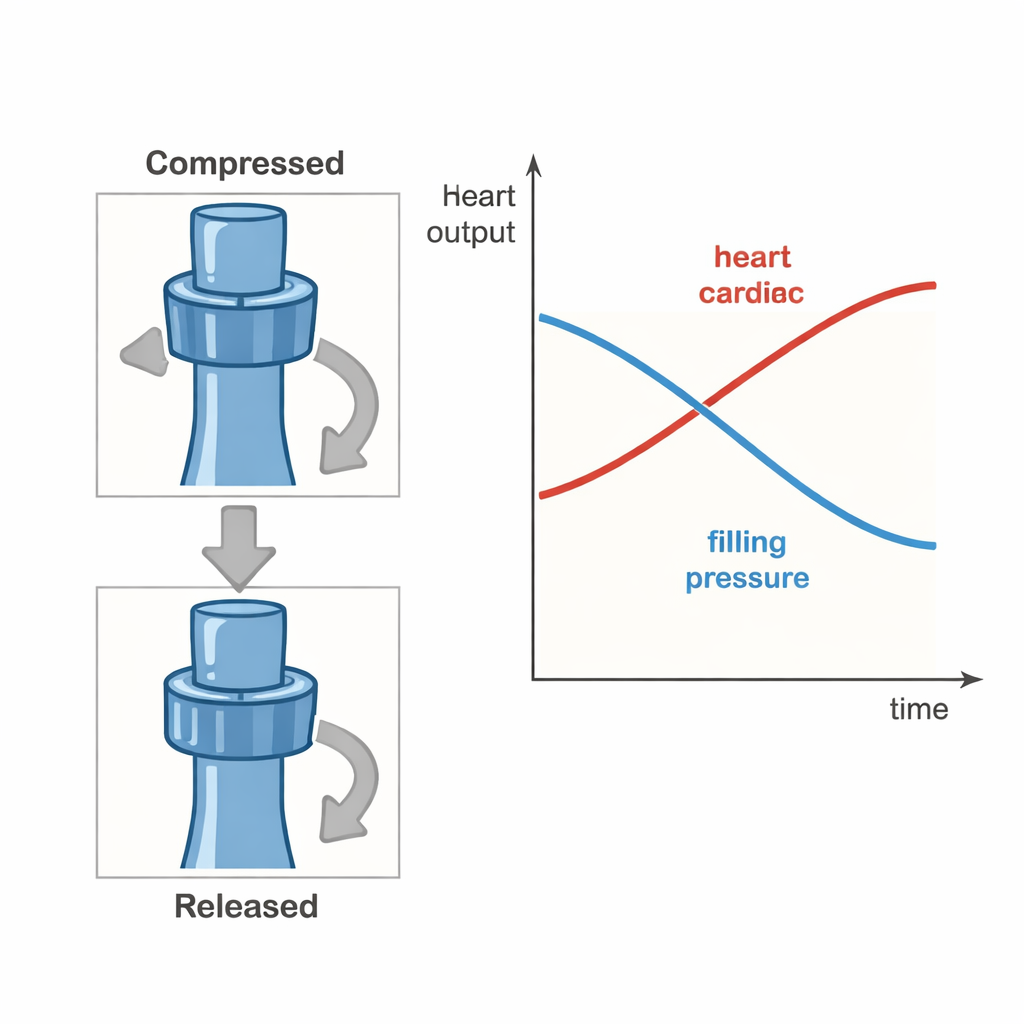
Finding the sweet spot for gentle squeezing
The key question was how much and how long to squeeze the vein. The team tried different levels of narrowing (70%, 85%, or complete closure) and patterns of compression. A continuous squeeze at 70% initially improved the heart’s output but soon caused it to fall again, suggesting that unbroken pressure was too much for the system. Complete closure in short cycles produced only modest gains and dropped blood pressure. The most promising results came from a protocol that narrowed the vein by about 85% for 20 minutes, followed by 5 minutes of full release, repeated several times. Under this pattern, average stroke volume (the amount of blood pumped with each beat) rose by about 20%, and total cardiac output increased by more than a quarter, without causing dangerous changes in blood pressure or lung pressures.
Stable body function and brain safety
Because the superior vena cava also drains blood from the head, a major concern was whether squeezing it would raise pressure in the brain. In a subset of animals, the team measured pressure in the fluid around the spinal cord as a stand-in for brain pressure. Even during strong compression at the 85% setting, brain-related pressures stayed within normal ranges. Blood tests also showed normal oxygen levels, acid–base balance, and lactate, all signs that vital organs continued to receive enough blood and oxygen throughout the cycles. These observations suggest that, at least in this short-term animal model, the device could unload the heart without harming the brain or other organs.
What this could mean for patients
Although this was a small pilot study in animals, the results hint at a future in which heart failure care includes smart, adjustable hardware alongside drugs. Cyclic squeezing of the superior vena cava appeared to lower the heart’s filling pressure while actually improving how much blood it pumped, and it did so without major instability. The authors envision a fully implantable version of the device, possibly equipped with sensors and automatic controls, that could fine-tune vein compression in real time based on a patient’s condition. For people with advanced heart failure—especially during surgery or severe flare-ups—such a reversible, non-blood-contacting device might offer a new way to protect an overworked heart and buy time for recovery. Much larger and longer studies in animals and, eventually, in humans will be needed, but this work lays the technical and physiological groundwork for that next step.
Citation: Kim, J., Kim, Y., Lee, J. et al. Device-driven cyclic compression of the superior vena cava as a preload reduction strategy to improve cardiac function in heart failure: a pilot feasibility study. Sci Rep 16, 4976 (2026). https://doi.org/10.1038/s41598-026-35769-y
Keywords: heart failure, venous compression, cardiac preload, superior vena cava, device-based therapy