Clear Sky Science · en
The role of the α7 nicotinic acetylcholine receptor in promoting M2 macrophage polarization at inflammatory sites
How Nerves Help Calm Runaway Inflammation
When we cut a finger or fight an infection, our bodies launch inflammation to protect us. But if that reaction rages too long, it can damage healthy tissue and drive chronic diseases. This study explores how a specific "calming" receptor on immune cells, called the α7 nicotinic acetylcholine receptor (α7nAChR), helps push inflammation toward healing rather than harm, offering clues for new treatments for conditions like sepsis, bowel disease, and arthritis.
The Two Faces of Immune Clean-Up Crews
Macrophages are immune cells that act as clean-up crews and repair teams. They can switch between two main modes. In their "attack" mode, often called M1, they pour out aggressive chemicals to kill germs and clear debris. In their "healing" mode, known as M2, they release soothing signals that quiet inflammation and promote tissue repair. A healthy response starts with more M1 cells and then gradually shifts toward M2 cells as the danger passes. The authors wanted to know whether α7nAChR, a receptor originally famous for its role in nerve communication and nicotine’s effects on the brain, also helps steer macrophages toward this healing M2 state during inflammation.
A Nerve-Linked Switch Toward Healing
To probe this, the researchers used mice that either had or lacked the α7nAChR and triggered inflammation in the abdominal cavity in two ways: with a bacterial component (mimicking infection) and with gentle intestinal handling (mimicking sterile surgical injury). They measured molecular markers that distinguish M1 from M2 behavior and used flow cytometry to count the proportions of each macrophage type. In normal mice, the early phase of inflammation was dominated by M1 signals, but over the next one to two days, markers of M2 cells rose, reflecting a natural shift into repair. In mice missing α7nAChR, however, pro-inflammatory markers were higher, healing markers were lower, and the proportion of M2 macrophages in the inflamed area was consistently reduced, tilting the local balance toward an M1-heavy, more damaging state.
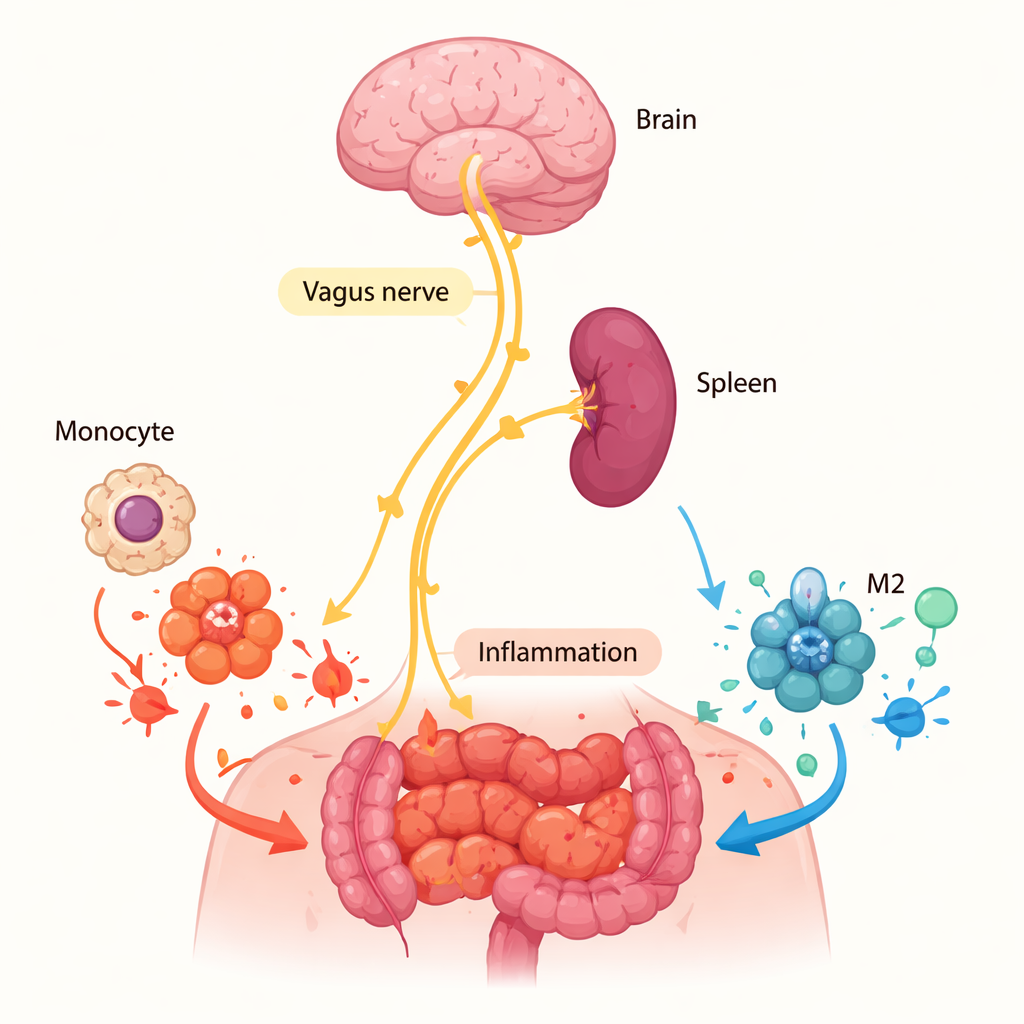
Why the Spleen Matters More Than the Wound
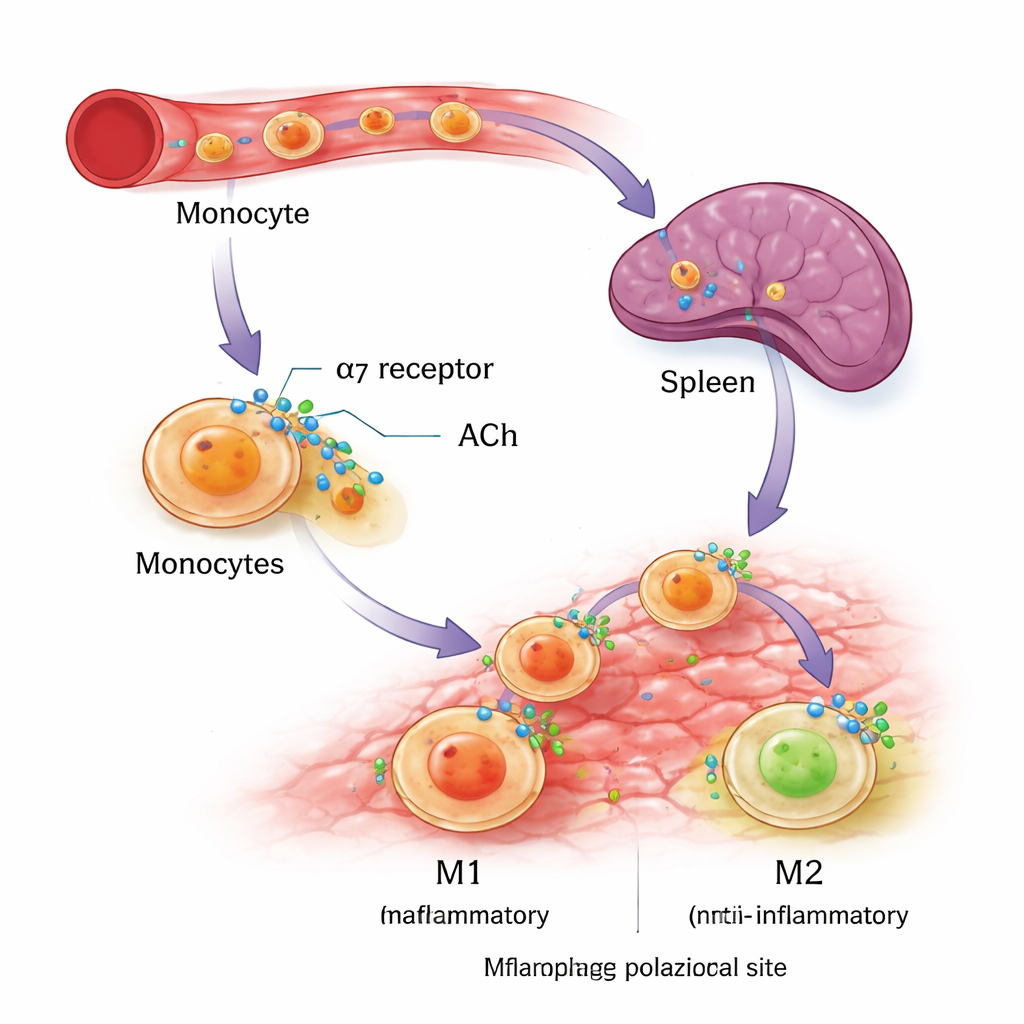
The team next asked where α7nAChR was doing this work. It might have acted directly at the inflamed site if local cells were releasing acetylcholine, the chemical that activates the receptor. But measurements of acetylcholine in abdominal fluid and in cell cultures from the inflamed tissue came up essentially empty, arguing against a strong local signal. Instead, attention turned to the spleen, a key immune organ already known to participate in a "cholinergic anti-inflammatory pathway" controlled by the vagus nerve. When the researchers surgically removed the spleen from normal mice and then induced abdominal inflammation, the proportion of M2 macrophages in the peritoneal cavity dropped, and overall macrophage numbers fell. This pattern mirrored what they saw in mice lacking α7nAChR, suggesting that nerve-driven signals in the spleen prime monocytes—the precursors of macrophages—to become M2 cells before they even arrive at the inflamed tissue.
Testing the Switch in Human Cells
To see whether the same receptor can tune human cells, the scientists turned to lab-grown monocytes from a leukemia cell line (THP-1) and from donated human blood. They guided these cells to develop into either M1 or M2 macrophages using standard immune signals and then added a specific α7nAChR-activating drug. Across both human cell sources, turning on α7nAChR did not boost M1 markers but clearly increased key hallmarks of M2 behavior, including the surface protein CD206 and the anti-inflammatory molecule interleukin-10. These experiments support the idea that α7nAChR acts like a biasing switch, making it easier for developing macrophages to adopt a healing identity without simply shutting down the immune response altogether.
From Nicotine Clues to Future Therapies
The findings help explain several puzzling observations, such as why stimulating the vagus nerve can improve inflammatory diseases and why smokers, despite many health risks, appear to have a somewhat lower risk of certain bowel conditions—nicotine can activate α7nAChR. Rather than just blocking inflammatory chemicals, α7nAChR seems to help the body convert its immune clean-up crews into more repair-focused M2 cells, particularly through spleen-based training of monocytes. For a layperson, this means our nervous system does more than sense pain or control muscles; it also quietly coaches immune cells to know when to fight and when to heal. Learning to safely flip this built-in switch with drugs or targeted nerve stimulation could open new ways to calm harmful inflammation while preserving the body’s ability to defend itself.
Citation: Mihara, T., Tanabe, H., Nonoshita, Y. et al. The role of the α7 nicotinic acetylcholine receptor in promoting M2 macrophage polarization at inflammatory sites. Sci Rep 16, 5267 (2026). https://doi.org/10.1038/s41598-026-35757-2
Keywords: macrophage polarization, inflammation, vagus nerve, nicotinic acetylcholine receptor, immune regulation