Clear Sky Science · en
Physiologically relevant forms of Tc- and Re-pyrophosphate radioactive tracers and the basis of their transthyretin amyloid sensitivity
Why this heart imaging story matters
Many people develop silent deposits of misfolded proteins, called amyloids, in the heart as they age. Some of these deposits, especially those made from a blood protein named transthyretin, can stiffen the heart and lead to serious disease. Doctors increasingly rely on a radioactive tracer known as technetium‑99m pyrophosphate (99mTc‑PYP) to see these deposits on scans. Yet, surprisingly, the exact form of this tracer in the body and why it seems to “prefer” certain amyloid types has not been clear. This paper combines theory and experiment to uncover what the tracer really looks like in living conditions and how that shape may let it home in on harmful transthyretin fibrils.
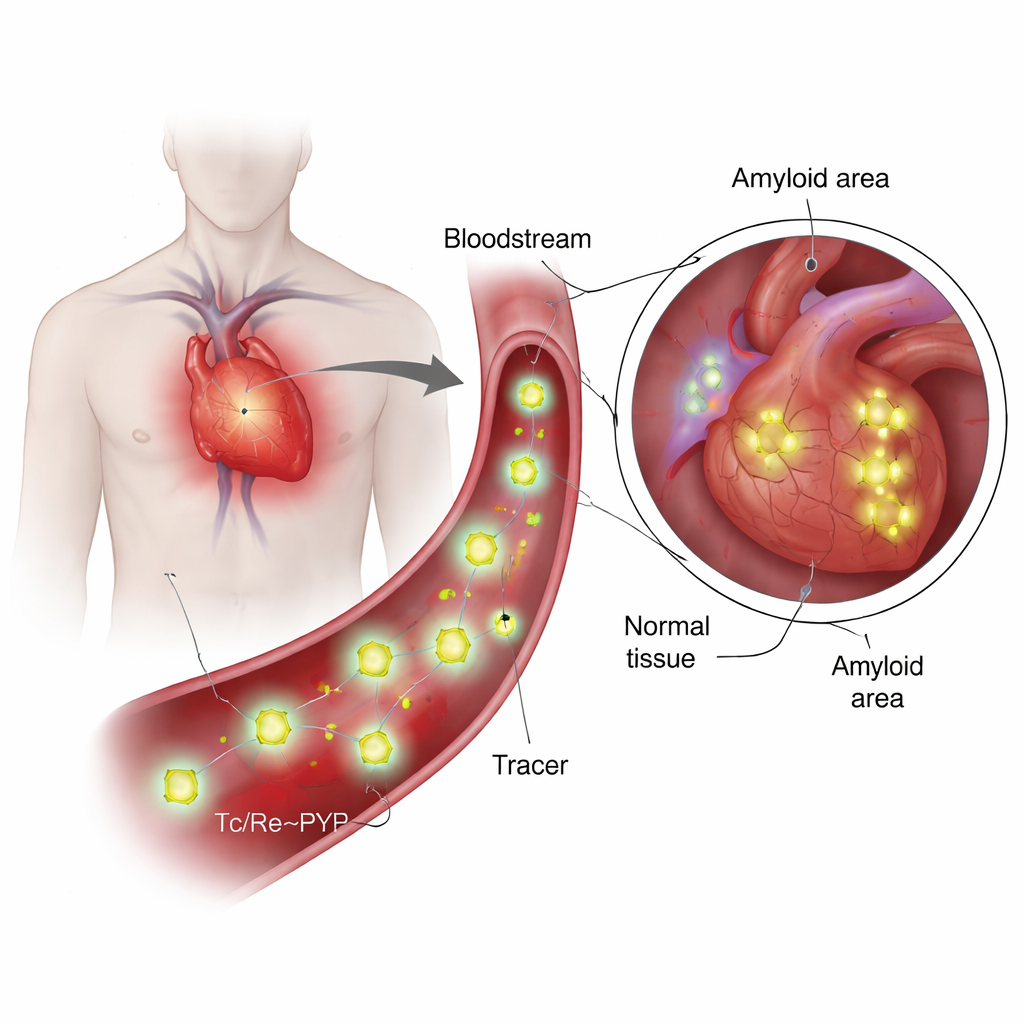
What are these tracers and why are they special?
99mTc‑PYP has been used for decades to image bone, because it tends to collect where calcium and mineral turnover are high. More recently, doctors discovered that it can also distinguish between two major kinds of cardiac amyloidosis: one built from antibody light chains (AL) and one from transthyretin (ATTR). In ATTR, the heart often lights up strongly on PYP scans, while AL cases tend to remain dim, even when calcium buildup looks similar. This mismatch raised a key question: is the tracer sticking only to calcium, or is it interacting directly with the amyloid protein itself? Answering that requires knowing the tracer’s true chemical structure under blood‑like conditions, something that earlier work had only sketched in broad strokes.
Using a safer stand‑in to see the invisible
Because technetium is radioactive and present only in tiny amounts in medical preparations, it is hard to study directly with many lab techniques. The authors therefore used rhenium, a closely related element with almost identical size and bonding preferences but more convenient chemistry, as a stand‑in. They prepared rhenium‑pyrophosphate mixtures under conditions that mimic clinical PYP kits and then probed them with a battery of tools: high‑level quantum chemistry calculations, UV‑visible light absorption, various vibrational spectroscopies (infrared and Raman), nuclear magnetic resonance, mass spectrometry, and tin Mössbauer spectroscopy. Together, these methods allowed them to test many candidate structures and to narrow down which species are likely to exist at neutral pH, like that of blood.
A flexible but recognizable molecular shape
The combined evidence points to a common “core” structure: an octahedral complex in which a technetium or rhenium atom in the +4 oxidation state is bound to two pyrophosphate groups and two water molecules. In simple terms, the metal sits at the center of an almost octahedral cage formed by oxygen atoms, with the pyrophosphates acting as multi‑toothed anchors and the water molecules occupying the remaining positions. This basic diaqua dipyrophosphate unit is not rigid. Because the pyrophosphate arms can twist and form internal hydrogen bonds with the bound waters, the molecule samples many slightly different shapes in solution. Calculations and spectra suggest that these variations shift its light‑absorption and vibrational fingerprints, explaining why experimental bands are broad and why earlier studies struggled to pin down a single clean structure.
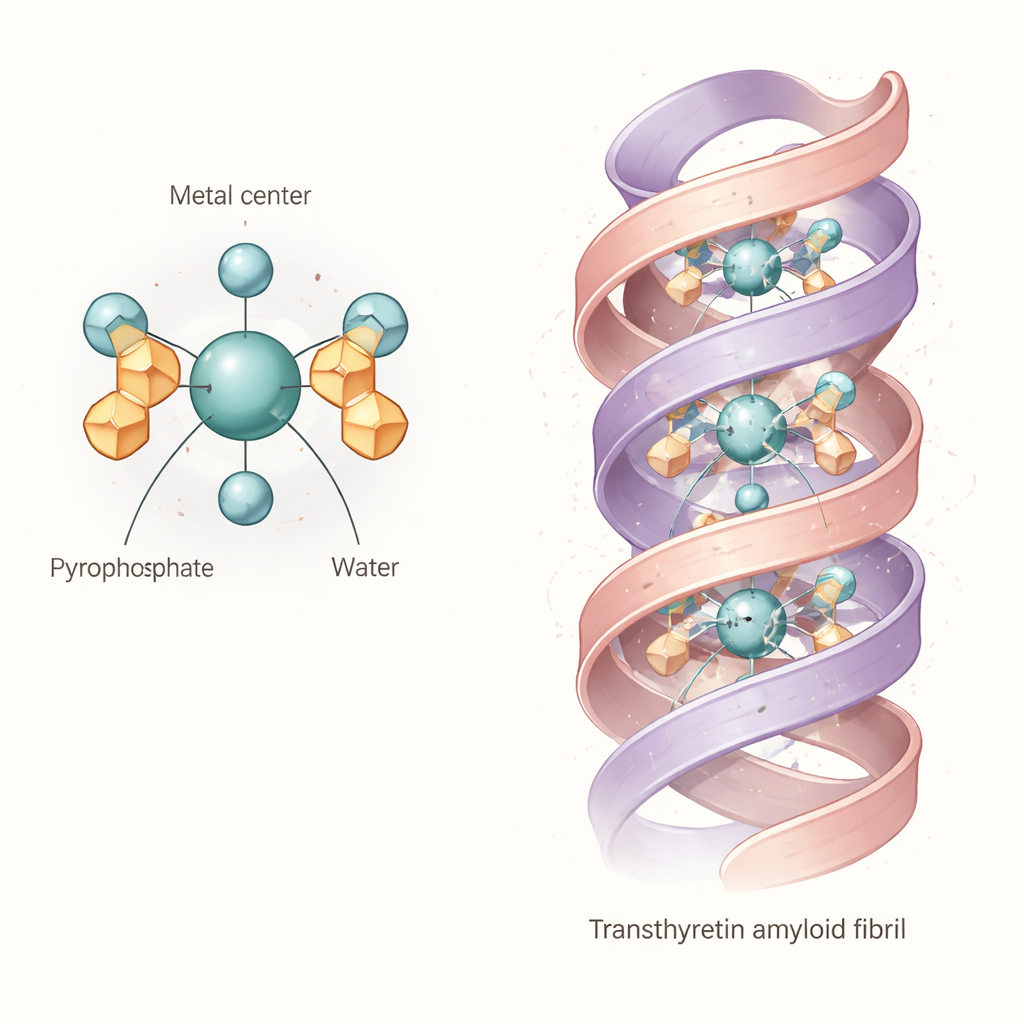
What this means for binding to heart amyloid
The authors then asked whether this flexible complex could plausibly nestle directly into transthyretin amyloid fibrils. Using a detailed cryo‑electron microscopy structure of a human transthyretin fibril, they performed computer docking searches with the modeled technetium‑pyrophosphate complex. The results show that the diaqua dipyrophosphate unit can fit into a central channel running along the fibril, making multiple hydrogen bonds and salt bridges with charged side chains lining the cavity. This suggests that, at least for some transthyretin fibril shapes, the tracer is not just tagging mineral deposits nearby; it may be clasped directly by the protein scaffold itself. The structural “give” in the tracer likely helps it adapt to slightly different pockets and charge patterns in real patient fibrils.
Implications for diagnosis and future tracers
To a lay reader, the takeaway is that the widely used PYP heart scan is based on a tracer that is more subtle and protein‑aware than previously appreciated. Under physiological conditions, it is best thought of as a small, water‑bearing metal–pyrophosphate cage that can flex and form multiple contact points with transthyretin amyloid channels. This insight helps explain why the tracer shows strong signals in some amyloid diseases but not others, and why tiny changes in the protein or its environment might cause puzzling losses of sensitivity. By clarifying the tracer’s working shape and charge pattern, the study lays a foundation for designing next‑generation imaging or therapeutic agents that more selectively recognize disease‑causing fibrils in the heart and beyond.
Citation: Simon, K.Z., Béres, K.A., Farkas, A. et al. Physiologically relevant forms of Tc- and Re-pyrophosphate radioactive tracers and the basis of their transthyretin amyloid sensitivity. Sci Rep 16, 6111 (2026). https://doi.org/10.1038/s41598-026-35746-5
Keywords: cardiac amyloidosis, transthyretin, technetium pyrophosphate, molecular imaging, radiotracer chemistry