Clear Sky Science · en
Rapid visual detection of Treponema pallidum using the RPA-CRISPR/Cas12a system
Why this matters for everyday health
Syphilis, a sexually transmitted infection once thought to be under control, is again on the rise worldwide. It can quietly damage the heart, brain, and unborn babies if it goes undetected. Yet today’s lab tests are not ideal for quick screening in clinics, rural areas, or busy emergency rooms. This paper describes a new one-hour, easy-to-read DNA test for the syphilis germ, Treponema pallidum, that aims to bring accurate diagnosis closer to the bedside and to low-resource settings.
A growing problem that is hard to spot
Syphilis is caused by a corkscrew-shaped bacterium that spreads mainly through sexual contact and from mother to child during pregnancy. Global reports counted about 8 million new cases in 2022 and 1.5 million cases of congenital syphilis in 2023, numbers that signal serious gaps in screening and treatment. Most current tests do not look for the germ itself; instead, they detect antibodies—the body’s response to infection. These blood tests are widely used because they are cheap and simple, but they can miss very early infections, give false positives in some conditions like pregnancy or autoimmune disease, and often stay positive for life. That means they cannot easily tell doctors whether a patient has an active infection or one that was cured long ago.
Building a faster DNA-based test
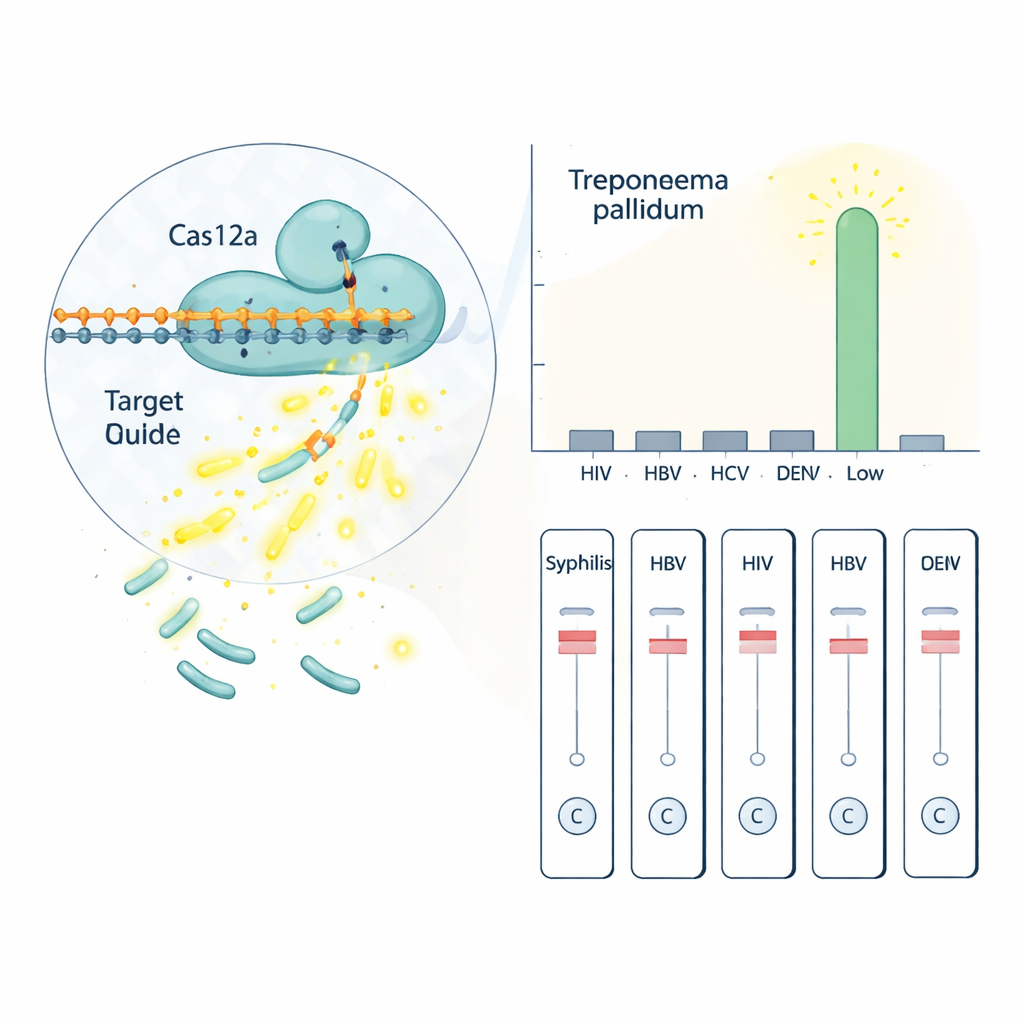
To solve these problems, the researchers created a test that directly detects syphilis DNA. They focused on a gene called tp47, a stable and widely used marker for this bacterium. The test combines two modern tools. The first is recombinase polymerase amplification (RPA), which quickly copies DNA at a constant, body-like temperature instead of cycling through high and low temperatures like traditional PCR machines. The second is CRISPR/Cas12a, an enzyme system best known for gene editing, but here repurposed as a highly selective sensor. When Cas12a is guided to the matching tp47 DNA sequence, it becomes activated and rapidly cuts nearby short DNA “reporter” pieces, releasing a strong fluorescent signal or triggering a line on a dipstick-like strip.
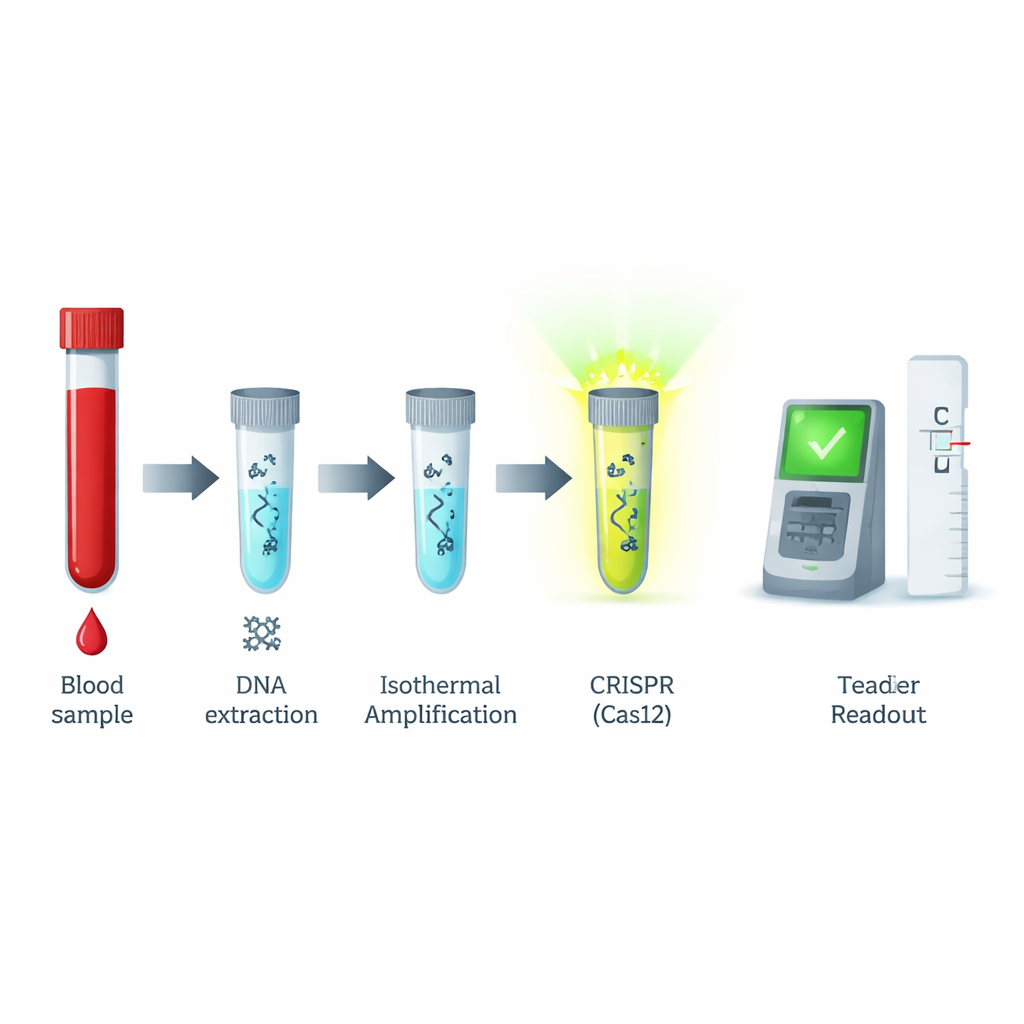
How the new test works in practice
In the lab, the team first optimized every step: the best primer pair to copy the tp47 gene, the ideal reaction times, and the amounts of the Cas12a protein, its guide RNA, and the signal-generating reporter. Under these refined conditions, the entire process—from adding the patient sample DNA to reading the result—takes about an hour. The fluorescence version of the test could reliably detect as few as about 11 copies of the target DNA per microliter, an extremely low level. It also distinguished syphilis DNA from the genetic material of several other blood-borne viruses that can cause similar symptoms, including HIV, hepatitis B and C, and dengue virus, with no cross-reaction.
From high-tech lab to simple strip
Recognizing that many clinics lack specialized instruments, the researchers also converted their method into a lateral flow assay—the same basic format used in home pregnancy tests and many rapid COVID-19 tests. After the RPA and CRISPR steps, the reaction mixture is applied to a small strip. If tp47 DNA is present, a test line appears alongside a control line. This strip-based version had a detection limit of about 5.56 × 10² DNA copies per microliter, slightly less sensitive than the fluorescence readout but still strong for a field test. In trials with 30 patient blood samples, the fluorescence test matched standard hospital diagnoses in 96.6% of cases, while the strip test correctly picked up positive cases but needs further tuning to reach the same performance.
What this could mean for patients
For non-specialists, the key message is that this new test looks directly for the syphilis germ’s DNA, works quickly at simple temperatures, and can give either a bright fluorescent signal in a small reader or a clear positive line on a disposable strip. That combination of speed, sensitivity, and simplicity could help doctors catch infections earlier, especially in clinics with limited equipment or in outreach programs. While more work is needed to improve the strip version and to test larger and more varied groups of patients, the platform offers a promising blueprint for rapid point-of-care syphilis diagnosis—and potentially for similar tests targeting many other infectious microbes.
Citation: Li, W., Sun, Y., Ye, M. et al. Rapid visual detection of Treponema pallidum using the RPA-CRISPR/Cas12a system. Sci Rep 16, 5120 (2026). https://doi.org/10.1038/s41598-026-35745-6
Keywords: syphilis, Treponema pallidum, CRISPR diagnostics, point-of-care testing, RPA-Cas12a assay