Clear Sky Science · en
Synergistic antipersister, efflux inhibitory & antibiofilm activities of vaginal Lactobacillus-derived postbiotics against UPEC: toward a novel therapeutic for utis
Why stubborn urinary infections matter
Urinary tract infections (UTIs) are among the most common bacterial infections, especially in women, and many people experience them again and again despite taking antibiotics. This study explores a new, antibiotic-sparing way to tackle those stubborn, returning infections: harnessing helpful vaginal bacteria and their chemical by-products to stop troublemaking E. coli before it can take hold, hide, and bounce back.
The hidden survivors behind repeat infections
Standard antibiotics can wipe out most bacteria, yet a tiny subpopulation known as “persister cells” survives by going into a dormant, low-activity state. These sleepers are not genetically resistant, but they tolerate very high antibiotic doses and can reawaken later, driving chronic and recurrent UTIs. The researchers worked with a common UTI-causing strain, E. coli UTI89, and showed that powerful antibiotics such as colistin and meropenem readily trigger these persister cells. In lab tests that mimic urine, small fractions of E. coli endured extreme antibiotic exposure, confirming how easily persisters can arise and why standard drug regimens often fail to give lasting relief.
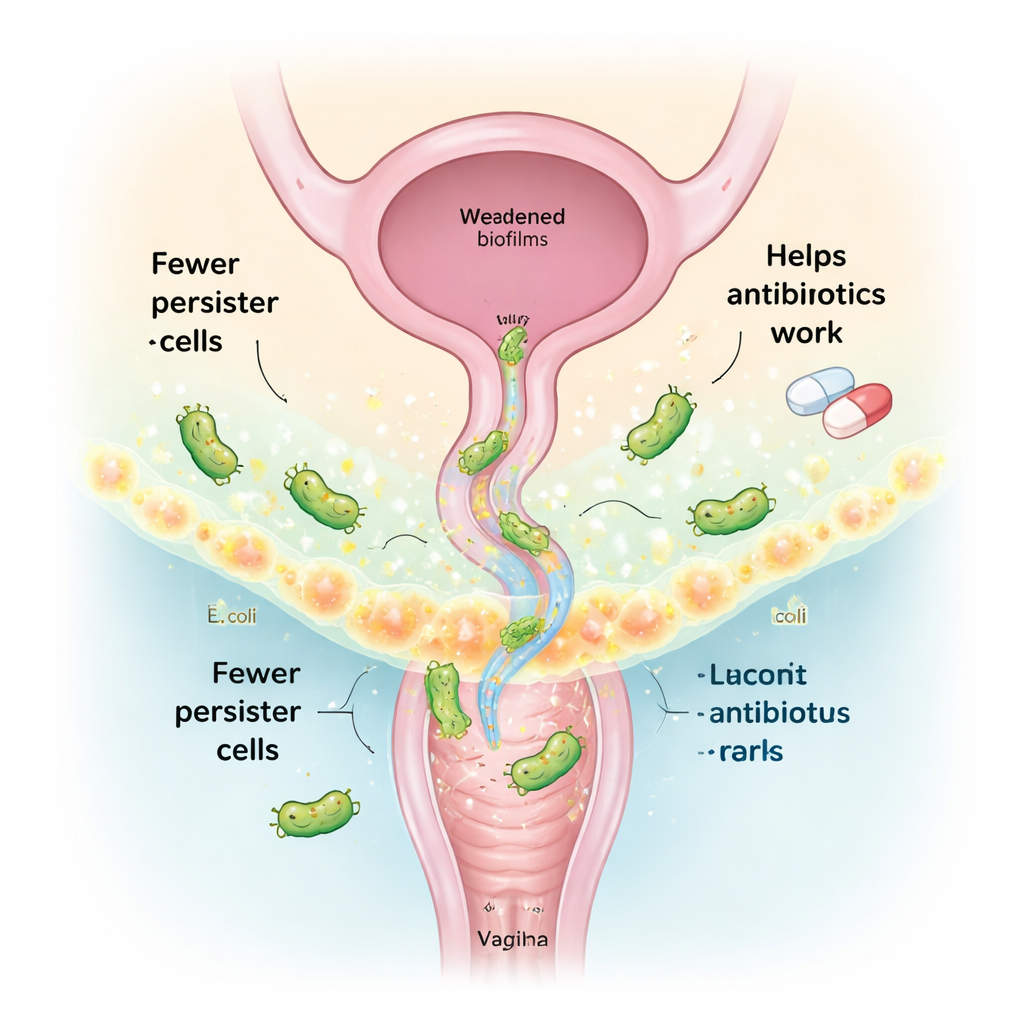
Turning friendly vaginal bacteria into a treatment
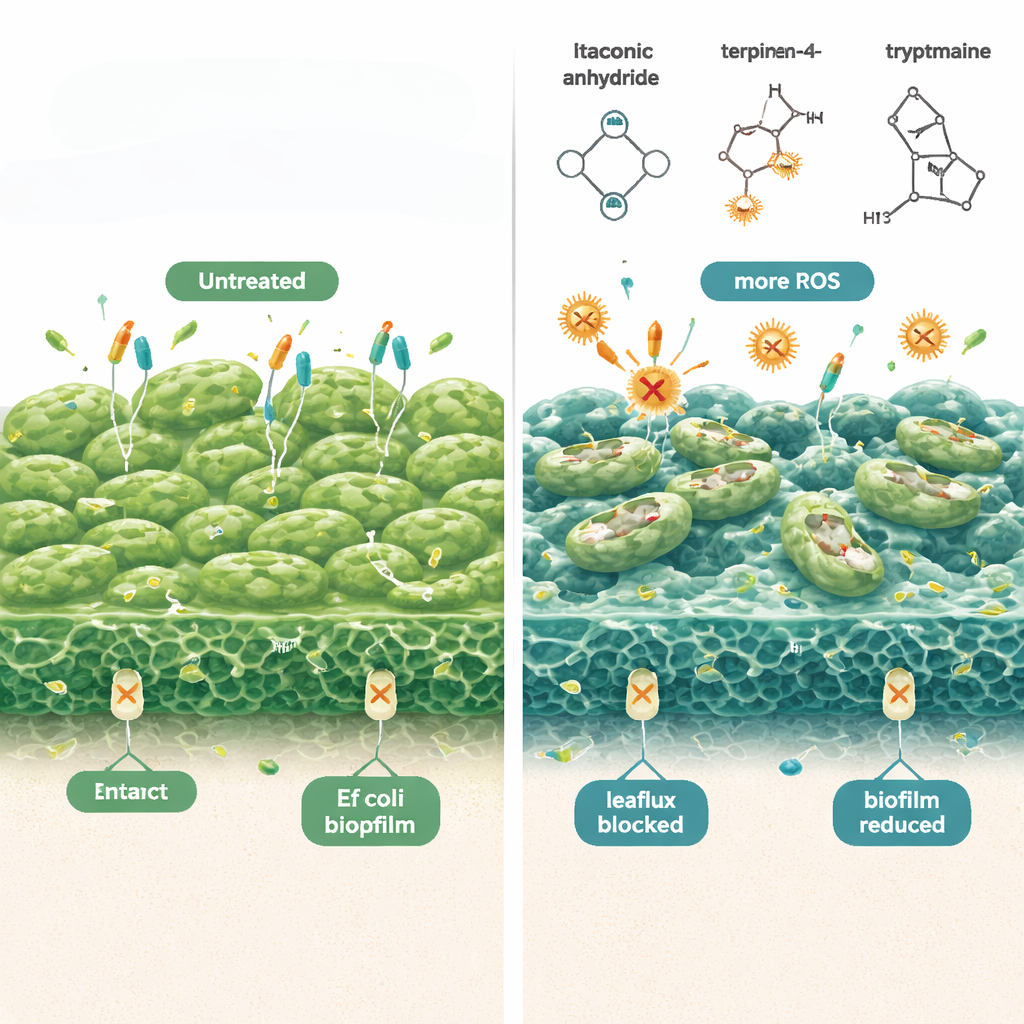
A healthy vagina is usually dominated by Lactobacillus species, which help keep harmful microbes in check. Instead of using live “probiotic” bacteria, this study focused on their cell-free supernatant—essentially the cocktail of molecules they secrete, called postbiotics. From vaginal Lactobacillus strains collected from healthy women, the team separated and analyzed these secreted compounds. Two key metabolites, itaconic anhydride and (−)-terpinen-4-ol, stood out for their ability to work together and sharply reduce the number of E. coli persister cells when combined with antibiotics. A third compound, tryptamine, previously shown to break down the slimy biofilm matrix that bacteria build for protection, was added to strengthen antibiofilm action.
How the new mixture weakens hard-to-kill bacteria
The scientists found that these Lactobacillus-derived molecules attack E. coli persisters on several fronts at once. First, they boost the production of reactive oxygen species—chemically reactive forms of oxygen that damage bacterial components—making antibiotics much more lethal to dormant cells. When antioxidants were added, this killing effect dropped, underscoring the role of oxidative stress. Second, the compounds make bacterial outer membranes more “leaky,” as shown by fluorescent dyes that enter cells more easily after treatment. Third, they inhibit cellular “efflux pumps,” tiny export machines that normally pump antibiotics back out of the cell. With pumps blocked, more drug stays inside the bacteria, and fewer persisters survive. Together, these changes reduced preformed E. coli biofilms by up to ten orders of magnitude in lab tests, without harming mammalian immune cells at the tested doses.
From lab bench to a practical vaginal wash
To turn these findings into something usable in everyday life, the team designed a customized vaginal wash using a thermosensitive gel base called poloxamer 407. At room temperature it behaves like a fluid for easy application, then gently thickens at body temperature to improve contact with the vaginal walls. The gel carries a defined, safe combination of itaconic anhydride, (−)-terpinen-4-ol, and tryptamine. In lab plates, this formulation worked over a range of pH values similar to vaginal conditions, strongly suppressing E. coli biofilm formation and slashing the number of live bacteria by about nine orders of magnitude. It remained stable and biologically active for at least three months in cold storage and also showed broad action against other problematic bacteria such as Klebsiella, MRSA, and Pseudomonas.
Testing safety and protection in mice
Next, the researchers evaluated the wash in a mouse model of vaginal E. coli infection. Mice were inoculated with glowing (GFP-tagged) E. coli and then treated with either the new metabolite wash, a probiotic wash containing live Lactobacillus, a commercial vaginal wash, or a placebo base. Animals receiving the customized metabolite wash showed markedly less vaginal inflammation, maintained their body weight, and had dramatically lower bacterial counts in vaginal discharge compared to the other groups. Most strikingly, treated mice had no detectable E. coli in their urine, bladders, kidneys, or vaginal tissue and maintained normal kidney function, indicating that the formulation not only reduced local infection but also prevented spread to the upper urinary tract.
What this could mean for people with repeat UTIs
The study concludes that a vaginal wash built from precise metabolites made by naturally protective Lactobacillus species can weaken UTI-causing E. coli in multiple ways: shrinking persister populations, disrupting biofilms, and making standard antibiotics work better. Because the approach relies on non-living molecules rather than live bacteria or long courses of systemic drugs, it may be safer, more stable, and easier to regulate. While human trials are still needed, this metabolite-based wash points toward a future in which recurring UTIs are prevented not just by stronger antibiotics, but by smartly boosting the body’s own microbial defenses at the source.
Citation: Nair, V.G., Chellappan, D.R., Durai, R.D. et al. Synergistic antipersister, efflux inhibitory & antibiofilm activities of vaginal Lactobacillus-derived postbiotics against UPEC: toward a novel therapeutic for utis. Sci Rep 16, 5005 (2026). https://doi.org/10.1038/s41598-026-35736-7
Keywords: urinary tract infection, vaginal microbiome, Lactobacillus postbiotics, antibiotic tolerance, bacterial biofilms